Synthesis of Highly Dispersible ZnO Nanoparticles via Oriented Attachment: A Comprehensive Study
Abstract
Understanding how nanocrystals grow is essential for tailoring their physical and chemical properties. While Ostwald ripening (OR) has long been considered the dominant growth pathway, recent evidence highlights oriented attachment (OA) as a significant complementary mechanism. In this work, we synthesized zinc oxide (ZnO) nanoparticles (NPs) using a simple wet‑chemical route and systematically investigated their size, shape, and surface evolution in ethanol by transmission electron microscopy (TEM), dynamic light scattering (DLS), and X‑ray diffraction (XRD). Our results show that reaction parameters—temperature, time, pH, and precursor concentrations—directly influence NP morphology and surface roughness. Notably, a “rough” surface state preceding OA markedly enhances colloidal stability, providing a robust platform for designing nano‑suspensions with tailored properties.
Introduction
Zinc oxide nanoparticles have attracted sustained interest across diverse fields, from physical chemistry to biomedical applications, owing to their unique optical, electronic, and antimicrobial attributes. Their versatility is evident in catalysis, transducers, semiconductors, microelectronics, textiles, cosmetics, and water remediation. Moreover, ZnO NPs exhibit intrinsic antibacterial and anti‑inflammatory effects, offering a safer alternative to conventional antibiotics.
Despite the plethora of synthesis strategies, controlling crystalline structure, dispersibility, and surface chemistry in common solvents remains challenging. A deep mechanistic understanding of NP formation is therefore imperative. While the general framework of crystal growth is reasonably well understood, the nuances of OA—particularly the evolution of particle surface structure—are still underexplored. This study bridges that gap by correlating reaction conditions with the OA process and the resulting colloidal stability of ZnO NPs.
We focused on optimizing the synthesis of a highly stable ZnO NP suspension in ethanol without surfactants, by systematically varying pH, reaction time, temperature, and precursor ratios. The growth of individual particles and their clusters was monitored by HR‑TEM and XRD, marking the first report that links reaction parameters to the suspension and dispersion behavior of ZnO NPs.
Methods
Reagents: zinc acetate dihydrate (Zn(O₂CCH₃)₂·2H₂O) and sodium hydroxide (NaOH) were sourced from Shanghai Aladdin Biochemical Technology Co. (China). Absolute ethanol was supplied by Tianjin Damao Chemical Reagents Co. (China). All chemicals were of analytical grade and used as received.
Standard synthesis conditions: 60 °C, 2 h, 7.22 mmol NaOH and 3.73 mmol zinc acetate. By varying precursor concentrations, reaction time, temperature, and pH, we obtained a series of white precipitates (see Additional file 5: Table S1).
Procedure: Two solutions were prepared—solution A (3.73 mmol zinc acetate in 40 mL ethanol) and solution B (7.22 mmol NaOH in 320 µL water, then diluted to 25 mL ethanol). Solution B was added dropwise to solution A under vigorous stirring for 2.25 h at 45–65 °C. The mixture cooled to room temperature, and ZnO was isolated by centrifugation, washed repeatedly with ethanol, and re‑dispersed or dried at 60 °C for 2 h. Samples were labeled 1–6. During NP formation, Zn(O₂CCH₃)₂·2H₂O reacted with NaOH in ethanol, with ethanol’s dehydration property preventing zinc hydroxide formation.
Aging experiments: For sample 4, aging times of 1, 1.5, 2.25, 6, 12, and 24 h were studied (samples 19–24). Additional runs varied precursor amounts: 1–18 mmol Zn(O₂CCH₃)₂·2H₂O and 3.73–9.33 mmol NaOH (samples 7–18).
Dispersion assessment: Post‑washing, NPs were sonicated and vortexed in fresh ethanol. Visual inspection and UV‑vis absorbance at 370 nm quantified suspension stability over 1, 7, 14, and 21 days. The synthetic workflow is illustrated in Scheme 1.
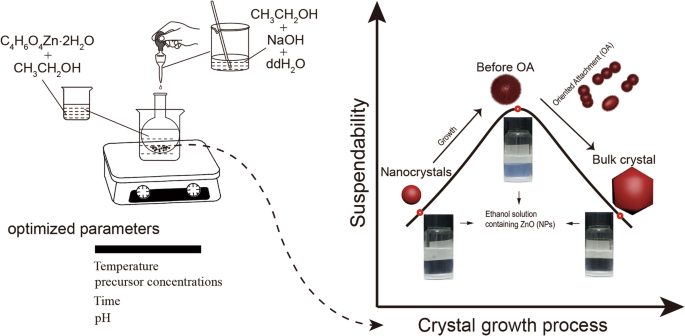
Schematic illustration of ZnO NP synthesis and suspendability study
Analytical instrumentation: UV‑Lambda 370 spectrometer (Perkin Elmer), SEM (Hitachi), HR‑TEM with EDX and SAED (FEI Tecnai G2 F20, Thermo Fisher), XRD (Smartlab with Cu Kα, λ = 1.5418 Å, 2θ = 20–80°), and DLS (ELSZ‑2, Otsuka). Samples were sonicated before DLS to minimize aggregation.
Results and Discussion
Suspendability Analysis
Colloidal stability was monitored by turbidity at 370 nm, a wavelength free from nanoparticle absorption. Figure 1 shows that samples with higher absorbance exhibit lower suspendability. Samples 3 and 21 were further examined by zeta potential, confirming a correlation between turbidity and surface charge.
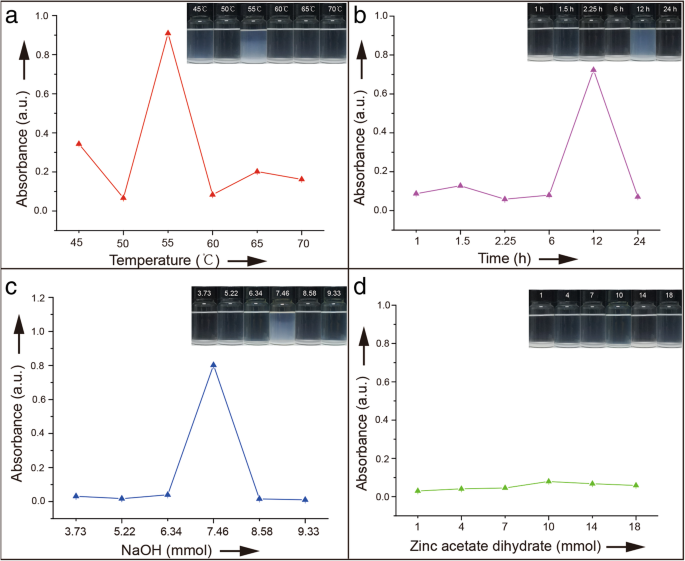
Absorbance of ZnO NPs in ethanol at 370 nm for varying a reaction temperature, b reaction time, c NaOH molarity, and d zinc acetate molarity, after 3 weeks of aging.
Optimal suspension performance was achieved at 55 °C, 12 h, and 7.46 mmol NaOH, yielding particles that remained transparent for weeks. Excessive temperature, time, or pH led to precipitation and reduced stability. Contrary to classical crystal theory, precursor concentration did not influence suspendability (Fig. 1d), suggesting that high concentrations do not necessarily promote aggregation in non‑classical crystallization.
XRD Analysis
All XRD patterns matched hexagonal wurtzite ZnO (JCPDS 36‑1451), with no secondary phases. Sample 4 exhibited a c‑lattice constant of 0.26 nm. Peak broadening reflected the nanoscale dimensions.
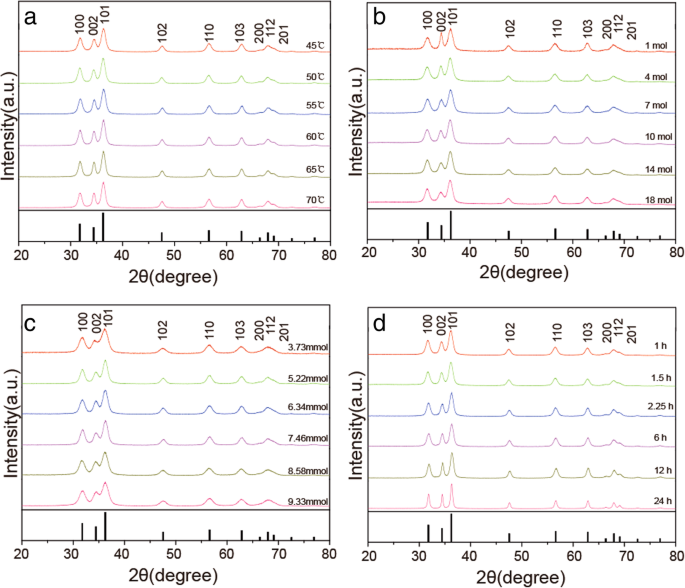
XRD patterns of ZnO NPs at varying a temperature, b zinc acetate, c NaOH, and d time. Bulk ZnO reference shown below each set.
Enhanced (002) intensity in samples 11 and 12 indicated nanorod growth along the c‑axis. Scherrer analysis yielded sizes of 5–15 nm for samples 1–21 and 10–100 nm for samples 22–24, consistent with TEM results.
Morphological Analyses
High‑resolution TEM revealed predominantly spherical particles with lattice fringes of 2.60 Å (wurtzite (002)). FFT patterns confirmed single‑crystalline hexagonal structure. SEM images showed larger apparent sizes due to aggregation during preparation. EDX confirmed Zn and Cu (grid). SAED patterns matched wurtzite symmetry.
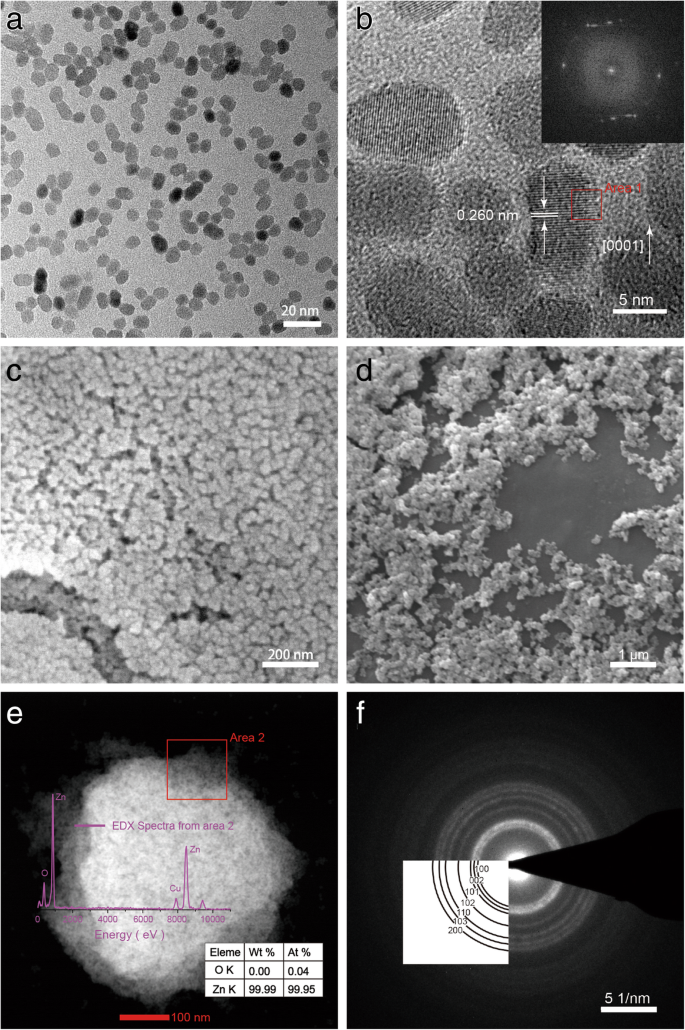
TEM, SEM, EDX, and SAED of ZnO NPs from sample 4. a, b Low‑ and high‑magnification TEM; c, d SEM images; e EDX spectrum; f SAED.
HRTEM showed that merged particles maintained near‑perfect crystallographic alignment, with occasional dislocations at junctions (Fig. 4a). Over time, misalignments diminished, indicating recrystallization during OA (Fig. 4b). These observations support a layered growth mechanism, forming nanorods along the c‑axis.
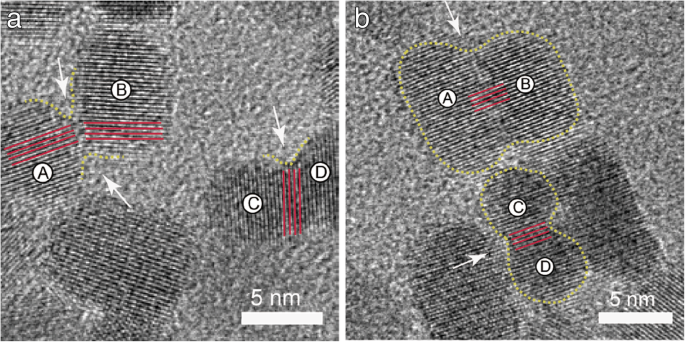
HRTEM of ZnO NPs: a Dislocations from OA; b Layer‑by‑layer assembly along the c‑axis.
OA is strongly influenced by solvent, water content, pressure, temperature, and pH. Ethanol proved superior to methanol for controlling OA. Temperature emerged as the most critical factor; at 40 °C no precipitation occurred under conditions matching sample 21, underscoring its role in crystal growth.

TEM images of ZnO NPs with morphologies varying by reaction a time, b temperature, and c NaOH content. Samples correspond to 2–4, 10–12, and 15–17 respectively.
Time‑dependent TEM revealed stages from isolated nuclei (Fig. 6a) to flake‑like aggregates (Fig. 6b) and fully merged, well‑crystallized particles (Fig. 6c). The progressive widening of the merging zone (Fig. 6d) indicates surface roughening preceding OA, corroborating the “rough” state hypothesis.
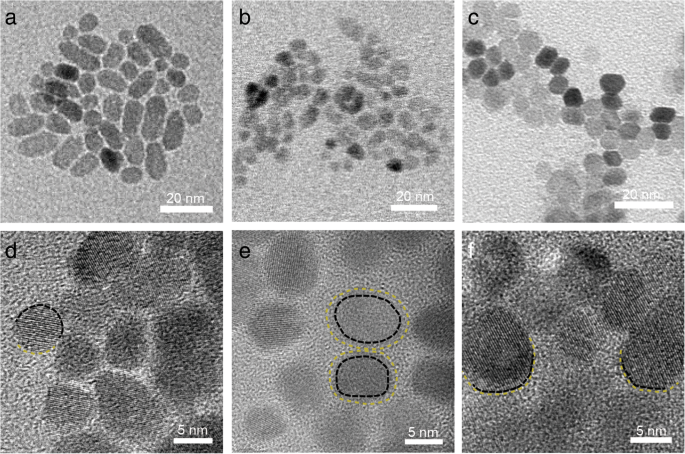
TEM snapshots of ZnO NP growth: a, d primary nucleation; b, e flake aggregates; c, f well‑crystallized particles.
Although pH had limited effect on morphology, it altered surface roughness (Fig. 5c). Rough surfaces likely represent early OA intermediates, as supported by literature on non‑classical nucleation.
DLS Analysis
Figure 7 displays DLS distributions for ZnO NPs synthesized at 55 °C, 7.46 mmol NaOH, and 0.1 mmol zinc acetate over various reaction times. Polydispersity indices ranged from 0.140 to 0.287, reflecting size evolution. Early-stage distributions (Fig. 7a) correspond to nucleation, while later stages (Fig. 7b–e) show a bimodal distribution, indicative of OA‑driven aggregation.
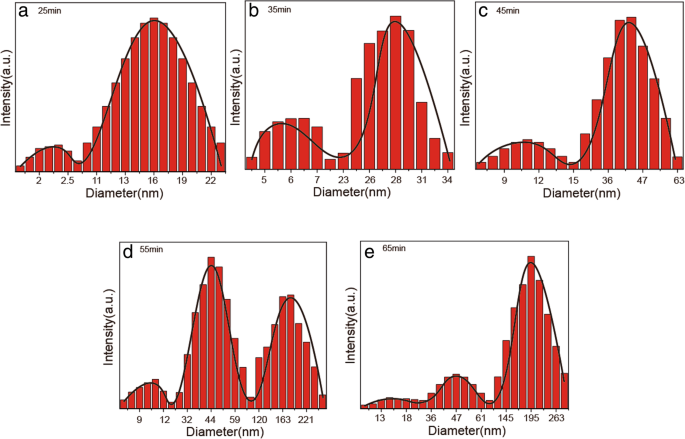
DLS size distributions for samples at 55 °C: a 25 min, b 35 min, c 45 min, d 55 min, e 65 min.
Bimodal distributions were consistently observed across multiple samples, a hallmark of OA. While similar patterns can arise from conventional aggregation, the concurrent TEM and XRD evidence confirms OA as the dominant growth pathway.
Analysis of Oriented Attachment
Classical Ostwald ripening predicts growth of larger particles at the expense of smaller ones. However, many experimental observations—such as nucleation at low supersaturation and unusual suspension morphologies—cannot be reconciled with this model. OA, by contrast, involves lattice‑matched attachment of adjacent particles, a process that naturally explains these anomalies.
Our data reveal that OA is preceded by a roughening phase where particle surfaces become atomically irregular, lowering the nucleation energy barrier (LNEB). Once the surface smooths, lattice alignment facilitates rapid coalescence, yielding elongated nanorods along the c‑axis. This sequence—classical nucleation, surface roughening, and oriented aggregation—provides a comprehensive framework for interpreting our observations.
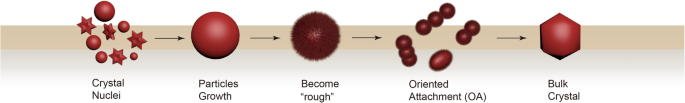
Stages of crystallization via OA.
Controlling the rough‑state duration offers a route to engineer long‑lived colloids, a strategy that can be extended to other nanomaterials.
Conclusions
We have demonstrated that ZnO NPs can be synthesized in ethanol at modest temperatures without surfactants, yielding highly stable suspensions. By tuning temperature, aging time, precursor ratios, and pH, we identified that the surface roughening phase preceding OA is pivotal for achieving long‑term colloidal stability.
Our findings confirm, for the first time, that ZnO NP suspendability in ethanol is controllable and suitable for practical applications that demand stable nano‑suspensions.
Understanding the role of surface structure in OA opens new avenues for designing nanomaterials with tailored properties, promising advances across industrial and biomedical sectors.
Availability of Data and Materials
Data supporting this study are available from the corresponding author upon reasonable request.
Abbreviations
- DLS:
Dynamic light scattering
- EDX:
Energy dispersive X‑ray spectroscopy
- HR‑TEM:
High‑resolution transmission electron microscopy
- LNEB:
Lower nucleation energy barrier
- NPs:
Nanoparticles
- OA:
Oriented attachment
- OR:
Ostwald ripening
- SAED:
Selected area electron diffraction
- SEM:
Scanning electron microscope
- TEM:
Transmission electron microscopy
- XRD:
X‑ray diffraction
Nanomaterials
- pH‑Responsive Ag@Polyacryloyl Hydrazide Nanoparticles: A Smart, Ultra‑Sensitive SERS Substrate for Trace Analysis
- Enhancing Silicon Carbide Densification: Spark Plasma Sintering with Si‑SiC Nanocomposite Additives from Thermal DC Plasma
- High‑Performance PEDOT:PSS/n‑Si Solar Cells with Textured Surfaces and Silver Nanowire Electrodes
- Hybrid Silicon–Graphene Metasurface: Ultra‑Thin, Tunable Polarization Converter with 96% Efficiency
- Room‑Temperature, Glycerol‑Stabilized Synthesis of ZnO Nanoparticles from Concentrated Zinc Chloride Solutions
- Tunable Solvothermal Synthesis of Phase‑Selective Cu(In,Ga)S₂ Nanoparticles via Solvent Composition Control
- High‑Performance Polyelectrolyte Microcapsules Encoded with PEGylated Quantum Dots for Advanced Bioimaging
- High‑Performance All‑Perovskite Photodetector Delivering Ultrafast Response
- Anion‑Controlled Fabrication of ZnO 1D Necklace‑Like Nanostructures for Superior Photocatalytic Performance
- Apoptosis Pathways Drive Cytotoxicity of Fresh vs. Aged Zinc Oxide Nanoparticles



