Highly Efficient, Cd‑Free Cu‑Doped ZnInS/ZnS Core–Shell Nanocrystals: Tunable Synthesis and Superior Photophysical Performance
We report the synthesis of composition‑tunable, cadmium‑free Cu‑doped ZnInS/ZnS core/shell colloidal nanocrystals (CNCs) via a colloidal, non‑injection route. By employing oleate precursors instead of powders, the resulting CNCs exhibit near‑defect‑free photoluminescence. Adjusting the Zn/In ratio controls Cu incorporation, allowing emission tunability from 550 to 650 nm. A ZnS shell boosts the quantum yield (QY) from 6 % to 65 %. Through careful stoichiometric control, the CNCs transition from mixed deep‑trap/dopant emission to a dominant, Stokes‑shifted dopant‑related peak. Steady‑state and time‑resolved fluorescence spectroscopy elucidate the emission mechanism. As a proof‑of‑concept, blue‑LED‑excited Cu‑doped ZnInS/ZnS CNCs generate white light with CIE coordinates (0.33, 0.31), a color‑temperature of 3,694 K, a luminous efficacy of 170 lm W⁻¹, and a CRI of 88. Colloidal nanocrystals (CNCs) have become pivotal in optoelectronics due to their size‑ and composition‑dependent emission across the visible spectrum. The use of toxic heavy metals such as Cd, Pb, and Te in traditional CNCs (e.g., CdSe, ZnCdS, ZnCdSe) poses environmental and regulatory challenges, limiting their deployment in devices that interact with biological systems. Consequently, there is a pressing need for cadmium‑free, high‑performance emitters. Transition‑metal‑doped ternary CNCs (e.g., Cu²⁺, Mn²⁺) have emerged as promising candidates, offering tunable, efficient photoluminescence and large Stokes shifts that mitigate self‑absorption. Cu‑doped ZnInS and related alloys combine a wide direct band gap with the ability to host dopant ions at substitutional or interstitial sites, thereby enabling controllable emission properties. The incorporation of ZnS as a shell material further passivates surface defects, suppresses non‑radiative recombination, and enhances quantum yield. In this study, we synthesize highly efficient, cadmium‑free Cu‑doped ZnInS/ZnS core/shell CNCs using a modified, non‑injection colloidal approach. By varying the Zn/In precursor ratio, we tune the Cu incorporation and achieve emission wavelengths spanning 550–650 nm. Detailed optical analyses reveal that the dominant emission originates from Cu‑related states, while deep‑trap contributions are minimized through shell passivation. Finally, we integrate these CNCs with commercial blue LEDs to produce white light exhibiting excellent color fidelity and luminous efficacy. Zinc acetate, indium acetate, copper acetate, sulfur powder, dodecanethiol, oleic acid, oleylamine, and 1‑octadecene were sourced from Sigma Aldrich and used without further purification. Precursors were dissolved in oleate form to yield 0.1 M Zn‑oleate, 0.1 M In‑oleate, 0.01 M Cu‑oleate, and 0.4 M sulfur‑in‑ODE solutions. The ZnS shell precursor was prepared as a 1.756 g (8 mmol) Zn‑oleate solution in 20 mL of ODE/OAm. All solutions were degassed under vacuum at 95 °C, then heated to 160 °C under Ar until clear. In an Ar‑filled flask, 2 mL ODE and 1 mL DDT were degassed, then 1 mL each of Zn‑oleate and In‑oleate (0.1 mmol), 0.5 mL Cu‑stock (0.01 mmol), and 0.5 mL sulfur‑stock (0.2 mmol) were added. The mixture was heated to 220 °C and maintained for 20 min. Quenching to 60 °C, followed by toluene addition and ethanol precipitation, yielded purified core CNCs. After 20 min of core growth, the reaction was cooled to 100 °C. A 1 mL aliquot of the Zn‑oleate shell precursor was injected, and the temperature was raised to 240 °C for 20 min to allow shell growth. Cooling to 60 °C, toluene addition, and standard purification yielded core/shell CNCs. Mixed‑composition CNC films were drop‑cast onto quartz wafers and mounted over a 455 nm blue LED. Optical characterization employed an integrating sphere and an Ocean Optics Maya 2000 spectrometer; color metrics were computed with MATLAB. UV‑Vis absorption (Varian‑Cary 100), PL/PLE (Cary Eclipse), TEM (FEI Tecnai Osiris 200 kV), XRD (Cu Kα), time‑correlated single‑photon counting (Pico‑Quant FluoTime 200), and QY (de Mello method) were used to assess structural and optical properties. TEM images reveal monodisperse, nearly spherical core CNCs (average diameter 2.5 nm) and triangular core/shell CNCs (average diameter 4.5 nm). XRD patterns confirm a zinc‑blende lattice without Cu‑related phase separation, with a slight shift to higher angles after shell growth, indicative of Zn incorporation. TEM images of (a) ZnInS:Cu core and (b) ZnInS:Cu/ZnS core/shell CNCs; (c) XRD pattern of core and core/shell CNCs. Core CNCs exhibit a broad defect‑related emission (~450 nm) and a Stokes‑shifted Cu‑induced peak (~600 nm) with a 6 % QY. Shell deposition suppresses the defect emission and boosts the Cu‑related peak, achieving a 65 % QY. Time‑resolved PL shows lifetime extension from 92 ns (core) to 283 ns (core/shell), confirming reduced surface recombination. Using indium oleate as precursor eliminates low‑energy trap tails observed with powdered indium. Cu concentration studies reveal a maximum QY of 50 % at 2 % Cu, slightly decreasing at higher doping due to defect formation. UV‑Vis absorption shifts subtly with Cu loading, reflecting size and band‑gap changes. PL spectra of (a) ZnInS:Cu/ZnS CNCs (method A: powdered indium precursor) and (method B: indium oleate); (b) undoped ZnInS/ZnS vs Cu‑doped ZnInS/ZnS CNCs. By varying the Zn/In ratio, we tune the host band‑gap from 3.67 to 4.02 eV, shifting the Cu‑induced emission continuously from 550 to 650 nm. The FWHM remains 90–110 nm, characteristic of dopant‑related broad emission. Zn diffusion from the shell into the core during growth reduces Zn vacancy states, thereby suppressing deep‑trap emission and enhancing dopant dominance. (a) UV‑Vis absorption and (b) photoluminescence spectra of ZnInS:Cu/ZnS CNCs as a function of Zn/In stoichiometry. QY values for Zn/In ratios 0.11, 0.33, 0.53, and 1.0 are 56 %, 65 %, 55 %, and 48 %, respectively. Inset: calculated band‑gap. (c) PL peak shift and QY vs Zn/In. (d) PL decay curves for different Zn/In ratios. Lifetime measurements across Zn/In ratios show average lifetimes of 374, 283, 226, and 184 ns for emission peaks at 540, 560, 590, and 630 nm, respectively, confirming dopant‑mediated recombination. The long lifetimes (hundreds of ns) rule out surface‑trap‑dominated emission. Emission mechanisms were further clarified via PL excitation spectra: excitation profiles match the absorption spectrum, indicating energy transfer from the host lattice to Cu states rather than direct Cu excitation. As a demonstration, we fabricated white‑light emitting diodes (WLEDs) by combining green, yellow, and orange‑emitting CNCs with a 455 nm blue LED. Optimal weight ratios (G : Y : O = 15 : 1 : 0.75) yielded CIE coordinates (0.333, 0.313), a CCT of 3,694 K, LER of 170 lm W⁻¹, and CRI of 88—meeting indoor lighting standards. Single‑color CNCs produced lower CRI, underscoring the advantage of multi‑emissive systems. Emission spectra of green, yellow, and orange ZnInS:Cu/ZnS CNCs integrated on a blue LED driven at different currents. We have developed cadmium‑free, Cu‑doped ZnInS/ZnS core/shell CNCs with defect‑free, symmetric emission and a tunable band‑gap (3.67–4.02 eV). The Cu‑induced, Stokes‑shifted emission spans 550–650 nm, achieving a record 65 % QY. Time‑resolved spectroscopy confirms that dopant states dominate the radiative process. These high‑performance CNCs enable white‑light generation with excellent color fidelity, positioning them as strong candidates for next‑generation lighting and display technologies. Conduction band Color coordinate temperature Commission Internationale de l’Enclairage Colloidal nanocrystals Colloidal quantum dots Color rendering index Full width at half maximum Luminous efficacy of optical radiation Photoluminescence Photoluminescence excitation Quantum dot‑based light‑emitting diode Quantum yield Time‑correlated single‑photon counting Transmission electron microscopy Time‑resolved fluorescence Zinc vacancy White‑light emission X‑ray diffraction Zinc interstitialAbstract
\n Introduction
\n Methods
\n Chemicals Used
\n Preparation of Stock Solutions
\n Synthesis of Cu‑doped ZnInS Core CNCs
\n Deposition of the ZnS Shell
\n White‑Light Emission
\n Characterization
\n Results and Discussion
\n 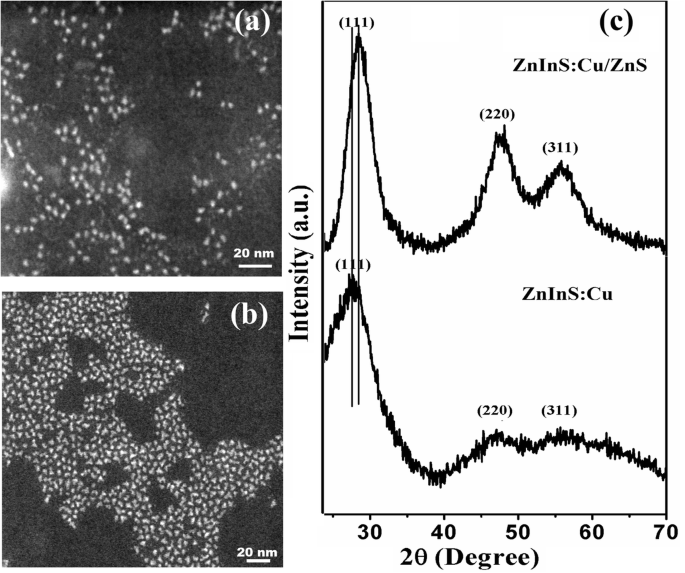 \n
\n 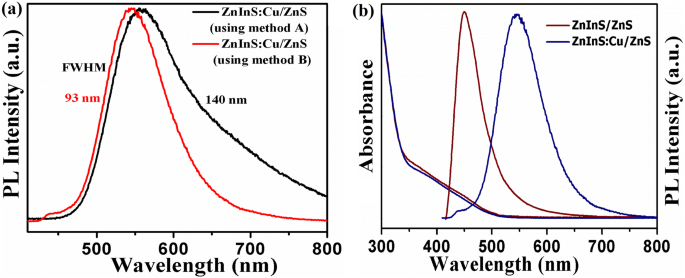 \n
\n 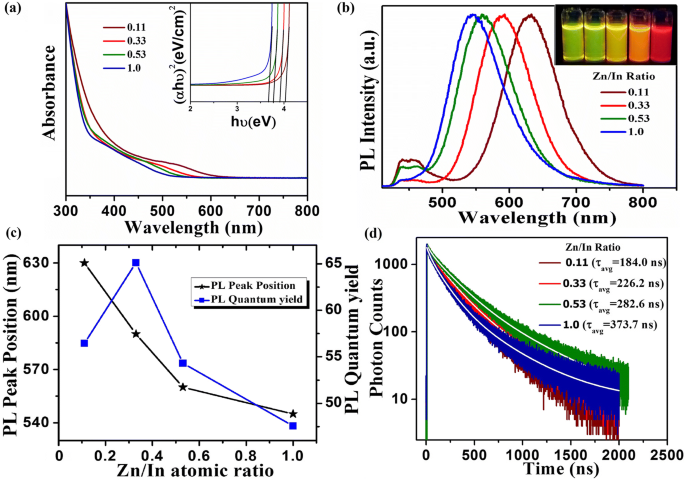 \n
\n 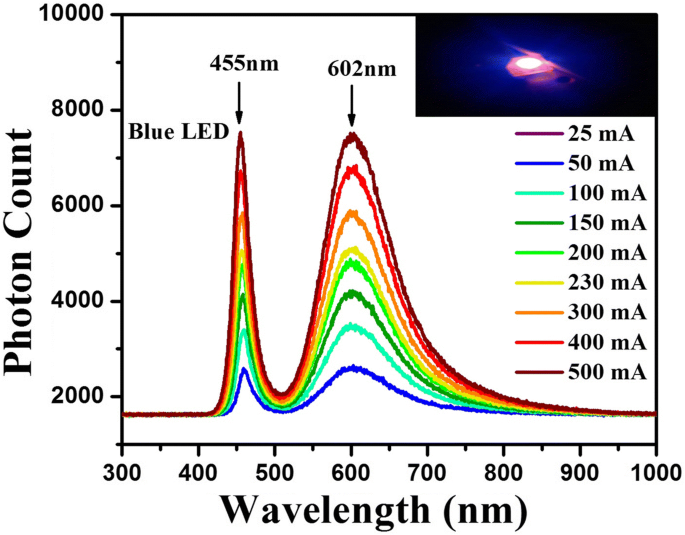 \n
\n Conclusions
\n Abbreviations
\n \n
\n
Nanomaterials
- Efficient Synthesis and Optical Characterization of Sub‑Micron Selenium Nanocrystals and Nanorods
- Phosphine‑Free Synthesis of Reabsorption‑Suppressed ZnSe/CdS/ZnS Core–Shell Quantum Dots and Their Application in Sensitive CRP Detection
- Eco-Friendly Synthesis of InP/ZnS Core/Shell Quantum Dots for High-Performance Heavy-Metal-Free LEDs
- Controlled Hydrothermal Synthesis of ZnO Nanocrystals for Enhanced Inverted Polymer Solar Cells
- Water‑Soluble Antimony Sulfide Quantum Dots: Efficient Synthesis and Superior Photo‑Electric Performance
- Blue‑Emitting Mn‑Alloyed CdTe Quantum Dots: Synthesis, Structural Insight, and Electrochemical Characterization
- High‑Performance Pr2CuO4 Nanosheet Adsorbent: Controlled Synthesis, Superior Selectivity for Malachite Green, and Mechanistic Insights
- Hierarchical Au@CdS‑CdS Nanoflowers: Stepwise Fabrication and Superior Photocatalytic Performance
- Co‑Doped 3d–4f Metal Polyoxometalates: Controlled Synthesis, Morphology, and Photoluminescence Properties
- Mastering 2D Semiconductor Nanomaterials: Precision Synthesis & Breakthrough Applications



