Optimizing Pretreatment for sp-ICP-MS: Unveiling Silver Nanoparticle Distribution in Biological Tissues
Abstract
The rapid proliferation of engineered nanoparticles (ENPs) has heightened human exposure, yet current analytical tools cannot simultaneously quantify and characterize ENPs within biological matrices. Single‑particle inductively coupled plasma‑mass spectrometry (sp‑ICP‑MS) offers simultaneous quantification and physical‑property assessment, but its application to tissues is limited by inadequate pretreatment protocols that can alter particle integrity or reduce recovery. In this study, we systematically evaluated five solubilizing agents—sodium hydroxide (NaOH), tetramethylammonium hydroxide (TMAH), nitric acid (HNO3), hydrochloric acid (HCl), and proteinase K—using silver nanoparticles (nAg) as a model system. NaOH treatment achieved >90 % tissue dissolution while preserving particle size and minimizing ionization, outperforming the other reagents. Extending the NaOH protocol to heart, lung, spleen, and kidney homogenates confirmed its broad applicability. Finally, we applied the optimized pretreatment to assess the fate of intravenously administered nAg versus Ag+ in mouse blood and liver. sp‑ICP‑MS revealed that nAg remained largely particulate (~80 %) in the liver but ionized (~95 %) in the bloodstream, whereas Ag+ remained ionic in both compartments. These findings demonstrate that NaOH pretreatment enables accurate sp‑ICP‑MS analysis of ENPs in vivo and highlights the importance of particle‑to‑ion conversion in determining biodistribution and kinetics.
Introduction
Advances in nanotechnology have accelerated the production of engineered nanoparticles (ENPs) smaller than 100 nm, offering unique advantages such as enhanced tissue penetration and reactive surfaces. Silver nanoparticles (nAg) are widely deployed in consumer goods and pharmaceuticals, prized for their sustained Ag+ release and conductive properties. However, the same nanoscale attributes that confer functionality can also pose health risks, including blood‑brain barrier disruption and inflammatory responses. Given the ubiquity of ENPs, a rigorous assessment of both hazard and exposure—collectively defining ENP risk—remains essential.
While extensive research has focused on ENP toxicity, fewer studies have quantified actual exposure scenarios. Recent work has shown distinct intracellular distributions for nAg versus Ag+ in cultured cells, underscoring the need to differentiate particles from ions within tissues. Conventional ICP‑MS quantifies total metal content but cannot resolve particle versus ionic species, whereas TEM provides size information but lacks quantitative capability. Thus, a method that couples precise quantification with physical‑property analysis is critical for elucidating ENP biotransformation.
Single‑particle ICP‑MS (sp‑ICP‑MS) can detect individual particles, providing size estimates from peak intensity and concentration from peak frequency, while distinguishing ions via background signal analysis. Prior studies have applied sp‑ICP‑MS to environmental and commercial samples, but its use in biological tissues has been limited, largely due to inadequate pretreatment that can degrade or alter ENPs. Different solubilizing reagents vary in their ability to lyse tissues, recover ENPs, and preserve particle integrity. This study therefore evaluates and optimizes pretreatment strategies for sp‑ICP‑MS analysis of nAg in mouse tissues, aiming to establish a reliable workflow for simultaneous quantification and physical‑property assessment of ENPs in vivo.
Materials and Methods
ENPs
The 30, 70, and 100 nm Biopure nAg (nAg30, nAg70, nAg100) were sourced from nanoComposix (San Diego, CA, USA). RM8013, a standard for transport‑efficiency calibration, was obtained from the National Institute of Standards and Technology (Gaithersburg, MD, USA). All ENP suspensions were sonicated for 10 min prior to use.
Reagents
Solutions of 0.1 mol L−1 NaOH, 25 % TMAH, 30 % HCl, and proteinase K were purchased from Wako (Osaka, Japan). A 70 % HNO3 solution was obtained from Kanto Kagaku (Tokyo, Japan).
Animals
Female BALB/c mice (6 weeks old) were obtained from Japan SLC (Shizuoka, Japan). Mice were housed under a 12‑h light/dark cycle with ad libitum access to food and water. All procedures complied with the ethical guidelines of Osaka University, Japan.
Measuring Particle Size Distributions by Dynamic Light Scattering
nAg was diluted in MilliQ water to 10 µg mL−1 Ag. A 1 mL aliquot was placed in a zeta‑capillary cell (Malvern Instruments) and analyzed on a Zetasizer Nano‑ZS (Malvern Instruments) to determine mean diameter and zeta potential.
Measuring Gross Mass of Ag
Total Ag concentrations were measured on an Agilent 7700x ICP‑MS (Agilent Technologies). Operating conditions were RF power 1550 W, Ar flow 1.05 L min−1, dwell time 100 ms. Rhodium served as an internal standard. Measurements were performed in triplicate.
Analysis of sp‑ICP‑MS and Its Calculation
sp‑ICP‑MS analysis used the same instrument with RF power 1550 W, Ar flow 1.05 L min−1, dwell time 10 ms, and a total run time of 30 s. Particle size calculations employed the single‑particle tool developed by RIKILT.
Critical Particle Concentration for sp‑ICP‑MS
Stock nAg solutions (1.0 mg mL−1) were serially diluted to produce 2000, 800, 700, and 600 pg mL−1 solutions, which were further diluted tenfold to generate 40 distinct concentrations for sp‑ICP‑MS evaluation.
Optimization of Pretreatment Methods for Mouse Liver
Liver homogenates were prepared by mixing tissue with phosphate‑buffered saline (PBS, w/v = 1:10) and homogenizing. Each homogenate was spiked with 100 ng mL−1 nAg and then treated with one of the following reagents at a 1:1 v/v ratio: 0.1 mol L−1 NaOH, 25 % TMAH, 30 % HCl, or proteinase K (10 U mL−1 in 0.01 M Tris‑HCl, 0.01 M EDTA, 0.5 % SDS). Samples were incubated for 3 h at 37 °C, residues collected, weighed, and supernatants diluted 500‑fold for sp‑ICP‑MS.
Versatility Evaluation of NaOH Pretreatment in Various Organs
Heart, lung, spleen, and kidney tissues were homogenized in PBS (w/v = 1:10) and spiked with 100 ng mL−1 nAg. A 1 mol L−1 NaOH solution was added at a 1:1 v/v ratio, and samples were incubated for 3 h at 37 °C. Residues were weighed, and supernatants diluted 500‑fold for sp‑ICP‑MS.
Evaluation of Quantity and Physical Properties of nAg100 and Ag+ in Mice After Single Intravenous Administration
nAg100 and AgNO3 were diluted to 0.25 mg mL−1 (Ag+) in 5 % glucose. Mice received intravenous injections of nAg100 (1.5 or 0.75 mg kg−1), AgNO3 (1.5 or 0.75 mg kg−1 as Ag+), or 5 % glucose (control). Twenty‑four hours post‑injection, blood and liver samples were collected. Liver homogenates were prepared in PBS (w/v = 1:10) and then treated with TMAH or NaOH (1:1 v/v) for ICP‑MS and sp‑ICP‑MS analysis.
Results and Discussions
Optimization of Particle Detection by sp‑ICP‑MS
Accurate sp‑ICP‑MS requires that no more than one particle enters the plasma during each dwell time; otherwise, multiple particles are erroneously summed as a single event, inflating size estimates. We serially diluted nAg100 and plotted detected versus theoretical particle counts. While low concentrations produced a linear relationship, higher concentrations yielded fewer detected events than expected, indicating multi‑particle interference (Fig. 1a). Subtracting detected from theoretical counts revealed that deviations became significant when detected events exceeded 500, establishing a practical threshold for reliable analysis (Fig. 1b).
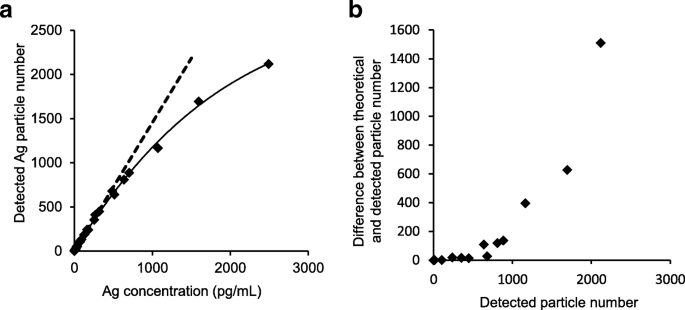
Determination of optimal particle number per dwell time for accurate sp‑ICP‑MS analysis. A series of nAg solutions (600 fg mL−1 to 2,500 pg mL−1) were analyzed by sp‑ICP‑MS. a To determine the relationship between the concentration of nAg100 and the number of detected particles, a curve for the detected particles (solid line) the theoretical values (dotted line) were plotted. b The number of detected particles subtracted from the theoretical value was plotted in the vertical axis to determine the optimal particle number. Each point is the result of a single trial (n = 1)
Validation with nAg of varying diameters confirmed that the sp‑ICP‑MS settings yielded accurate size estimates: measured diameters of 30.0 ± 1.2 nm, 65.1 ± 0.6 nm, and 97.4 ± 0.6 nm for nAg30, nAg70, and nAg100, respectively. Dynamic light scattering (DLS) measurements (36.4 ± 1.6 nm, 70.6 ± 1.7 nm, 101 ± 1.0 nm) closely matched sp‑ICP‑MS results, validating the analytical protocol.
Optimization of Pretreatment Methods for Detecting nAg in Mouse Liver Tissue
Effective pretreatment must lyse tissue, recover ENPs without altering their physical or chemical state, and avoid ionization or aggregation. We compared five solubilizing agents—NaOH, TMAH, HNO3, HCl, and proteinase K—using liver homogenates spiked with 100 ng mL−1 nAg.
Residue analysis revealed that NaOH, TMAH, and proteinase K dissolved >90 % of the liver matrix, whereas HNO3 and HCl dissolved only ~75 % (Fig. 2a). Given that ~80 % of tissue is water, the lower dissolution by acidic reagents indicates incomplete matrix removal.
sp‑ICP‑MS recovery data showed that acidic treatments fully ionized nAg, while proteinase K partially ionized it. In contrast, NaOH and TMAH preserved nAg as intact particles with negligible ion detection (Fig. 2b). Size distribution analysis indicated that TMAH treatment caused a shift from 100 nm to 120 nm and broadened the distribution (Fig. 2c‑d), suggesting aggregation. NaOH treatment maintained the original 100 nm size (Fig. 2c‑d), confirming its suitability.
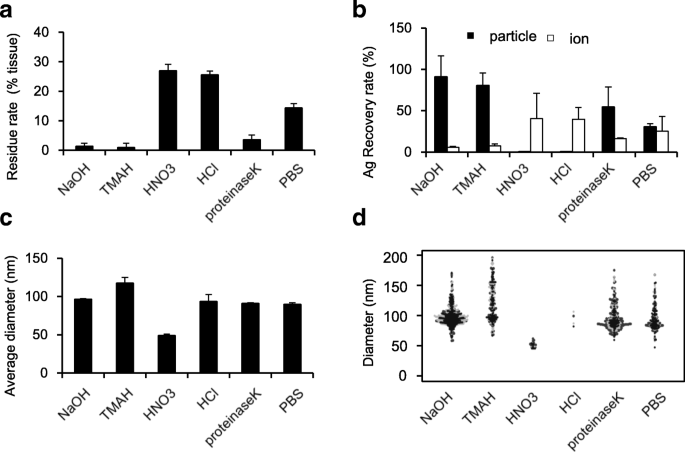
NaOH pretreatment is the optimal method for detecting nAg100 in mouse liver. Five solubilizing reagents were screened as pretreatment solvents to lyse the tissues (NaOH, TMAH, HNO3, HCl, and proteinase K). The liver homogenate was mixed with nAg100 solution to obtain a final Ag concentration of 100 ng mL−1 and treated with each solubilizing reagent at 37 °C. After 3 h, a residue rates in each group as an indicator of tissue solubility, b recovery rates (black and white bars represent the rate of silvers detected as particles and as ions, respectively), c average particle diameters shown in a bar chart, and d particle size distribution shown in a beeswarm chart were analyzed by sp‑ICP‑MS analysis. The results are expressed as mean ± SD (n = 3)
TMAH’s tendency to increase the dielectric constant through decomposition into trimethylamine and methanol can diminish the zeta potential of nAg, promoting aggregation. Short‑time (1 min) TMAH exposure, however, preserved particle size (~100 nm), confirming that prolonged treatment is responsible for aggregation.
Evaluation of Versatility of NaOH Pretreatment in Various Organs
To confirm NaOH’s broad applicability, we applied the protocol to heart, lung, spleen, and kidney homogenates spiked with nAg. Residue analysis showed >95 % dissolution (Fig. 3a). sp‑ICP‑MS detected the added Ag as particles in all tissues, with recovery rates within the 80–120 % range acceptable by FDA standards (Fig. 3b). Particle diameters remained close to 100 nm across all organs (Fig. 3c‑d), underscoring the method’s consistency.
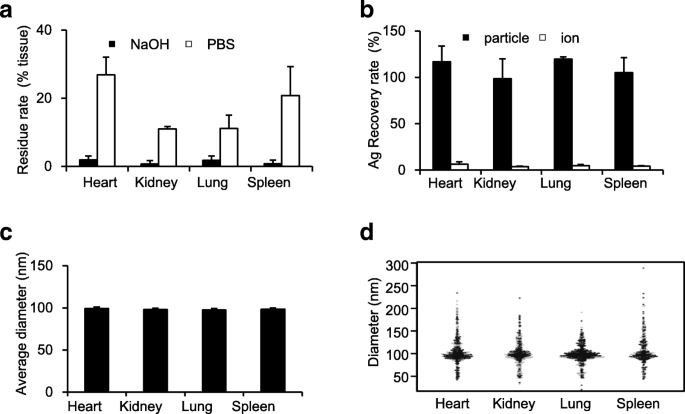
NaOH pretreatment is the optimal method for detecting nAg100 in various organs. As in Fig. 2, the heart, kidney, lung and spleen homogenates were mixed with nAg100 and incubated with NaOH solution. After 3 h, a residue rates (black and white bars represent the residue rates in NaOH- or PBS-treatment, respectively), b recovery rates (black and white bars represent the rate of Ag detected as particles and as ions, respectively), c average particle diameters shown in a beeswarm chart, and d particle size distribution shown in a beeswarm chart were analyzed by sp‑ICP‑MS analysis in each tissue sample. The results are expressed as mean ± SD (n = 3)
These data collectively confirm that NaOH pretreatment provides a robust, reproducible platform for quantifying and characterizing nAg in diverse animal tissues via sp‑ICP‑MS.
Evaluation of sp‑ICP‑MS for Quantitative and Physical Property Analyses of nAg and Ag+ in Biological Tissues
To demonstrate practical relevance, we compared the fate of intravenously administered nAg100 versus Ag+ in mouse blood and liver. ICP‑MS confirmed Ag presence in both compartments for both treatments (Fig. 4a,b). However, sp‑ICP‑MS revealed distinct speciation: in blood, Ag predominantly existed as ions (>95 %) regardless of the administered form, while in liver, ~80 % of Ag from nAg100 remained as particles, with minimal particle presence after Ag+ administration (Fig. 4c,d). Particle size analysis of liver nAg100 showed an average diameter of ~80 nm, smaller than the administered 100 nm, reflecting partial ionization and possible transformation within hepatic tissue (Fig. 4e).
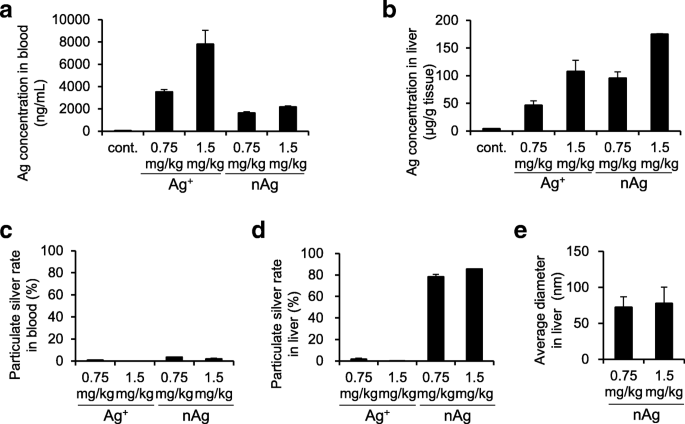
Simultaneous quantification and physical property analyses of intravenously administered nAg100 and Ag+. nAg100 and Ag+ were intravenously administrated in mice (0.75 or 1.5 mg kg−1). After 24 h, their livers and blood were collected. All samples were pretreated with NaOH solution. Ag concentration in a blood and b liver were measured by ICP‑MS. nAg in c blood and d liver were measured by sp‑ICP‑MS. The average diameter of the particles detected in the liver is shown in e. The results are expressed as mean ± SE (n = 3)
Conclusions
We have established an optimized NaOH pretreatment protocol that enables accurate sp‑ICP‑MS analysis of silver nanoparticles in mouse tissues, providing simultaneous quantification and particle‑size characterization. Application of this workflow to intravenously administered nAg100 and Ag+ revealed that nAg partially ionizes in vivo, with ~80 % remaining particulate in the liver and ~95 % ionized in the bloodstream. These findings underscore the importance of accounting for particle‑to‑ion conversion when assessing ENP biodistribution and kinetics. The method is broadly applicable to diverse tissues and can be employed in risk‑assessment studies to elucidate ENP exposure dynamics and biological responses.
Availability of Data and Materials
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Abbreviations
- Ag:
Silver
- Ag+:
Silver ion
- DLS:
Dynamic light scattering
- ENPs:
Engineered nanoparticles
- HCl:
Hydrochloric acid
- HNO3:
Nitric acid
- ICP‑MS:
Inductively coupled plasma‑mass spectrometry
- nAg:
Silver nanoparticles
- nAg100:
100 nm nAg
- nAg30:
30 nm nAg
- nAg70:
70 nm nAg
- NaOH:
Sodium hydroxide
- PBS:
Phosphate‑buffered saline
- sp‑ICP‑MS:
Single particle ICP‑MS
- TEM:
Transmission electron microscope
- TMAH:
Tetramethylammonium hydroxide
Nanomaterials
- Nanoparticle-Based Cancer Therapy: Advances, Mechanisms, and Clinical Translation
- Optimized Post‑Treatment Strategy for Monodisperse FePt–Fe3O4 Binary Nanoparticles
- Au@TiO₂ Yolk–Shell Nanostructures: Tailored Synthesis and Their Superior Visible‑Light Photocatalytic Degradation and SERS Detection of Methylene Blue
- Green Synthesis of Gold and Silver Nanoparticles with Platycodon Saponins from Platycodi Radix
- Magnesium‑Doped Zinc Oxide Nanoparticles: Optimizing Photocatalytic Degradation and Antibacterial Efficacy
- Optimizing Glycyrrhetinic Acid Liposomes via Lyophilization Monophase Solution: Preformulation, Design, and In‑Vitro Evaluation
- Controlling Silver Nanoparticle Morphology via CTAB‑Capped Seeds and Aging Time
- Reducing Cytotoxicity of Silver Nanoparticle Coatings with Graphene Oxide: Enhanced Antibacterial Performance and Biocompatibility of an Ag–GO Nanocomposite
- A Novel Analytical System for Assessing Silver Nanoparticle Physicochemical Properties in Human Skin
- Ultrasmall Superparamagnetic Iron Oxide Nanoparticles for Stem Cell Tracking in Parkinson’s Therapy



