How Synthesis Route Shapes the Structure, Magnetism, and Hyperthermia Performance of La₁₋ₓSrₓMnO₃ Nanoparticles
Abstract
Lanthanum‑strontium manganite (La₁₋ₓSrₓMnO₃, LSMO) nanoparticles were synthesized by three distinct routes: a sol‑gel approach, precipitation from a diethylene glycol (DEG) solution, and reverse microemulsion with two different surfactants. Employing organic solvents and reagents significantly lowered the crystallization temperature to 600–800 °C, allowing single‑phase crystalline products to form in a single step. Morphology and magnetic properties were strongly influenced by the synthesis conditions. The heating efficiency in an alternating magnetic field correlated closely with the saturation magnetization, which was highest for the sol‑gel derived particles. All three methods produced weakly agglomerated, superparamagnetic nanoparticles; however, the sol‑gel samples exhibited the most promising hyperthermia performance (specific loss power ≈ 38 W g⁻¹).
PACS: 61.46.Df 75.75.Cd 81.20.Fw
Background
When magnetic materials are reduced to the nanoscale, their crystal structure, surface chemistry, and magnetic response can deviate markedly from bulk behavior, opening new opportunities for sensors, recording media, and medical technologies. Among the most promising biomedical applications is magnetic hyperthermia, wherein nanoparticles are locally heated to 43–45 °C under an alternating magnetic field to selectively kill tumor cells. Achieving uniform, controllable heating requires nanoparticles that are small, weakly agglomerated, single‑domain, superparamagnetic, and possess high specific loss power (SLP).
Magnetite (Fe₃O₄) is widely studied for hyperthermia due to its high Curie temperature (~580 °C), but its heating is largely uncontrollable above 43 °C, risking damage to healthy tissues. Thus, alternative materials with Curie temperatures tuned to the therapeutic window are needed. Lanthanum‑strontium manganite (La₁₋ₓSrₓMnO₃, LSMO) perovskites exhibit a magnetic phase transition near 45 °C, providing an inherent temperature limit suitable for hyperthermia.
However, perovskite manganites crystallize only after high‑temperature treatment (>1100 °C) when synthesized from aqueous solutions, leading to particle growth and aggregation. Non‑aqueous and organic‑based synthesis routes—such as precipitation from DEG, microemulsion, and sol‑gel—can lower the crystallization temperature, reduce agglomeration, and allow finer control over particle size and magnetic properties. This study compares these three methods for producing weakly agglomerated, single‑phase LSMO nanoparticles and evaluates their suitability for magnetic hyperthermia.
Methods
Synthesis Routes
Sol‑gel: Metal nitrates (La(NO₃)₃, Mn(NO₃)₂, Sr(NO₃)₃) were dissolved in deionized water. Citric acid (CA) and ethylene glycol (EG) were added in a molar ratio of CA:EG = 1:4, with a salt to gel‑forming mixture ratio of 1:10. The solution was heated to 80 °C while stirring, forming a polymer gel via polyesterification. Pyrolysis at 200 °C yielded a precursor powder, which was subsequently heat‑treated at 600–800 °C for 2 h to achieve crystallization.
Precipitation from DEG: Concentrated aqueous metal nitrate solutions (La(NO₃)₃ Cₗₐ = 1.2 M, Mn(NO₃)₂ Cₘₙ = 1.5 M, Sr(NO₃)₃ Cₛᵣ = 1.6 M) were mixed with diethylene glycol (DEG) under argon at 200 °C. Sodium hydroxide (0.5 M in DEG) was added dropwise, and the mixture was stirred at 200–220 °C for 1 h. After cooling, the precursor was coated with oleic acid, isolated by centrifugation, washed, and dried at 30–50 °C. Crystallization was achieved by heating at 600–800 °C for 2 h.
Reverse microemulsion: Aqueous solutions of the same metal nitrates were combined with cetyltrimethylammonium bromide (CTAB) or Triton X‑100 as surfactants, n‑butanol as a co‑surfactant, cyclohexane as the organic phase, and water as the dispersed phase. Aqueous ammonia served as precipitant. Two microemulsions (M₁ with salts, M₂ with precipitant) were prepared and mixed dropwise at 70 °C, followed by 1 h stirring. The resulting precipitate was collected by centrifugation, washed with isopropanol and water, and heat‑treated at 600–800 °C for 2 h.
Crystalline structure was confirmed by X‑ray diffraction (CuKα radiation, DRON‑4 diffractometer). Morphology and size distribution were examined using transmission electron microscopy (TEM, JEOL JEM‑1400). Magnetic properties were measured with a vibrating sample magnetometer (VSM, LDJ‑9500). Heating efficiency was assessed by preparing magnetic fluids (0.1 % agarose) and measuring temperature rise under a 300 kHz, 9.5 kA/m AC magnetic field. Specific loss power (SLP) was calculated from the initial slope of the temperature‑time curve using the formula: $$\mathrm{SLP}=\frac{C_{\mathrm{fluid}}\cdot V_{s}}{m_{\text{powder}}}\cdot\frac{\mathrm{d}T_{\text{fluid}}}{\mathrm{d}\tau}$$
Results and Discussion
In all three synthesis routes, the as‑prepared powders were initially amorphous. X‑ray diffraction (Figure 1) shows that crystallization initiates at 600 °C and completes by 800 °C, regardless of the method, demonstrating that non‑aqueous or organic‑based routes lower the required temperature by ~300 °C compared with conventional aqueous synthesis.
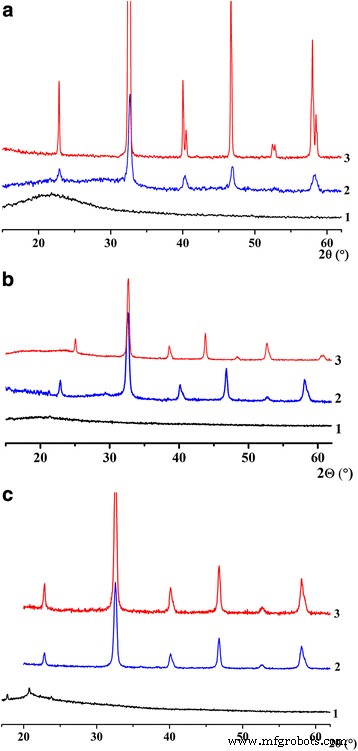
Figure 1. X‑ray diffraction of LSMO nanoparticles prepared via sol‑gel (a), DEG precipitation (b), and reverse microemulsion (c) at 1–200 °C, 2–600 °C, and 3–800 °C.
TEM imaging (Figure 2) reveals that all samples exhibit narrow size distributions, with average diameters of 20–40 nm—well below the ~70 nm threshold for single‑domain behavior in manganites. The microemulsion route, particularly with Triton X‑100, yields the smallest particles due to the larger hydrophilic headgroup that restricts the nanoreactor volume.
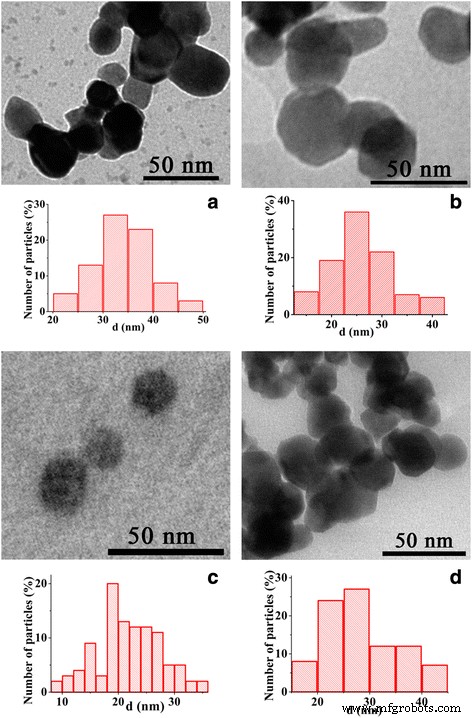
Figure 2. TEM images and size distributions for (a) sol‑gel, (b) DEG, (c) Triton X‑100 microemulsion, and (d) CTAB microemulsion nanoparticles.
Magnetization curves (Figure 3) indicate negligible coercivity (<12 A/m) at room temperature for all samples, confirming superparamagnetic behavior. Saturation magnetization decreases with particle size; the sol‑gel nanoparticles exhibit the highest value (~60 emu g⁻¹), while the microemulsion samples reach ~35 emu g⁻¹.
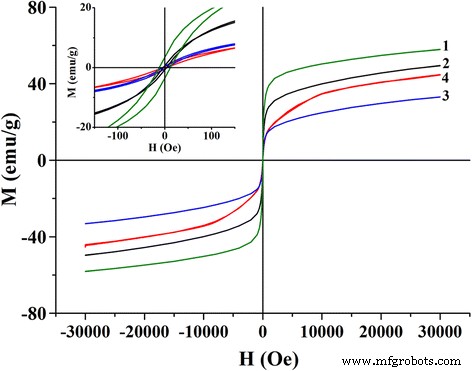
Figure 3. Magnetization versus field for sol‑gel (1), DEG (2), Triton X‑100 (3), and CTAB (4) nanoparticles. Inset shows low‑field behavior.
Heating tests (Figure 4) demonstrate that SLP is directly proportional to saturation magnetization. Sol‑gel derived nanoparticles reach ~38 W g⁻¹, whereas DEG and microemulsion samples achieve 20–25 W g⁻¹. Importantly, the temperature stabilizes after a brief period, ensuring controlled heating within the therapeutic window (43–45 °C) and preventing overheating of healthy tissues.
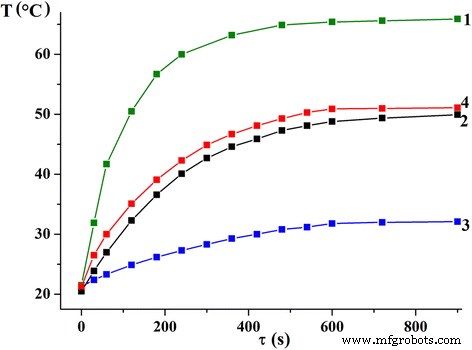
Figure 4. Temperature versus time for sol‑gel (1), DEG (2), Triton X‑100 (3), and CTAB (4) nanoparticles under AC field.
Overall, the synthesis route exerts a pronounced influence on particle morphology, magnetic properties, and hyperthermia efficiency. The sol‑gel method, offering the highest magnetization and SLP, is particularly attractive for biomedical applications.
Conclusions
La₁₋ₓSrₓMnO₃ nanoparticles produced via sol‑gel, DEG precipitation, and reverse microemulsion all crystallize in a single phase at 600–800 °C, a significant reduction from the 1100 °C required for conventional aqueous synthesis. The particle size remains in the 20–40 nm range, ensuring single‑domain, superparamagnetic behavior. Saturation magnetization scales inversely with particle size, directly impacting heating performance. Sol‑gel derived nanoparticles exhibit the highest magnetization and SLP (~38 W g⁻¹), making them the most promising candidates for magnetic hyperthermia. All routes yield weakly agglomerated particles, but the sol‑gel approach offers the best combination of magnetic properties and heating efficiency.
Nanomaterials
- Semiconductor Nanoparticles: Properties, Synthesis, and Applications
- Cobalt‑Doped FeMn₂O₄ Spinel Nanoparticles: Size‑Controlled Synthesis and Magnetic Behavior
- Efficient Synthesis and Optical Characterization of Sub‑Micron Selenium Nanocrystals and Nanorods
- Effect of Contact Non‑Equilibrium Plasma on Mn_xFe_3−xO_4 Spinel Nanoparticles: Structural and Magnetic Insights
- Fe₃O₄–PNIPAAm Nanocomposites: Preparation Method Determines Antibacterial Efficacy and Physicochemical Performance
- Green Synthesis of Metal‑Oxide Nanoparticles with Gum Karaya and Their Ecotoxicological Impact on *Chlamydomonas reinhardtii*
- Zinc Oxide Nanoparticles: Antimicrobial Properties, Mechanisms, and Applications
- Ligand‑Free Iridium Nanoparticles: A Simple Aqueous Synthesis and Demonstrated In‑Vitro Biocompatibility
- Rapid One‑Pot Synthesis of Monodisperse CoFe₂O₄@Ag Core–Shell Nanoparticles with Exceptional Plasmonic and Magnetic Properties
- PVP‑Enhanced SnO₂ Nanoflowers Deliver Ultra‑Fast, Highly Selective H₂S Sensing



