Tunable Solvothermal Synthesis of Phase‑Selective Cu(In,Ga)S₂ Nanoparticles via Solvent Composition Control
Abstract
Cu(In,Ga)S₂ (CIGS) is a leading contender for next‑generation thin‑film photovoltaics, owing to its favorable band alignment, high absorption coefficient, and compositional flexibility. Despite extensive research on its stable chalcopyrite form, practical methods for controlling the formation of metastable wurtzite (WZ) and zincblende (ZB) polymorphs remain scarce. In this study, we demonstrate a straightforward solvothermal route that yields phase‑selective CIGS nanoparticles (NPs) by merely adjusting the solvent mixture. When the reaction is carried out in pure ethylenediamine (en), WZ‑CIGS NPs with a 1.6 eV band gap and 4.85 cm² V⁻¹ s⁻¹ carrier mobility are obtained. Switching to an en/water blend selectively produces ZB‑CIGS NPs. Detailed structural, optical, and electrical characterizations confirm that the solvent composition dictates the Cu‑S nucleation environment, thereby steering the final crystal structure. This low‑cost, additive‑free approach opens a scalable pathway for tailoring CIGS polymorphs for high‑efficiency solar cells and other optoelectronic devices.
Introduction
I‑III‑VI₂ quaternary semiconductors have garnered significant attention for photoelectric applications because of their tunable band gaps, high absorption, and benign elemental composition. The record 22 % efficiency achieved by Cu(In,Ga)Se₂ (CIGSe) thin‑film solar cells illustrates the potential of this family, yet the high cost of selenium and vacuum‑based processing hinders commercial deployment. Replacing Se with the less toxic, inexpensive sulfur to form CuInS₂ (CIS) or CIGS offers a practical alternative. Nanoparticle (NP)‑based, non‑vacuum synthesis is particularly attractive for cost reduction and scalability. Consequently, the development of phase‑controlled, low‑temperature CIGS NP routes is a critical research frontier.
Phase control is paramount because the optoelectronic properties of chalcogenides are intrinsically linked to crystal symmetry. While the tetragonal chalcopyrite (CH) phase is thermodynamically favored, the cubic zincblende (ZB) and hexagonal wurtzite (WZ) metastable forms can exhibit distinct band gaps and carrier dynamics. Prior work has reported metastable CIS or CIGS synthesis via hot‑injection, solvothermal, or thermal‑decomposition methods, but these protocols often rely on expensive ligands, complex precursors, or additional additives. A simple, additive‑free strategy that can reliably produce either WZ or ZB CIGS NPs is therefore highly desirable.
In this paper, we present a one‑pot solvothermal synthesis that exploits the solvent’s coordination chemistry to direct Cu‑S nucleation, thereby enabling selective formation of metastable WZ or ZB CIGS. We also examine the resulting nanoparticles’ morphology, composition, and optoelectronic performance, establishing a clear structure–property relationship that informs future device design.
Experimental Methods
Materials
CuCl₂·2H₂O (99.9 %), sulfur powder (99.9 %), anhydrous ethylenediamine (en, 99 %), anhydrous ethanol (99.7 %), InCl₃ (99.9 %), and Ga(acac)₃ (99.99 %) were sourced from Chengdu Kelong Chemical Co., Ltd. and Sigma‑Aldrich. All reagents were used as received without further purification.
Solvothermal Synthesis of CIGS NPs
For WZ‑CIGS, the metal precursor solution was prepared by dissolving 0.164 g CuCl₂·2H₂O (0.96 mmol), 0.192 g InCl₃ (0.868 mmol), and 0.068 g Ga(acac)₃ (0.186 mmol) in 5 mL en. Sulfur (0.0826 g, 2.58 mmol) was dispersed in 5 mL en by ultrasonic agitation at 60 °C for 15 min. The precursor and sulfur solutions were combined in a 20‑mL Teflon‑lined autoclave, and an additional 6 mL en was added to reach 80 % of the autoclave volume. After sonication at 60 °C for 15 min, the autoclave was sealed and heated to 200 °C for 24 h, then cooled naturally to room temperature. The resulting precipitate was collected by centrifugation, washed five times with an ethanol‑water mixture, and dried under vacuum at 60 °C for 4 h.
For ZB‑CIGS, the only difference was that the metal salts were dissolved in 5 mL deionized water rather than en. All other synthesis steps remained unchanged.
Characterization
Phase identification was performed by X‑ray diffraction (XRD) on a Bruker D8 Advance with Cu‑Kα radiation (λ = 1.5418 Å), using 0.02° step size and 0.1 s/step scan rate. Morphology and elemental composition were examined by a ZEISS EVO LS15 SEM (15 kV) equipped with a Bruker Nano XFlash 5010 detector for energy‑dispersive X‑ray spectroscopy (EDS). Optical absorption was measured with an Agilent Cary 5000 UV‑Vis‑IR spectrophotometer. Hall effect measurements (Swin Hall 8800) were carried out on spray‑coated films on glass substrates, with silver paste contacts. Raman spectra were recorded on a Renishaw InVia system.
Results and Discussion
The solvothermal reaction in pure en yielded XRD patterns that match the reported wurtzite CIS structure, confirming the formation of hexagonal WZ‑CIGS. In contrast, the en/water mixture produced XRD peaks consistent with cubic zincblende CIS, demonstrating that the solvent composition alone can dictate the metastable phase. This phase‑selectivity is illustrated in Fig. 1.
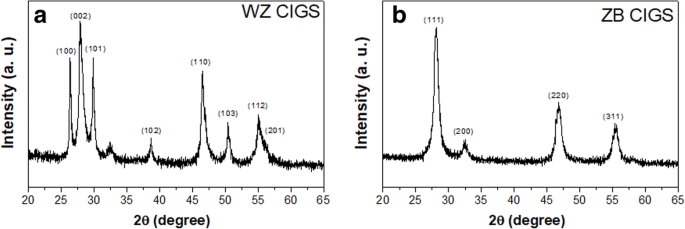
XRD patterns of CIGS NPs synthesized in pure en (a) and en/water mixture (b)
To rationalize this observation, we examined the Cu–S nucleation environment. Raman spectroscopy of the sulfur solution in en revealed a split peak at 811 cm⁻¹, absent in the en/water mixture, indicating a different S coordination state. En, with its dual amine groups, can partially reduce elemental sulfur and strongly chelate Cu²⁺. In pure en, Cu²⁺ is largely complexed (Cu(en)₂²⁺), limiting free metal ions at low temperature; nucleation therefore occurs only at elevated temperatures when the complex dissociates and S is reduced. In the en/water mixture, free Cu²⁺ concentration is high (≈0.192 M), allowing Cu–S cluster formation at room temperature and subsequent growth at 200 °C. This kinetic pathway favors the cubic ZB structure, while the delayed nucleation in pure en promotes the hexagonal WZ structure.
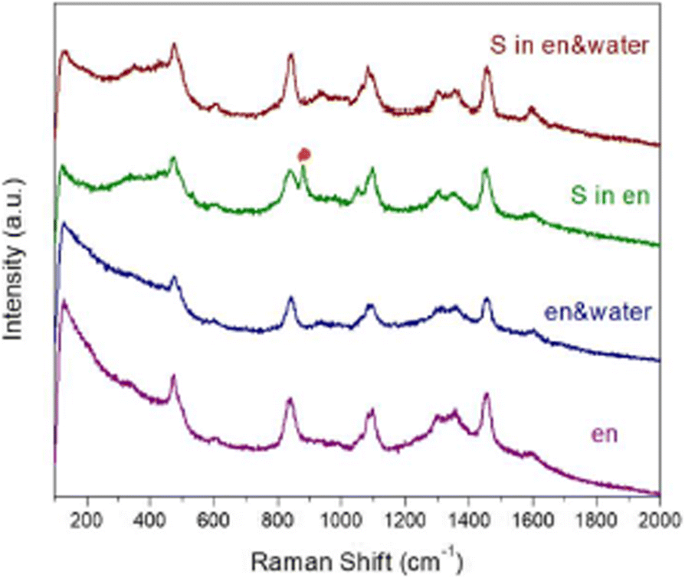
Raman spectra of pure en (purple), en/water mixture (blue), S in en (green), and S in en/water (red)
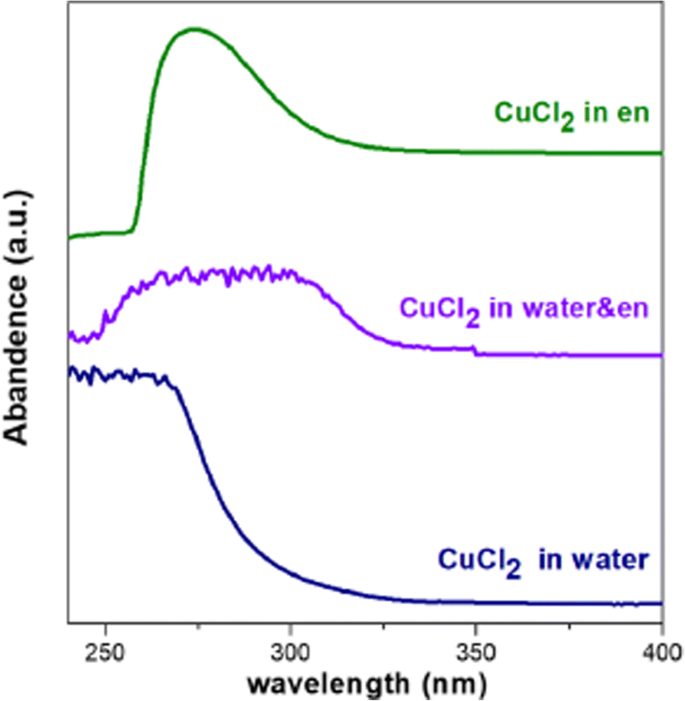
UV‑Vis absorption of Cu precursors in en (green), water (blue), and en/water (violet)
Morphologically, WZ‑CIGS NPs are uniform, ~50 nm spheres with narrow size distribution (Fig. 4a,b), whereas ZB‑CIGS displays irregular shapes—pellets, flakes, and rods (Fig. 4c,d). The homogeneous nucleation in pure en leads to a high density of nuclei that consume most precursors, limiting growth and producing monodisperse particles. Conversely, early‑stage nucleation in the en/water medium results in fewer nuclei and continued growth at higher temperatures, yielding heterogeneous morphology.

Representative SEM images of WZ‑structured (a, b, c) and ZB‑structured (c, b, d) CIGS NPs
EDS analysis (Table 1) confirms near‑stoichiometric Cu:(In+Ga) ratios for both phases, with a slight sulfur deficit (S ≈ 0.91–0.96 S per metal). The WZ‑CIGS shows a modest Cu‑poor composition (Cu:(In+Ga) = 1:1.16), favorable for p‑type conductivity in solar‑cell absorbers. Optical absorption indicates a band gap of ~1.6 eV for both polymorphs, consistent with theoretical predictions for CIGS. Hall measurements reveal n‑type behavior (expected for sulfur‑poor materials) with sheet resistivity and carrier mobilities comparable to literature values, but WZ‑CIGS demonstrates superior mobility (4.85 cm² V⁻¹ s⁻¹) relative to ZB‑CIGS.
To assess the versatility of this solvent‑based approach, we extended the method to CIS and CuGaS₂ (CGS). XRD patterns confirm that both compounds can be synthesized in either WZ or ZB phases by simply selecting the solvent, underscoring the generality of the strategy for tuning the In/Ga ratio and band gap.
Conclusion
We have established a simple, additive‑free solvothermal protocol that yields phase‑selective CIGS nanoparticles by adjusting the en/water ratio. The solvent’s coordination environment controls Cu‑S nucleation kinetics, steering the growth toward either hexagonal wurtzite or cubic zincblende structure. The resulting WZ‑CIGS NPs possess uniform morphology, a 1.6 eV band gap, and high carrier mobility, making them attractive for heterojunction solar cells and other optoelectronic devices. This strategy provides a scalable platform for tailoring metastable phases in copper‑based multinary chalcogenides, with broad implications for advanced material synthesis.
Nanomaterials
- Resveratrol-Loaded Human Serum Albumin Nanoparticles with PEG–RGD Targeting: Extended Circulation, Enhanced Biocompatibility, and Potent Pancreatic Tumor Suppression
- One‑Pot Synthesis of Color‑Tunable, Conductive CuS‑Coated CuSCN Composites
- Polypyrrole‑Coated FePt Nanoparticles: A Multifunctional Platform for Photothermal Therapy and Photoacoustic Imaging
- Green Synthesis of Metal‑Oxide Nanoparticles with Gum Karaya and Their Ecotoxicological Impact on *Chlamydomonas reinhardtii*
- Nanoparticle‑Induced Modulation of Cellular Mechanics: Comparative Analysis of SiO₂ and TiO₂ Effects on Caco‑2 and A549 Cells
- Synthesis of Highly Dispersible ZnO Nanoparticles via Oriented Attachment: A Comprehensive Study
- Efficient Charge Transfer in Au/CdSe Janus Nanoparticles Boosts Photocatalytic Hydrogen Production
- Biotin‑Functionalized Redox‑Sensitive Chitosan Nanoparticles Coated with Phycocyanin for Targeted Curcumin Delivery
- Controlling Gold Nanoparticle Size via Solvent Polarity in Chemical Reduction Synthesis
- Biogenic Au@Ag Core–Shell Nanoparticles Synthesized Using Rumex hymenosepalus Extract for Antimicrobial Applications



