Blue‑Emitting Mn‑Alloyed CdTe Quantum Dots: Synthesis, Structural Insight, and Electrochemical Characterization
Abstract
We report the first synthesis of water‑soluble CdTe quantum dots alloyed with Mn²⁺ that emit blue photoluminescence. Using a controlled ion‑exchange protocol at neutral pH, we produced a series of Cd1‑xMnxTe QDs with a narrow size distribution (average diameter 2.3 ± 0.3 nm). Photoluminescence peaks shift from 542 nm to 496 nm as Mn content increases, demonstrating a blue‑shift driven by band‑gap modification. Cyclic voltammetry reveals Mn‑induced states within the band gap, enabling an estimate of their energy positions. Electron paramagnetic resonance confirms the presence of isolated and aggregated Mn²⁺ ions, and X‑ray diffraction confirms the cubic Cd1‑xMnxTe phase. These findings establish Mn‑alloyed CdTe QDs as a promising platform for fluorescence sensing and magnetic imaging.
Background
Quantum dots (QDs) are tunable nanoscale semiconductors whose optical properties can be engineered by doping or alloying. Mn‑alloyed QDs are particularly attractive for biosensing and magnetic resonance imaging due to their combined fluorescence and magnetic characteristics. Previous work has shown successful Mn doping in ZnS, ZnSe, CdSe, CdS, and CdTe/CdS core/shell systems. However, incorporating Mn into CdTe in aqueous media has proven challenging, largely because alkaline conditions promote hydroxide precipitation and inhibit ion exchange. Our strategy leverages a neutral‑pH environment to suppress Mn(OH)2 formation, enabling efficient ion exchange of Cd²⁺ by Mn²⁺ and yielding blue‑emitting Cd1‑xMnxTe QDs.
Methods
Synthesis of CdTe and Cd1‑xMnxTe‑alloyed QDs
Thioglycolic acid–stabilized CdTe nanoclusters were prepared at room temperature via a modified three‑step method. The resulting colloids were divided into six 50 mL aliquots. MnSO4 (1, 5, 10, 15, 20 % of Cd²⁺ concentration) was added under sonication to each aliquot, facilitating ion exchange. A final microwave anneal (700 W, 3 min) completed the alloying. One aliquot of unmodified CdTe served as a control.
Characterization
Absorption and photoluminescence spectra were recorded at room temperature with an OceanOptics USB‑2000 spectrophotometer. Cyclic voltammograms (CV) were obtained using a ΠИ‑50‑1 potentiostat in a three‑electrode setup (Pt working, glassy carbon counter, Ag/AgCl reference) from −2 V to 2 V at 100 mV s⁻¹. Transmission electron microscopy (TEM) employed a Selmi TEM‑125 K microscope at 80 kV. Elemental composition was quantified by AES (С115М1). EPR spectra were recorded on a Radiopan X‑band spectrometer at 300 K with 100 kHz modulation. Powder X‑ray diffraction (XRD) used a Bruker D8 Advance diffractometer; phases were identified against ICSD 040413.
Results and Discussion
Structural Characterization
TEM Analysis
TEM images of Cd0.91Mn0.09Te QDs show a uniform spherical population with an average diameter of 2.3 ± 0.3 nm, matching the size inferred from optical spectra. Aggregates of smaller QDs were occasionally observed but did not affect the overall size distribution.
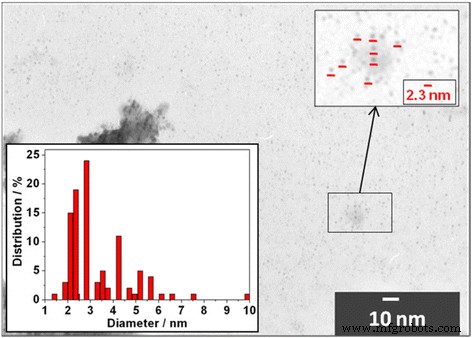
TEM image of Cd0.91Mn0.09Te QDs. Inset: size‑distribution histogram.
Atomic Emission Spectroscopy
Only ~20 % of the Cd²⁺ precursor participates in QD formation, consistent with previous findings. AES confirmed Mn incorporation across the series, with measured Cd:Mn ratios matching the intended stoichiometry. Mn alloying efficiency increased with higher Mn²⁺ loading, indicating a threshold for effective ion exchange.
Optical Properties
Absorption spectra exhibit a systematic blue‑shift with rising Mn content, while PL peaks shift from 542 nm (pure CdTe) to 496 nm (high Mn). The PL intensity diminishes at higher Mn levels, likely due to non‑radiative pathways introduced by excess Mn²⁺ or thioglycolate quenching. The observed spectral shifts confirm band‑gap widening induced by Mn alloying.
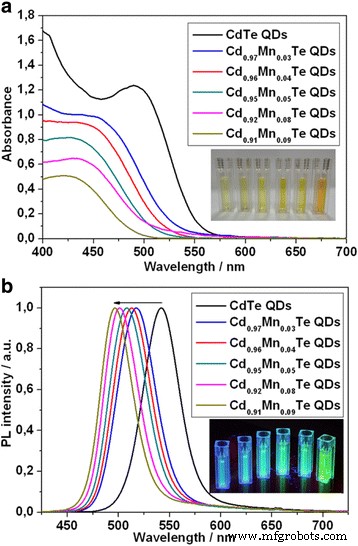
Absorption (a) and normalized PL (b) spectra of CdTe and Cd1‑xMnxTe QDs. Inset: daylight (a) and UV (b) images of the colloids.
Cyclic Voltammetry
CV of CdTe shows anodic (1.48 V) and cathodic (−1.00 V) peaks, yielding a band‑gap of 2.48 eV, in agreement with optical data. For Mn‑alloyed QDs, the separation between anodic and cathodic peaks remains ~2.18 V, indicating the emergence of Mn‑related states within the band gap. This supports a thermally assisted back‑energy‑transfer mechanism that increases the effective band‑gap energy as Mn content rises.
XRD Analysis
All samples display the cubic CdTe (111) reflection at 25°, which shifts to higher angles with increasing Mn, confirming lattice contraction. For higher Mn loadings, the (111) peak diminishes, while the (200)/(220) peaks broaden, indicating formation of a thicker Cd1‑xMnxTe shell around a CdTe core or complete alloying at high Mn. The XRD data corroborate successful Mn substitution.
EPR Measurements
Cd0.97Mn0.03Te QDs exhibit a complex EPR spectrum comprising a broad Gaussian line (g = 2.0069) from Mn²⁺ clusters and two sextets (Δ ≈ 10 mT and 6 mT) from isolated Mn²⁺ ions. Analysis with a spin‑Hamiltonian confirms surface‑localized Mn²⁺ (signal 2) and lattice‑substituted Mn²⁺ (signal 3). The presence of both isolated and aggregated Mn⁺⁺ ions explains the magnetic behavior observed in CV.
Conclusions
We have developed a reproducible, aqueous synthesis of blue‑emitting Cd1‑xMnxTe QDs that retain a narrow size distribution while exhibiting a tunable PL shift from 542 nm to 496 nm. Structural (XRD, TEM) and spectroscopic (PL, CV, EPR) analyses confirm successful Mn incorporation and reveal a Mn‑induced widening of the band gap through thermally assisted back‑energy transfer. These findings establish Mn‑alloyed CdTe QDs as a robust platform for dual‑function optical and magnetic applications.
Nanomaterials
- Phosphine‑Free Synthesis of Reabsorption‑Suppressed ZnSe/CdS/ZnS Core–Shell Quantum Dots and Their Application in Sensitive CRP Detection
- Eco-Friendly Synthesis of InP/ZnS Core/Shell Quantum Dots for High-Performance Heavy-Metal-Free LEDs
- Water‑Soluble Antimony Sulfide Quantum Dots: Efficient Synthesis and Superior Photo‑Electric Performance
- Efficient One‑Step Photo‑Ultrasonic Synthesis of rGO/Ag₃PO₄ Quantum‑Dot Composites for Enhanced Visible‑Light Photocatalysis
- High‑Performance Dual‑Emissive Mn‑Doped InP/ZnS Quantum Dots with 78 % Photoluminescence Quantum Yield: A Growth‑Doping Approach
- One‑Pot Room‑Temperature Synthesis of 10‑nm 2D Ruddlesden–Popper Perovskite Quantum Dots with Tunable Emission
- Tailoring Optical Properties of InAs Quantum Dots with InAlAs Interlayers in GaAsSb Strain‑Reducing Layers
- High‑Quality In₂S₃ Quantum Dots Synthesized at Room Temperature for Ultra‑Sensitive UV Photodetectors
- Synthesis, Structure, and Superior Capacitance of Sulfur‑Nitrogen Co‑Doped Graphene Quantum Dots
- Eco‑Friendly One‑Pot Hydrothermal Synthesis of Water‑Soluble WS₂ Quantum Dots for Sensitive Luminescent Detection of Hydrogen Peroxide and Glucose



