Easily Synthesized Wormhole‑Like Mesoporous SnO₂ via Evaporation‑Induced Self‑Assembly: Superior Ethanol Gas‑Sensor Performance
Abstract
Wormhole‑like mesoporous tin oxide (SnO₂) was prepared through a simple evaporation‑induced self‑assembly (EISA) route. The resulting materials were evaluated for their gas‑sensing capabilities against several target gases. Calcination temperature emerged as a critical factor: the sample calcined at 400 °C exhibited outstanding selectivity toward ethanol vapour, coupled with an optimal operating temperature, rapid response, and recovery. The high specific surface area and interconnected pore network of the mesoporous SnO₂ provide abundant active sites and facilitate the diffusion of reactant molecules, thereby enhancing the adsorption of oxygen species and the interaction with target gases. A plausible synthesis pathway and sensing mechanism are proposed, underscoring the material’s promise for practical gas‑sensor applications.
Background
Among semiconducting metal oxides, tin dioxide (SnO₂) – a wide‑bandgap (3.6 eV) rutile semiconductor – has attracted significant interest for applications ranging from lithium‑ion battery anodes[1] to photocatalysis[3–5] and gas sensors[7]. Its high electronic mobility, chemical stability, and non‑stoichiometric flexibility make it an attractive candidate for detecting a variety of analytes, including acetone[24], nitrogen dioxide[25], toluene[26], ethanol[27], formaldehyde[28–29], and methanol[30].
Gas‑sensor performance is intrinsically linked to the microstructure of SnO₂. Nanostructuring strategies that produce high surface areas and tailored pore architectures have been shown to significantly improve sensitivity and selectivity[18–23]. Mesoporous SnO₂, in particular, offers a narrow pore‑size distribution and large active surface, facilitating efficient gas diffusion and interaction[31–34]. While several synthesis approaches exist (sol‑gel, sonochemical, template‑based), a straightforward, scalable, and cost‑effective route remains desirable.
The EISA technique combines micelle formation, solvent evaporation, and template removal to produce uniform mesoporous nanostructures under mild conditions[33,34]. In this study, we employ EISA to fabricate wormhole‑like mesoporous SnO₂ and systematically investigate how calcination temperature influences structure and gas‑sensing performance.
Methods
Analytical‑grade reagents were sourced from Sinopharm Chemical Reagent Co. Ltd. A typical synthesis involved dissolving 0.42 g SnCl₄·5H₂O and 0.336 g citric acid in 10 mL deionized water, followed by adding 0.144 g P123 (EO₂₀(PO₇₀)EO₂₀) dissolved in 10 mL ethanol. One milliliter of nitric acid was introduced as a condensation inhibitor. The P123 solution was incorporated into the tin precursor with vigorous stirring, then the mixture was sealed with polyethylene film, stirred at 60 °C for 2 h, and subsequently dried at 60 °C to promote solvent evaporation. The resulting solid was calcined in air for 3 h at 350, 400, or 450 °C, yielding SnO₂‑350 °C, SnO₂‑400 °C, and SnO₂‑450 °C, respectively.
Phase identification was performed using a D/MAX2550VB⁺ X‑ray diffractometer (Cu Kα, λ = 1.5405 Å) over 2θ = 0.5–80° at 0.02°/s. TEM and HRTEM imaging employed a Tecnai G²‑20ST microscope (220 kV). N₂ adsorption–desorption isotherms were measured at 77 K with an ASAP 2020 analyzer; BET surface areas and BJH pore‑size distributions were calculated accordingly. FTIR spectra were recorded on a Nicolet Nexus 670 (KBr pellets), and photoluminescence (PL) was measured with a HITACHI FL‑4500 (excitation 310 nm).
Sensor fabrication involved mixing mesoporous SnO₂ powders with terpineol saturated with methylcellulose to form a slurry, coating it onto an alumina tube (gold electrodes and Pt wires), drying at ambient conditions, and heating at 350 °C for 3 h. A Ni‑Cr coil served as the heating element.
Gas‑sensing measurements used a WS‑30A system (Weisheng Electronics). Sensors were aged at 350 °C for 48 h to ensure stability. The response was defined as Ra/Rg (air vs. target gas). Response and recovery times were the intervals to reach 90 % of the total resistance change. Humidity effects were examined at 24, 43, 75, and 97 % RH using saturated salt solutions.
Results and Discussion
Figure 1 illustrates the EISA synthesis mechanism: tin species and P123 micelles self‑assemble into a liquid‑crystalline mesophase, subsequently forming a crown‑ether‑type complex that inhibits SnO₂ growth. Upon slow evaporation and template removal by calcination, a wormhole‑like mesoporous network emerges, conferring high surface area and pore volume.
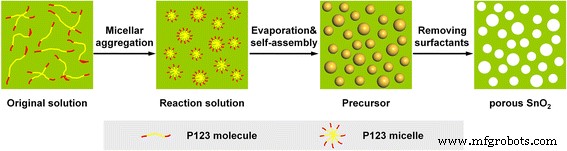
Schematic illustration of the synthetic procedure of mesoporous SnO₂ powders
XRD analysis (Figure 2) confirms the rutile SnO₂ phase (JCPDS 41‑1445) across all calcination temperatures. Small‑angle XRD peaks at ~1.7° (mesostructure) are pronounced for SnO₂‑400 °C but absent for SnO₂‑350 °C, indicating optimal mesopore formation at 400 °C. Higher calcination (450 °C) broadens the mesoporous peak, suggesting slight structural collapse. Wide‑angle patterns show increasing crystallinity with calcination temperature, yet the nanoscale crystallites observed in TEM are consistent across samples.
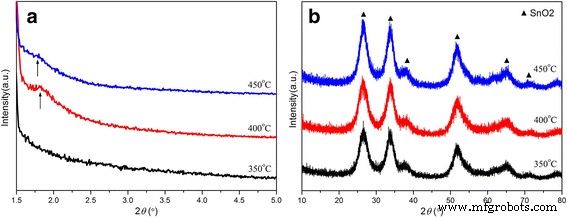
a Small‑angle and b wide‑angle XRD patterns of mesoporous SnO₂ powders
BET analysis (Figure 3) shows specific surface areas of 281 m² g⁻¹ (350 °C), 356 m² g⁻¹ (400 °C), and 307 m² g⁻¹ (450 °C). Pore volumes (0.14, 0.28, 0.22 cm³ g⁻¹) and average diameters (2.9, 5.3, 4.7 nm) peak at 400 °C, reflecting complete template removal and interconnected pore networks.
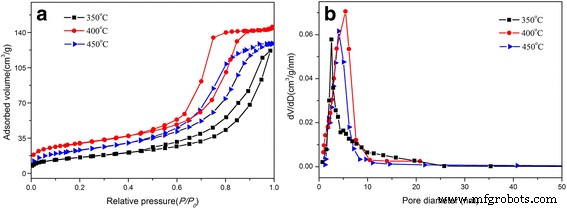
a Nitrogen adsorption–desorption isotherms. b Corresponding BJH pore size distribution curves of mesoporous SnO₂ powders
TEM images (Figure 4) reveal wormhole‑like mesopores formed by the aggregation of uniform nanoparticles. The mesostructure is most pronounced for SnO₂‑400 °C, with clear lattice fringes (0.32 nm) corresponding to the (110) plane. Even at 450 °C, the wormhole network remains intact, underscoring thermal robustness.

TEM images of a SnO₂‑350 °C, b SnO₂‑400 °C (inset SAED), and d SnO₂‑450 °C. c HRTEM of SnO₂‑400 °C
FTIR spectra (Figure 5a) show disappearance of organic template peaks (1658, 2803, 1381, 1349 cm⁻¹) upon calcination above 350 °C, confirming successful template removal. PL spectra (Figure 5b) exhibit two emission bands at ~390 nm and ~458 nm for samples calcined at 400 and 450 °C, indicative of defect‑related luminescence. The 350 °C sample shows stronger PL due to residual organic species and surface defects.
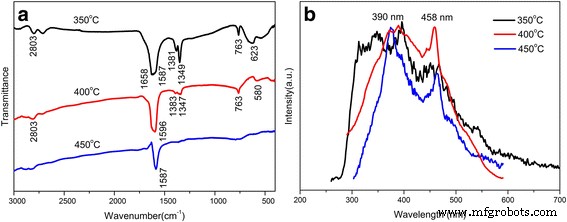
a FTIR spectra. b Photoluminescence spectra of mesoporous SnO₂ samples (excitation 310 nm)
Gas‑sensing tests (Figure 6) demonstrate that the SnO₂‑400 °C sensor achieves the highest response (Ra/Rg = 41.6) to 200 ppm ethanol at 200 °C, with response and recovery times of 31 s and 2 s, respectively. Response increases with ethanol concentration up to 200 ppm, then plateaus, indicating saturation at higher levels. Selectivity tests (Figure 6d) confirm preferential detection of ethanol over methanal, methanol, and acetone. Relative humidity experiments (Figure 7a) reveal that the sensor’s performance diminishes at higher RH, yet the 400 °C sample retains the most robust response. Long‑term stability over 10 days shows <10 % deviation, underscoring reliability.
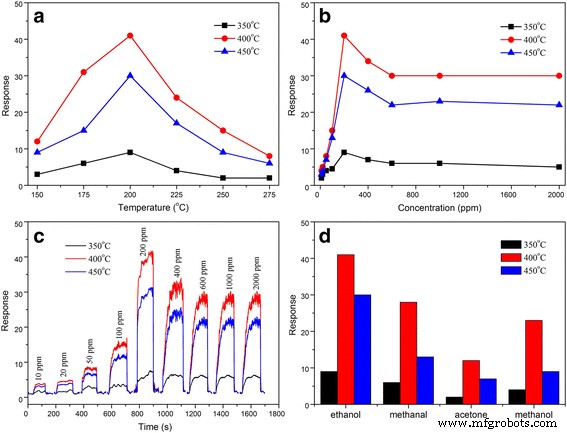
a Responses to 200 ppm ethanol at various operating temperatures, b responses vs. ethanol concentration, c response–recovery curve at 200 °C, d gas responses to ethanol, methanal, methanol, and acetone at 200 °C
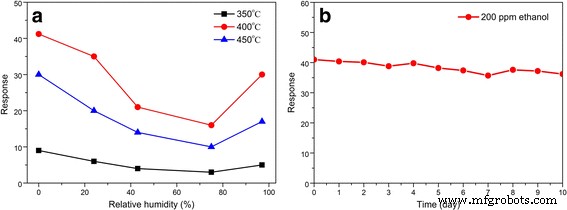
a Response vs. RH at 200 °C, b Long‑term stability over 10 days
Mechanistically, the high surface area and wormhole pores facilitate rapid oxygen adsorption (O₂⁻, O⁻) and efficient electron depletion. Upon exposure to ethanol, surface‑bound O⁻ reacts to form water and CO₂, releasing electrons back to the conduction band and reducing resistance. The interconnected pore network ensures uniform gas distribution and minimizes diffusion barriers, accounting for the superior sensitivity and rapid kinetics of the 400 °C sample.
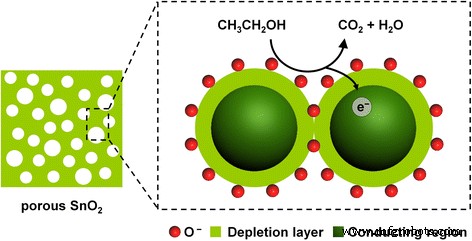
Schematic illustration of ethanol sensing mechanism of mesoporous SnO₂
Conclusions
We have demonstrated a facile, scalable EISA route to produce wormhole‑like mesoporous SnO₂ with high thermal stability. Calcination at 400 °C yields the most favorable microstructure—high surface area, interconnected pores, and complete template removal—leading to exceptional ethanol‑gas sensing: response 41.6 at 200 °C for 200 ppm, response time 31 s, and recovery time 2 s. The material’s robustness across humidity levels and over 10 days of operation suggests strong potential for environmental monitoring, medical diagnostics, and industrial safety applications.
Nanomaterials
- Efficient Synthesis and Optical Characterization of Sub‑Micron Selenium Nanocrystals and Nanorods
- Cellulose–POSS–Silica/Gold Core–Shell Hybrid Nanocomposites: One‑Step Sol‑Gel Synthesis and Multifunctional Properties
- Effect of Contact Non‑Equilibrium Plasma on Mn_xFe_3−xO_4 Spinel Nanoparticles: Structural and Magnetic Insights
- Efficient One‑Step Photo‑Ultrasonic Synthesis of rGO/Ag₃PO₄ Quantum‑Dot Composites for Enhanced Visible‑Light Photocatalysis
- Green Synthesis of Metal‑Oxide Nanoparticles with Gum Karaya and Their Ecotoxicological Impact on *Chlamydomonas reinhardtii*
- PVP‑Enhanced SnO₂ Nanoflowers Deliver Ultra‑Fast, Highly Selective H₂S Sensing
- Hierarchical Au@CdS‑CdS Nanoflowers: Stepwise Fabrication and Superior Photocatalytic Performance
- Efficient Fabrication of Mesostructured NR/SiO₂ Nanocomposites with Superior Thermal Stability and Hydrophobicity
- Enhanced Photocatalytic and Antibacterial Performance of Annealed Tin Oxide Nanostructures: A Molecular Docking Study
- Eco‑Friendly, Cost‑Effective Synthesis of Tin Oxide Nanoparticles: A Comprehensive Review of Methods, Formation Mechanisms, and Applications



