Reducing Cytotoxicity of Silver Nanoparticle Coatings with Graphene Oxide: Enhanced Antibacterial Performance and Biocompatibility of an Ag–GO Nanocomposite
Abstract
Silver nanoparticle (AgNP)–coated surfaces are widely considered a promising alternative to conventional antibiotics. Yet, their high surface reactivity raises safety concerns in biomedical contexts. Here, we report that integrating graphene oxide (GO) into AgNP coatings substantially lowers cytotoxicity toward human fibroblasts, endothelial cells, and the chicken embryo chorioallantoic membrane, while simultaneously enhancing antibacterial activity against Salmonella enteritidis. The Ag–GO composite demonstrated superior bacterial inhibition and markedly reduced inflammatory cytokine release, indicating a safer nanoplatform suitable for medical applications.
Introduction
Antimicrobial surfaces incorporating nanomaterials—especially silver nanoparticles—are emerging as effective alternatives to traditional antibiotics and chemical disinfectants. However, the very properties that confer antibacterial potency also contribute to potential cytotoxicity and immunogenicity, limiting clinical translation. A key strategy to mitigate toxicity is to immobilise nanoparticles firmly onto the substrate, thereby restricting their release while preserving antimicrobial function. Ultrasonic deposition techniques have proven effective for coating and forming stable nanocomposites, such as AgNPs on graphene oxide (GO). GO’s abundant oxygenated functional groups promote strong electrostatic or covalent bonding with metal ions, enhancing adhesion and reducing particle leaching. In this study, we hypothesised that polyurethane foils coated with an Ag–GO composite would exhibit lower toxicity compared to AgNP‑only coatings, without compromising antibacterial efficacy.
Results
AgNPs and GO Formed a Stable Nanocomposite
Transmission electron microscopy (TEM) revealed spherical AgNPs (~55 nm) tightly adhered to GO sheets (Fig. 1a‑e). Zeta‑potential measurements showed that the Ag–GO hydrocolloid stabilized after 24 h (−15.68 mV to −27.7 mV), whereas AgNPs alone remained unstable (−31.11 mV). Dynamic light scattering (DLS) indicated a Z‑average size of 1157 nm for the composite, intermediate between AgNPs (93 nm) and GO (1485 nm), confirming successful hybridisation (Fig. 1b,d,f). Raman spectroscopy confirmed the characteristic D (1347 cm−1) and G (1578 cm−1) bands of GO, with an ID/IG ratio of 1.34, indicating a highly disordered but functionalised structure (Fig. 2a). FT‑IR spectra further verified the presence of hydroxyl, carboxyl, and epoxy groups (Fig. 2b).
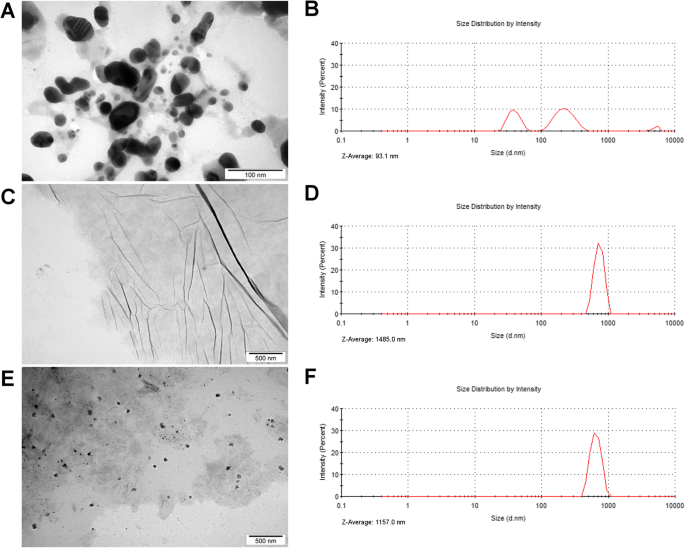
Nanoparticle morphology and size distribution. Transmission electron microscopy images of a silver nanoparticles, c graphene oxide and e silver nanoparticles and graphene oxide composite. Size distribution of b silver nanoparticles, d graphene oxide and f silver nanoparticles and graphene oxide composite
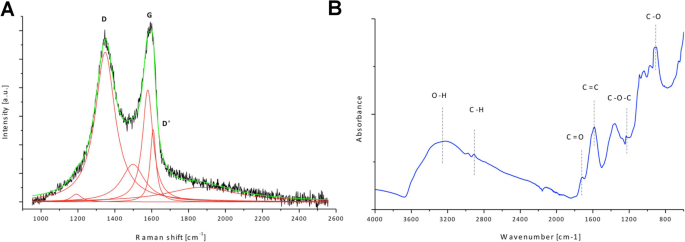
Structural feature analysis of graphene oxide. a Raman spectrum of graphene oxide with proposed deconvolution of the D, G and D’. b Fourier transform infrared spectroscopy (ATR, attenuated total reflectance) spectrum of graphene oxide with assignment of functional groups
Antibacterial Efficacy Against Salmonella enteritidis
After 24 h incubation at 37 °C, all nanoplatforms—AgNP, GO, and Ag–GO—reduced bacterial growth relative to uncoated foils. The Ag–GO composite achieved the most pronounced inhibition, with a >90 % reduction in colony‑forming units as quantified by PrestoBlue assay (Fig. 3c). Scanning electron microscopy (SEM) revealed sparse bacterial colonies and membrane disruptions on Ag–GO surfaces, confirming physical contact–mediated killing (Fig. 3b).
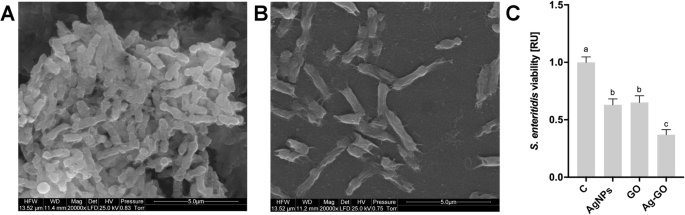
Nanoplatforms coated with silver nanoparticles and graphene oxide decreased the viability of S. enteritidis. Scanning electron microscope images of a control S. enteritidis bacteria and b S. enteritidis incubated on a silver nanoparticle- and graphene oxide-coated nanoplatform, after incubation at 37 °C for 24 h. c Viability of S. enteritidis after incubation on the nanoplatform for 24 h was assessed with a PrestoBlue assay. Values are expressed as mean ± standard deviation (n = 3, each experiment in triplicate). Statistical significance is indicated by different superscripts (one‑way ANOVA; P < 0.05). Abbreviations: C, control group (foil without nanoparticles); AgNPs, nanoplatform coated with silver nanoparticles; GO, nanoplatform coated with graphene oxide; Ag‑GO, nanoplatform coated with composite of graphene oxide and silver nanoparticles; RU, relative units
Reduced Cytotoxicity in Human Cells
Human fibroblasts and HUVECs were cultured directly on each nanoplatform for 24 h. Viability assays showed a 40–50 % decrease on AgNP‑only foils, while GO and Ag–GO surfaces maintained viability comparable to uncoated controls (Fig. 4e,f). Morphological assessment revealed that AgNP coatings induced pronounced cell aggregation, whereas GO and Ag–GO promoted normal spreading and morphology (Fig. 4a‑d). These findings underscore the protective effect of GO in the composite.
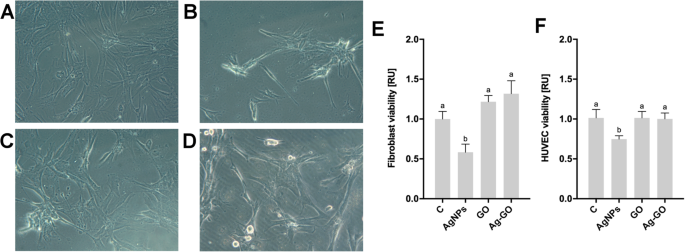
Nanoplatforms coated with graphene oxide decreased the cytotoxicity of silver nanoparticles. Morphology of fibroblasts cultured on a non‑coated nanoplatforms, b silver nanoparticle‑coated nanoplatforms, c graphene oxide‑coated nanoplatforms, d silver nanoparticles and graphene oxide composite‑coated nanoplatforms. Morphology was assessed by light microscopy using phase contrast with × 200 magnification. Fibroblast (e) and HUVEC (f) viability after 24 h of incubation on the nanoplatforms was determined using a PrestoBlue assay. Values are expressed as mean ± standard deviation (n = 3, each experiment in triplicate). Statistical significance is indicated by different superscripts (one‑way ANOVA; P < 0.05). Abbreviations: HUVECs, human umbilical vein endothelial cells; C, control group (foil without nanoparticles); AgNPs, nanoplatform coated with silver nanoparticles; GO, nanoplatform coated with graphene oxide; Ag‑GO, nanoplatform coated with a composite of graphene oxide and silver nanoparticles; RU, relative units
Chorioallantoic Membrane Compatibility
Direct contact of the nanoplatforms with the chicken embryo chorioallantoic membrane for 48 h revealed that AgNP‑only coatings caused capillary loss and surface roughening, whereas GO and Ag–GO maintained a smooth, vascularised morphology similar to controls (Fig. 5b). This in‑vivo‑like model corroborates the reduced endothelial toxicity observed in vitro.
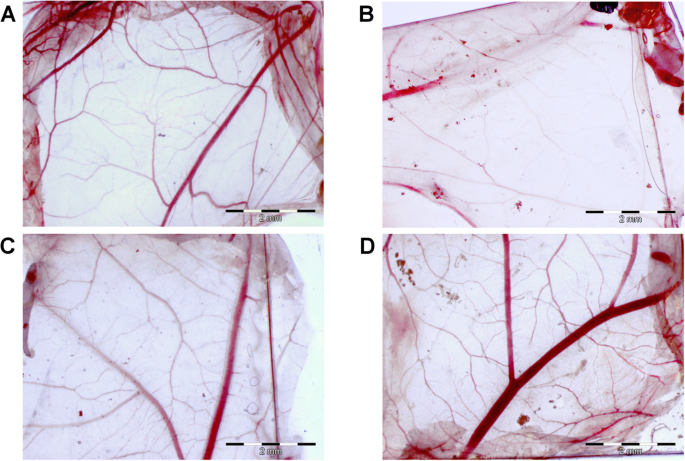
Graphene oxide decreased the morphological changes of the chorioallantoic membrane caused by silver nanoparticles. The morphology of the chicken embryo chorioallantoic membrane after 48 h of incubation with the nanoplatforms. a Control group (foil without nanoparticles), b nanoplatform coated with silver nanoparticles, c nanoplatform coated with graphene oxide, d nanoplatform coated with composite of graphene oxide and silver nanoparticles
Modulation of Inflammatory Cytokine Release
Antibody array profiling of fibroblast supernatants revealed that both AgNP and Ag–GO coatings markedly suppressed IL‑8 secretion, while GO alone had no effect (Fig. 6a). All nanoplatforms reduced IL‑6 release, and GO/Ag–GO increased TNF‑β production, suggesting a balanced inflammatory response. Other cytokines remained unchanged, indicating low immunogenicity of the composite.
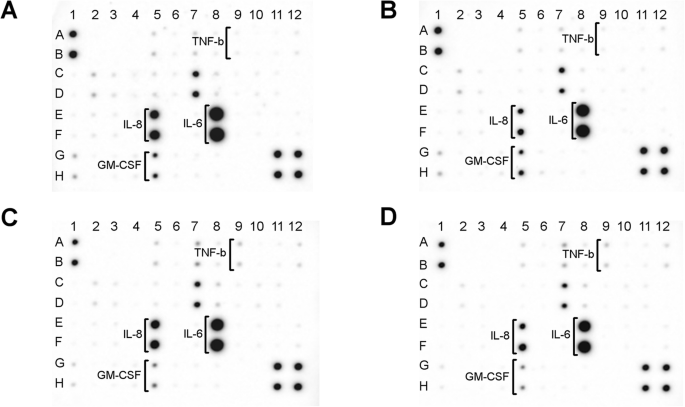
Antibody array analysis of the inflammatory cytokine release of fibroblasts after 24 h of incubation. a Control group (foil without nanoparticles), b nanoplatform coated with silver nanoparticles, c nanoplatform coated with graphene oxide, d Ag‑GO nanoplatform coated with composite of graphene oxide and silver nanoparticles. The AgNP and Ag‑GO nanoplatforms decreased the release level of IL‑8 (dots: E5, F5). Both the GO and Ag‑GO nanoplatforms decreased the synthesis of GM‑CSF (dots: G5, H5). Additionally, the GO and Ag‑GO nanoplatforms led to the increased synthesis of TNF‑β (dots: A9, B9). The AgNP, GO and Ag‑GO nanoplatforms decreased the release level of IL‑6 (dots: E8, F8). A full array map is available in Additional file 1
Discussion
Our data confirm that integrating GO into AgNP coatings not only enhances bacterial eradication but also mitigates cytotoxic and inflammatory responses in human cells and in a vertebrate model. The improved stability of AgNPs within the GO matrix likely limits ion release and particle detachment, thereby reducing cellular uptake and oxidative stress. Moreover, the larger composite size (≈1150 nm) decreases endocytosis rates, as observed for other nanostructures, further lowering toxicity. Importantly, the composite does not provoke significant cytokine storms, maintaining a neutral immunological profile that is essential for clinical translation.
These findings align with previous reports of GO’s ability to stabilize metal nanoparticles and reduce their leaching, while preserving—or even enhancing—antimicrobial potency. The present work extends this concept to a realistic polymer substrate (polyurethane foils) and validates biocompatibility across multiple cell types and an in‑vivo‑like model.
Conclusions
The Ag–GO nanocomposite outperformed both AgNP‑only and GO‑only coatings in antibacterial efficacy against Salmonella enteritidis and in reducing cytotoxicity toward fibroblasts, HUVECs, and the chorioallantoic membrane. The composite’s balanced cytokine profile further supports its suitability for biomedical devices, such as wound dressings and implant coatings. Future work will explore long‑term stability, release kinetics, and in‑vivo infection models to advance clinical deployment.
Materials and Methods
Preparation and Characterisation of Nanoplatforms
Polyurethane foils (15 × 15 mm, 0.05 mm) were dip‑coated in aqueous suspensions of AgNPs (100 mg l−1), GO (200 mg l−1), or Ag–GO (200 mg l−1) prepared via ultrasonication (20 kHz, 60 % efficiency, 30 °C). Coated samples were rinsed in deionised water and dried under sterile conditions. TEM, Raman, FT‑IR, zeta‑potential, and DLS measurements confirmed the morphology and stability of each formulation (details provided in Supplementary Material).
Bacterial Cultivation and Viability Assay
Salmonella enteritidis (ATCC 13076) was grown on tryptic soy agar, harvested, and quantified by OD600 to achieve 5 × 103 CFU ml−1 in MH broth. Foils were incubated with bacteria for 24 h at 37 °C. Post‑incubation, bacterial viability was measured using PrestoBlue, and CFU counts were obtained by serial dilution and plating.
Scanning Electron Microscopy
Bacterial and cellular samples were fixed in 2.5 % glutaraldehyde, contrasted with osmium tetroxide, dehydrated, critical‑point dried, and sputter‑coated with gold before SEM imaging at 15 kV.
Human Cell Culture
HUVECs and fibroblasts were cultured in low‑serum media with supplements (details above). Cells were seeded directly onto foils (1 × 104 cells ml−1) and incubated for 24 h. Viability was assessed by PrestoBlue fluorescence (λex = 560 nm, λem = 590 nm). Morphology was examined by phase‑contrast microscopy.
Chorioallantoic Membrane Assay
Embryos (Day 8) were exposed to foils placed on the chorioallantoic membrane for 48 h. Membranes were harvested, imaged stereoscopically, and evaluated for vascular integrity.
Antibody Array Analysis
Fibroblast supernatants (80 µl per sample) were pooled, diluted, and probed on an antibody array (Abcam, catalogue ab134003) to quantify 40 inflammatory cytokines. Signals were detected with HRP‑conjugated streptavidin and imaged on a ChemiDoc system.
Statistical Analysis
Data were analysed with one‑way ANOVA and Tukey’s HSD post‑hoc test (GraphPad Prism 8). Significance was set at *p* < 0.05.
Availability of Data and Materials
The datasets generated during this study are available from the corresponding author upon reasonable request.
Abbreviations
- Ag‑GO
Composite of silver nanoparticles and graphene oxide
- AgNPs
Silver nanoparticles
- CFU
Colony‑forming units
- DLS
Dynamic light scattering
- FT‑IR
Fourier transform infrared spectroscopy
- GM‑CSF
Granulocyte‑macrophage colony‑stimulating factor
- GO
Graphene oxide
- HUVECs
Human umbilical vein endothelial cells
- IL‑6
Interleukin 6
- IL‑8
Interleukin 8
- SEM
Scanning electron microscope
- TEM
Transmission electron microscope
- TNF‑α
Tumour necrosis factor alpha
- TNF‑β
Tumour necrosis factor beta
Nanomaterials
- One‑Pot Synthesis of Color‑Tunable, Conductive CuS‑Coated CuSCN Composites
- Assessing the Biosafety and Antibacterial Efficacy of Graphene and Graphene Oxide for Orthopedic Implant Applications
- Gold Nanoparticle–Modified 6‑Mercaptopurine and Rabies‑Derived Peptide Enhance SH‑SY5Y Neural Cell Proliferation and Neurite Outgrowth
- Green Production of Copper Nanoparticles: Catalytic, Antibacterial, Cytotoxic, and Antioxidant Potentials
- Graphene Oxide–Silver Nanoparticle Nanocomposites: A Potent Antibacterial and Antifungal Agent
- Green‑Synthesized Silver Nanoparticles from Pomegranate Peel: Potent Antimicrobial and Colon‑Cancer Cytotoxicity
- Magnetic MnFe₂O₄/Reduced Graphene Oxide Nanocomposite: Efficient and Recyclable Adsorption of Tetracycline from Water
- Controlling Silver Nanoparticle Morphology via CTAB‑Capped Seeds and Aging Time
- In‑Situ Synthesis of Silver Nanoparticles on Amino‑Grafted Polyacrylonitrile Fibers for Enhanced Antibacterial Performance
- Apoptosis Pathways Drive Cytotoxicity of Fresh vs. Aged Zinc Oxide Nanoparticles



