Room‑Temperature, Glycerol‑Stabilized Synthesis of ZnO Nanoparticles from Concentrated Zinc Chloride Solutions
Abstract
A simple, room‑temperature route has been developed to produce ZnO nanoparticles directly from highly concentrated aqueous zinc chloride (65–80 wt %) and alkaline solutions in the presence of glycerol as a stabilizer. The ZnCl₂ concentration and the glycerol‑to‑Zn²⁺ molar ratio strongly influence the particle size and morphology: with increasing ZnCl₂ concentration and glycerol ratio, the nanoparticles transition from 50–120 nm rods to 20 nm globules. Glycerol, a water‑soluble polyol, effectively prevents agglomeration and acts as a template even under extreme zinc concentrations.
Background
Zinc oxide (ZnO) nanoparticles are among the most versatile semiconductors, finding use in electronics, optoelectronics, solar cells, field‑emission devices, sensors, and photocatalysis. Their intrinsic biocompatibility and low toxicity also enable applications in drug delivery, biosensing, UV protection, and antimicrobial coatings.
Traditional synthesis routes—chemical vapor deposition, sol‑gel, hydrothermal, laser ablation, and microemulsion—often require low precursor concentrations and high temperatures, leading to significant solvent usage and particle agglomeration. Polyol synthesis, which employs high‑boiling diols as both solvent and stabilizer, has proven effective for metal oxides but still suffers from low precursor loading and elevated reaction temperatures.
Zinc chloride is highly soluble in water (432 g/100 g at 25 °C, up to 81.2 wt %) and can serve as a concentrated ZnO precursor. Previous work has shown that ZnO can be obtained from concentrated ZnCl₂ solutions in the presence of starch or cellulose, but those methods require elevated temperatures and multi‑step processing. This study introduces a one‑step, room‑temperature method that uses glycerol—a green, highly water‑soluble polyol—as a stabilizer and template to generate ZnO nanoparticles from extremely concentrated ZnCl₂ solutions.
Methods/Experimental
Materials
Analytical‑grade ZnCl₂, NaOH, KOH, LiOH, ammonia water, and glycerol were used without further purification (Nanjing Chemical Reagent Factory, China).
Preparation of the ZnO Nanoparticle
Glycerol was first mixed with an aqueous ZnCl₂ solution at a prescribed molar ratio (Zn²⁺:glycerol). Then an alkaline solution (NaOH, KOH, LiOH, or NH₄OH) was added dropwise under continuous stirring at room temperature to reach pH 12. The mixture was stirred for an additional 5 min. A white emulsion formed, which was washed twice with water and ethanol, then centrifuged (6000 rpm, 10 min). After drying at 80 °C, ZnO nanoparticles were recovered.
Characterization of ZnO Nanoparticles
Crystalline structure was assessed by XRD (Ultima IV, Japan). Morphology was examined with SEM (JSM‑7600F, JEOL) and TEM (JEM‑2100, JEOL). XPS (AXIS Ultra DLD, UK) identified chemical states. UV–vis absorption spectra were recorded with a Lambda 950 (Perkin Elmer), with a peak at 325 nm.
Results and Discussion
Effect of Glycerol‑to‑Zn²⁺ Ratio on Size and Morphology
Figure 1 illustrates the pronounced influence of glycerol on nanoparticle formation when 65 wt % ZnCl₂ and 50 wt % NaOH are used. Without glycerol (Fig. 1a), particles are large and heterogeneous. Increasing the glycerol ratio progressively yields smaller, more uniform globules: 0.33 : 1 produces 180 nm rods; 1 : 1 gives 40–80 nm spheres; 3.33 : 1 delivers 15–25 nm globules. Thus, glycerol serves both as a steric stabilizer and a template.

SEM images of ZnO nanoparticles obtained from 65% ZnCl₂ under different glycerol ratios (a 0:1; b 0.33:1; c 1:1; d 1.67:1; e 3.33:1) and (f) the statistics table.
Mechanistically, glycerol’s three hydroxyl groups coordinate Zn²⁺, forming Zn–glycerol complexes. Upon addition of NaOH, these complexes generate Zn(OH)₄²⁻ that dehydrates to ZnO near glycerol. The high NaOH concentration (50 wt %) induces a burst of homogeneous nucleation, while glycerol blocks agglomeration. At low glycerol content, growth is uneven and particle sizes increase; at high glycerol, the blocking effect yields uniform, sub‑30 nm particles.
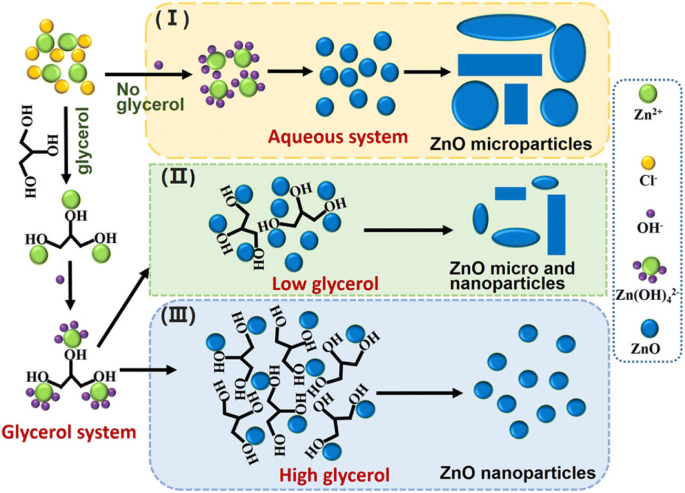
Schematic representation of glycerol’s role in ZnO nanoparticle synthesis.
Figure 3 confirms these observations: TEM images show rods at 0.33 : 1 (20–160 nm) and globules at 1 : 1 (≈40–50 nm) and 3.33 : 1 (≈15–25 nm). XPS analysis (Fig. 4) indicates fully oxidized Zn (Zn²⁺) with minimal residual glycerol. XRD patterns (Fig. 5a) match the wurtzite ZnO structure, while UV–vis spectra (Fig. 5b) display a strong absorption peak near 380 nm, confirming the nanoscale ZnO’s UV‑blocking capability.
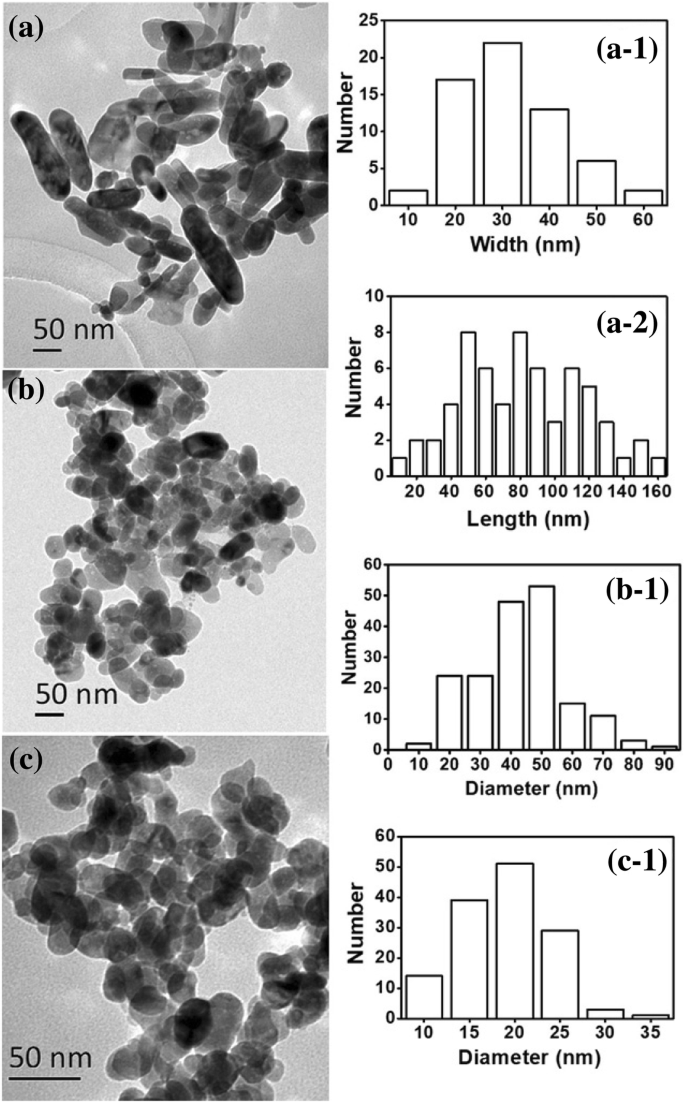
TEM images of ZnO nanoparticles from varying glycerol ratios (a 0.33:1; b 1:1; c 3.33:1). Corresponding histograms are shown.
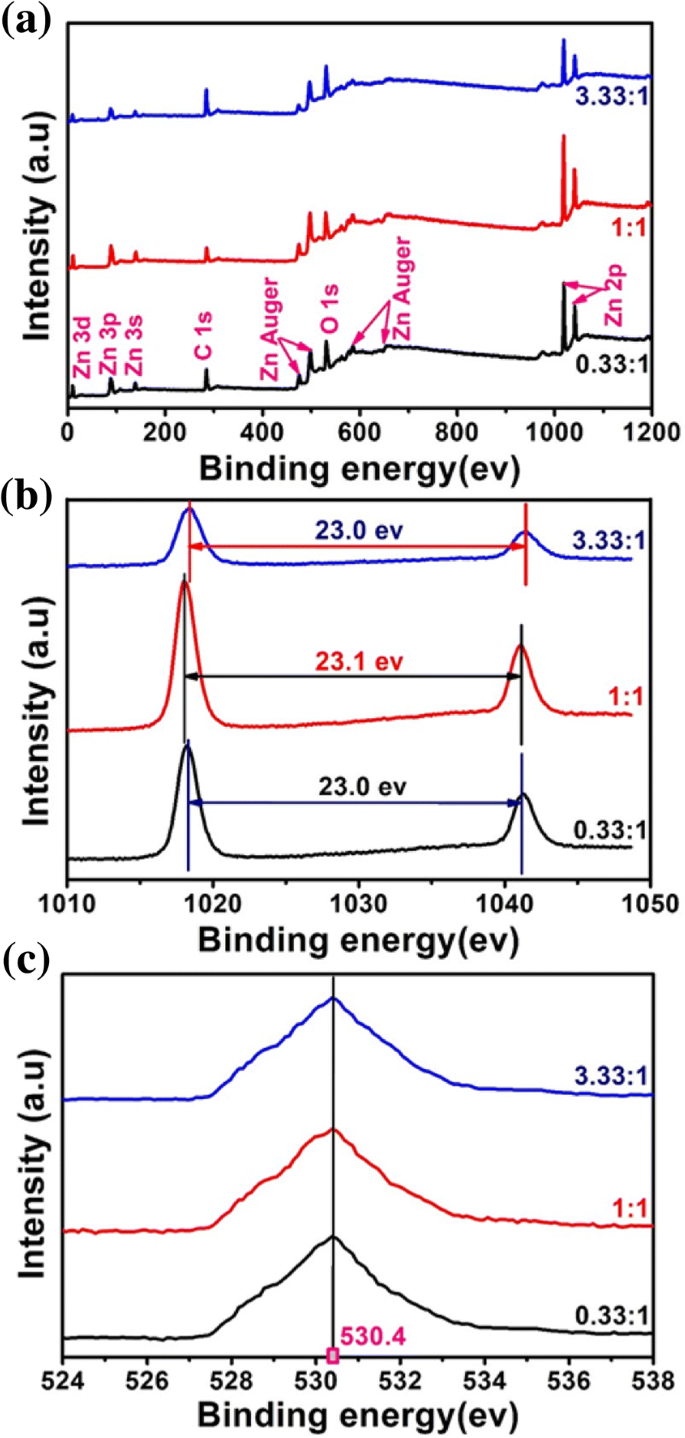
XPS spectra of ZnO nanoparticles (a wide; b Zn‑2p; c O‑1s).
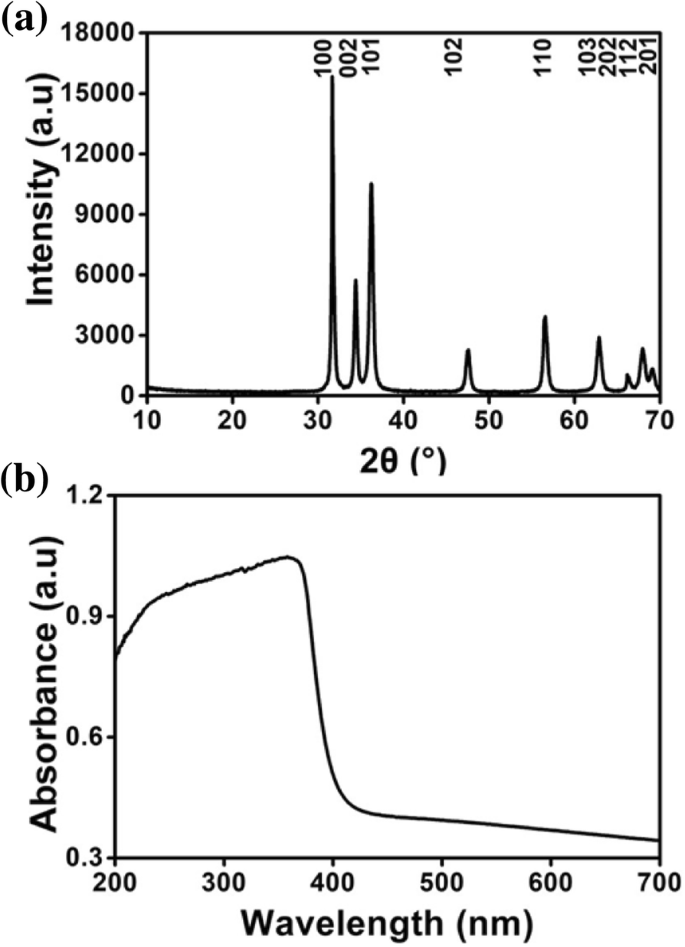
XRD pattern (a) and UV–vis absorption spectrum (b) of ZnO nanoparticles.
Effect of ZnCl₂ Concentration on Nanoparticle Morphology
Using a 1:1 glycerol:Zn²⁺ ratio and 50 wt % NaOH, ZnCl₂ concentration was varied from 50 to 80 wt %. Figure 6 shows that increasing ZnCl₂ concentration (decreasing hydration ratio) transforms rods into globular particles and reduces size. At 50 wt % ZnCl₂, rods measure ≈130 nm; at 65 wt % they become 40–80 nm spheres; at 80 wt % they shrink to ≈40 nm globules. The enhanced glycerol blocking effect at higher ZnCl₂ concentrations explains the smaller particles.

SEM images of ZnO nanoparticles from 50 wt % (a), 65 wt % (b), and 80 wt % (c) ZnCl₂ solutions.
Influence of Hydroxide Type on Nanoparticle Size
We investigated saturated NaOH, KOH, LiOH, and NH₄OH solutions at room temperature (concentrations: 50 wt %, 60 wt %, 8 wt %, and 25 wt % respectively). With a 3.33 : 1 glycerol:Zn²⁺ ratio, ZnO particles ranged from ≈20 nm (NaOH) to ≈300 nm (NH₄OH). The size trend reflects the ability of the cation to passivate the ZnO surface; larger hydrated cations (Li⁺ > Na⁺ > NH₄⁺ > K⁺) require higher concentrations to achieve similar passivation. Low LiOH and NH₄OH concentrations reduce glycerol’s blocking effect, leading to larger particles.

SEM images of ZnO nanoparticles from 65 % ZnCl₂ with various hydroxides (a NaOH; b KOH; c LiOH; d NH₄OH).
Comparison with Starch or Cellulose Systems
Previous work synthesized ZnO from starch or cellulose dissolved in concentrated ZnCl₂. Figure 8 compares the resulting particles: starch‑ and cellulose‑based routes yield 40–60 nm globules, whereas the glycerol system produces 15–25 nm particles. The lower viscosity of the glycerol solution allows more efficient droplet formation during milling, enhancing particle uniformity.
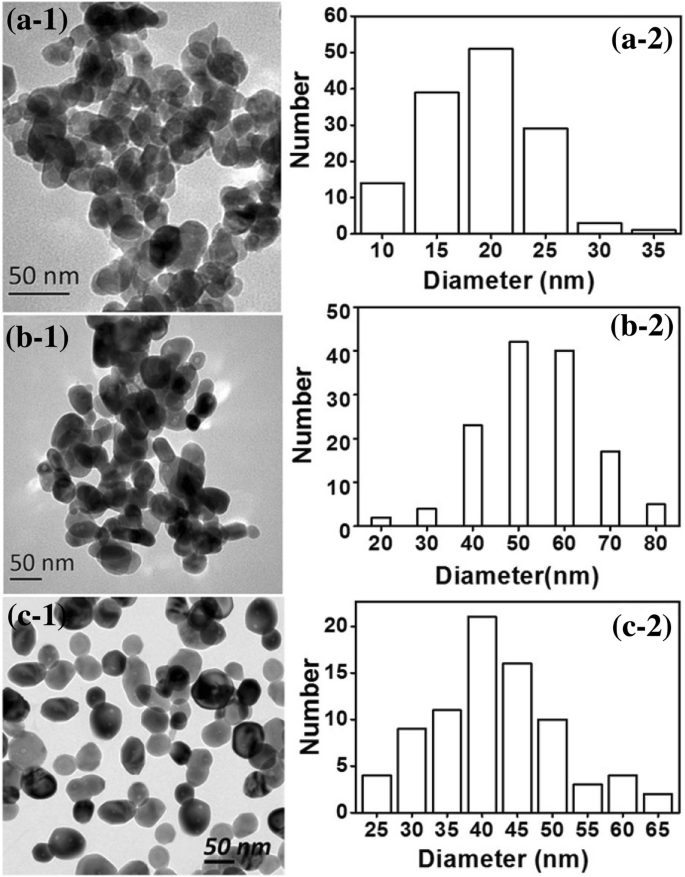
TEM images of ZnO nanoparticles from 65 % ZnCl₂ using glycerol (a), starch (b), and cellulose (c) systems.
Thus, the glycerol approach is simpler, more cost‑effective, and yields smaller, more uniform ZnO nanoparticles.
Conclusions
By adding an alkaline solution to a glycerol‑ZnCl₂ mixture at room temperature, ZnO nanoparticles of ≈20 nm can be produced from extremely concentrated zinc chloride (up to 80 wt %). The particle size and morphology are tunable via the glycerol:Zn²⁺ ratio, ZnCl₂ concentration, and hydroxide type. Glycerol’s steric blocking becomes more pronounced at higher ZnCl₂ loadings, shifting morphology from rods to globules and reducing size. NaOH and KOH produce 20–50 nm particles, while LiOH and NH₄OH yield larger particles due to less effective surface passivation. This method offers a facile, scalable, and environmentally benign route to size‑controlled ZnO nanoparticles.
Abbreviations
- BD
1,4-butanediol
- DEG
Diethylene glycol
- EG
Ethylene glycol
- i-PrOH
Iso‑propanol
- PD
1,3-propanediol
- SEM
Scanning electron microscope
- TEG
Tetraethylene glycol
- TEM
Transmission electron microscope
- XRD
X‑ray diffraction patterns
- ZnO
Zinc oxide
Nanomaterials
- Au@TiO₂ Yolk–Shell Nanostructures: Tailored Synthesis and Their Superior Visible‑Light Photocatalytic Degradation and SERS Detection of Methylene Blue
- Efficient Fabrication of Cu₂O/TiO₂ Nanotube Heterojunctions for Enhanced Visible‑Light Photocatalysis
- Optimizing Zinc Oxide/Porous Anodic Alumina Composite Films for Superior Antibiofilm Performance
- PdO‑CeO₂ Rod‑Like Nanoporous Catalysts with Superior CO Oxidation and Methane Combustion Performance
- Synthesis of Highly Dispersible ZnO Nanoparticles via Oriented Attachment: A Comprehensive Study
- Enhanced Photoelectrochemical Water Splitting with TiO₂ Nanosheet Arrays, Layered SnS₂, and CoOx Nanoparticles
- Reducing Cytotoxicity of Silver Nanoparticle Coatings with Graphene Oxide: Enhanced Antibacterial Performance and Biocompatibility of an Ag–GO Nanocomposite
- Biodegradable Fe‑Doped Zinc Oxide Nanoparticles: A Promising Iron Delivery System for Biological Applications
- Easy Fabrication of rGO@Fe3O4 Microspheres for Magnetically Targeted, NIR-Responsive Chemo‑Photothermal Cancer Therapy
- Apoptosis Pathways Drive Cytotoxicity of Fresh vs. Aged Zinc Oxide Nanoparticles



