Al₂O₃ and SiO₂ Nanoparticles with Ultrasound Significantly Reduce Water Supercooling
Abstract
Al₂O₃ and SiO₂ nanoparticles, together with ultrasound, were employed to lower the supercooling degree of water. The study examined how nanoparticle concentration, contact angle, and ultrasonic intensity influence supercooling, while also monitoring nanoparticle dispersion stability during solidification. Results show that the combined use of ultrasound and nanoparticles reduces supercooling more effectively than either treatment alone. The reduction increases with higher ultrasonic intensity and nanoparticle concentration, and with lower nanoparticle contact angle. However, the combined effect never exceeds the sum of the individual effects, and in some cases it is even less than the effect of ultrasound alone. Maintaining nanoparticle dispersion requires that the joint treatment outperform ultrasound alone; otherwise, aggregation occurs, undermining the benefit. Optimal ranges of nanoparticle concentration, contact angle, and ultrasonic intensity that achieve both low supercooling and stable dispersion were identified, and the underlying mechanisms were discussed.
Introduction
Energy storage is a pivotal technology for addressing environmental concerns, energy shortages, and high operational costs. Latent‑heat thermal energy storage (LHTES) offers a high volumetric storage density, especially when water is used as the phase‑change material. Yet, water’s tendency to supercool during solidification limits its practical efficiency: a larger temperature difference between charging and discharging is required, reducing overall system performance. Thus, minimizing water supercooling is essential for advancing LHTES.
Nanoparticles are widely used as nucleating agents to control supercooling. Hydrophilic metal oxides such as TiO₂, Al₂O₃, and CuO, as well as hydrophobic carbon nanomaterials, can lower the Gibbs free energy barrier for ice nucleation by providing high‑surface‑area sites. Studies show that high‑specific‑area nanoparticles (e.g., graphene nanoplatelets at 0.02 wt.%) can eliminate supercooling, while TiO₂ at 1.0 wt.% only reduces it by ~70 %. Consequently, increasing the number of nucleation sites is more effective than enhancing hydrophilicity alone.
However, high‑area nanoparticles tend to aggregate, especially above 1.0–2.0 wt.%, compromising long‑term thermal performance. Ultrasound has emerged as an alternative strategy: acoustic cavitation can act as nucleation sites or generate pressure fluctuations that trigger ice formation. While the exact mechanism remains debated, evidence suggests that ultrasound reduces supercooling by creating or collapsing microbubbles.
Previous work combining graphene oxide nanoparticles with ultrasound demonstrated a synergistic reduction in supercooling, yet the phenomenon was not fully explained. Our own studies with TiO₂ nanoparticles revealed that ultrasound not only lowers supercooling but also promotes nanoparticle aggregation at high power. Therefore, a systematic investigation of Al₂O₃ and SiO₂ nanoparticles, which are hydrophilic and can remain stable in water, is needed to clarify how nanoparticle concentration, contact angle, and ultrasonic intensity jointly affect supercooling and dispersion.
Experimental
Al₂O₃ and SiO₂ nanoparticles (Aladdin Chemical Reagent Co. Ltd.) were selected for their strong water affinity. Contact angles were measured via static sessile drop using a DataPhysics OCA40 Micro goniometer; five replicates yielded <1 % variance (Fig. 1). Suspensions were prepared in de‑ionized water (pH 8, adjusted with NaOH) without surfactant. Nanoparticles were dispersed by 1‑h sonication (Sonics Vibra‑Cell, 600 W, 20 kHz). Concentrations of 0.2, 0.4, 0.6, 0.8, and 1.0 wt.% were investigated.
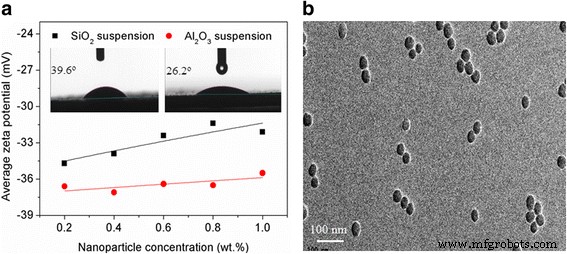
a Zeta potentials of the Al₂O₃ and SiO₂ suspensions. b TEM image of SiO₂ suspension.
Zeta potentials exceeded ±30 mV across all concentrations, confirming electrostatic stability. TEM images (Fig. 1b) confirmed uniform dispersion, and the suspensions remained stable for 4 days without sedimentation.
The solidification apparatus (Fig. 2a) consisted of a thermostatic bath (CDC‑1), an ultrasound generator (Sonics Vibra‑Cell JY88‑IIN), a temperature data logger (Agilent 34970A), and a PC for real‑time monitoring. Samples (~20 mL) were placed 2 in. from a vertically positioned ultrasound source. Cooling to −20 °C was performed at ultrasonic intensities ranging from 0.14 to 1.27 W cm⁻² (80 % duty cycle). Ultrasound was applied once the sample reached 0 °C and ceased upon ice nucleation; the total exposure was <2 min, negligible for heat transfer.
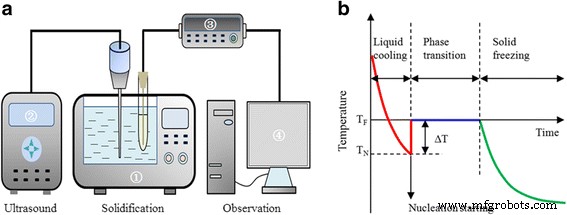
a Schematic of the experimental setup. b Typical temperature profile: T_F (freezing point), T_N (nucleation temperature), ΔT (supercooling degree).
Cooling curves were recorded using a copper–constantan T‑type thermocouple (±0.2 °C). Each condition was repeated ≥15 times; deviations were ±1.5 %.
Cavitation bubble states were assessed via a capillary method. UV–vis absorbance before and after solidification/melting quantified nanoparticle dispersion (five replicates). All data were averaged.
Results and Discussion
Supercooling with Nanoparticles or Ultrasound Alone
Figure 3 shows the ratio R₁ = ΔT_N/ΔT_W for various nanoparticle concentrations. Pure water exhibited ΔT_W ≈ 11.6 °C. R₁ < 1 and decreased with concentration, confirming that Al₂O₃ and SiO₂ act as effective nucleators. Al₂O₃ achieved a 37.4 % reduction at 0.6 wt.%, while SiO₂ reached 28.3 % at the same concentration; increasing SiO₂ to 0.8 wt.% matched Al₂O₃’s performance (37.1 %). Thus, higher concentration compensates for a larger contact angle.
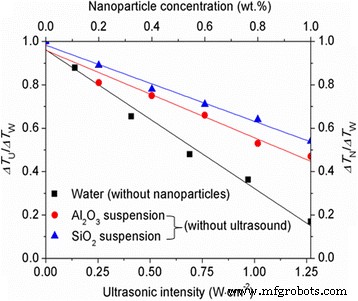
Effect of ultrasound and nanoparticles on supercooling. ΔT_U/ΔT_W (red) and ΔT_N/ΔT_W (blue) indicate reductions achieved by ultrasound and nanoparticles, respectively.
For ultrasound alone, the ratio R₂ = ΔT_U/ΔT_W dropped below 1, with the greatest reduction of 83.1 % at 1.27 W cm⁻², illustrating the powerful nucleating role of acoustic cavitation.
Combined Effect of Nanoparticles and Ultrasound
Figure 4 presents R₃ = ΔT_N‑U/ΔT_W, showing that the joint treatment always lowered supercooling relative to the control. The extent of reduction depends on nanoparticle concentration, ultrasonic intensity, and contact angle. For example, 63.7 % reduction was observed with 0.2 wt.% Al₂O₃ at 1.27 W cm⁻²; at the same intensity, 58.1 % reduction occurred when Al₂O₃ increased from 0.2 to 1.0 wt.%. Al₂O₃ consistently outperformed SiO₂ at equal conditions due to its smaller contact angle.
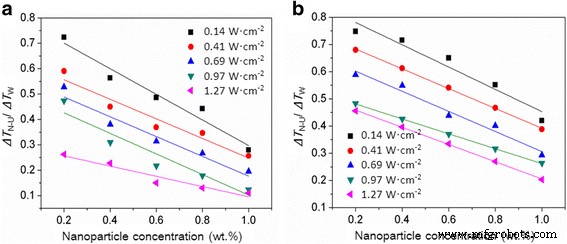
Combined impact of ultrasound and nanoparticles on supercooling. Red (Al₂O₃) and blue (SiO₂) curves depict ΔT_N‑U/ΔT_W.
Comparison of Separate and Combined Treatments
The combined reduction never exceeds the sum of the individual effects; in some cases, it is lower. For instance, at 0.6 wt.% Al₂O₃ and 0.69 W cm⁻², the joint reduction (70.6 %) is less than the combined individual reductions (37.4 % + 52.1 %). Conversely, the joint effect always surpasses the nanoparticle‑only effect, and can be greater or lesser than the ultrasound‑only effect depending on the parameters.
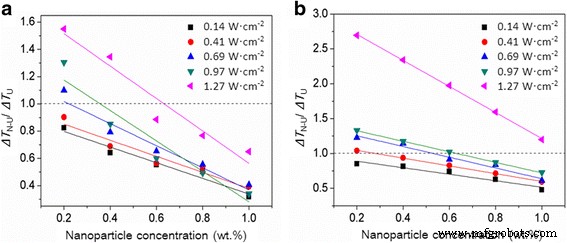
Ratio R₄ = ΔT_N‑U/ΔT_U across concentrations and intensities (Al₂O₃: green, SiO₂: purple). Values <1 indicate a positive combined effect.
Figure 6 maps the critical boundary where R₄ = 1. Above the red line (negative zone), R₄ > 1; below it (positive zone), R₄ < 1. The boundary shifts toward higher concentrations and lower intensities as the contact angle increases, shrinking the positive zone for SiO₂ relative to Al₂O₃.
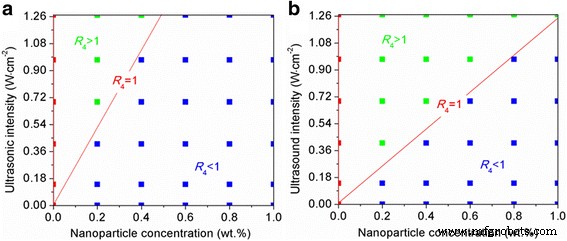
Phase diagram of combined versus single‑mode effects (Al₂O₃: blue, SiO₂: red). Points indicate whether the combined treatment outperforms ultrasound alone.
Nucleation Mechanism
Figure 7 shows volume changes and absorbance ratios (R₅ = A_A/A_B) for Al₂O₃ suspensions in positive and negative zones. Positive zones exhibit negligible volume change and stable absorbance, indicating retained dispersion. Negative zones show significant volume increase and absorbance loss, confirming aggregation.
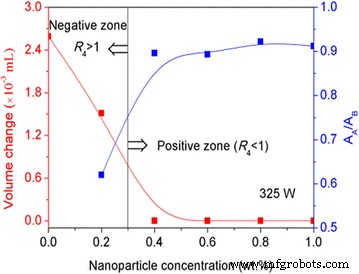
Al₂O₃ suspension behavior under ultrasound at varying concentrations.
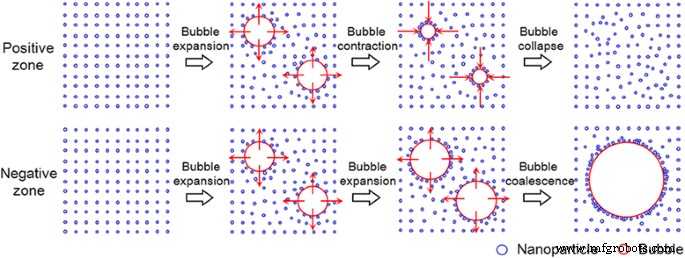
Schematic of cavitation bubble formation in positive (rectified diffusion) and negative (bubble coalescence) zones.
In the rectified‑diffusion zone, small bubbles grow via asymmetric mass transport and collapse, providing numerous nucleation sites while leaving nanoparticles dispersed. In the bubble‑coalescence zone, larger bubbles form through coalescence, adsorbing nanoparticles and reducing effective nucleation sites. The transition between these regimes is governed by ultrasonic intensity and nanoparticle concentration: higher intensity favors coalescence, while higher concentration and lower contact angle promote rectified diffusion.
Conclusions
Al₂O₃ and SiO₂ nanoparticles combined with ultrasound act as heterogeneous nucleation sites, lowering water supercooling. The effect depends on the type of cavitation bubbles formed:
- Rectified‑diffusion‑driven solidification: joint treatment yields a lower supercooling degree than either treatment alone because nucleation sites are additive.
- Bubble‑coalescence‑driven solidification: joint treatment requires a higher supercooling degree due to aggregation of nanoparticles on large bubbles.
Optimal performance is achieved when ultrasonic intensity is low and nanoparticle concentration is high, placing the system in the rectified‑diffusion zone. Contact angle critically influences the critical intensity and concentration: smaller angles expand the beneficial zone, while larger angles shrink it. These insights guide the design of nanoparticle‑ultrasound systems for efficient latent‑heat storage.
Abbreviations
- TEM:
Transmission electron microscope
Nanomaterials
- Raspberry Pi–Controlled Aquaponics System: Build Guide & Code
- Nanoparticle-Based Cancer Therapy: Advances, Mechanisms, and Clinical Translation
- Cobalt‑Doped FeMn₂O₄ Spinel Nanoparticles: Size‑Controlled Synthesis and Magnetic Behavior
- Water‑Soluble α‑NaGdF₄/β‑NaYF₄:Yb,Er Core–Shell Nanoparticles: Controlled Synthesis and Superior Upconversion Luminescence
- Comprehensive Review of Luminescent Silica Nanoparticles: Classification, Synthesis, and Practical Applications
- Bibliometric Trends in Drug Delivery and Magnetic Nanoparticles (1980‑2017)
- Enhanced Photoelectrochemical Water Splitting with TiO₂ Nanosheet Arrays, Layered SnS₂, and CoOx Nanoparticles
- Ultrasound-Enhanced Multifunctional Nanoparticles: A Novel Platform for Imaging and Synergistic Therapy of Metastatic Breast Cancer
- Mastering 2D Semiconductor Nanomaterials: Precision Synthesis & Breakthrough Applications
- Advanced Wastewater & Water Treatment Solutions: Sustainable Technologies for Clean Water



