All‑Carbon 3‑D Nanofiltration Membrane Achieves 99 % Antibiotic Removal and Rapid Water Flux
Industrial expansion and rising energy demands have amplified environmental pollutants, posing a global threat. We introduce a novel, all‑carbon nanofiltration (NF) membrane composed of multi‑walled carbon nanotubes (MWCNTs) interleaved within graphene oxide (GO) nanosheets, creating a 3‑D architecture with abundant two‑dimensional nanochannels. This ultrathin membrane (4.26 µm) demonstrates an exceptional tetracycline hydrochloride (TCH) adsorption of 99.23 % while maintaining a high water permeation rate of 16.12 L m-2 h-1 bar-1. Additionally, the cationic dye methylene blue (MB) is removed by 83.88 %, highlighting the membrane’s versatility for water purification.
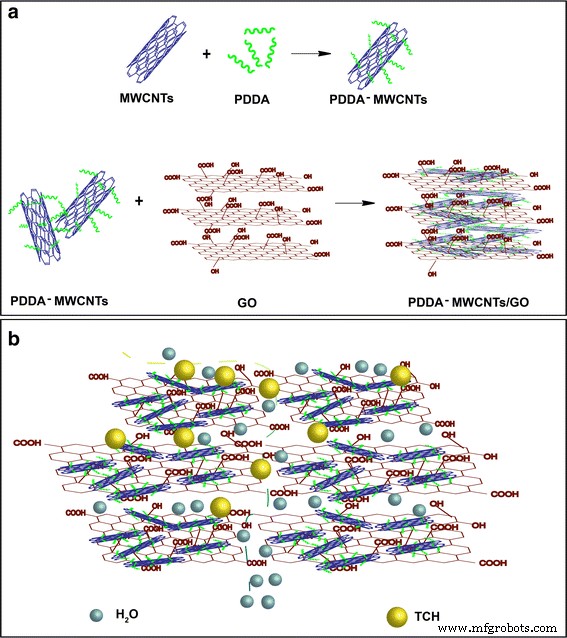
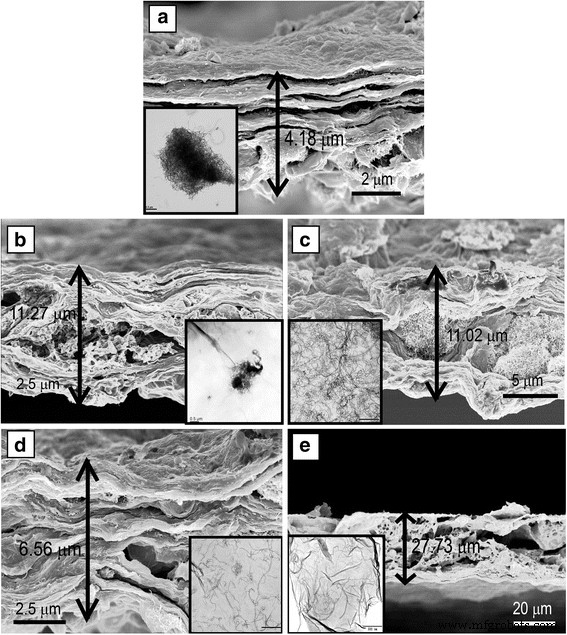
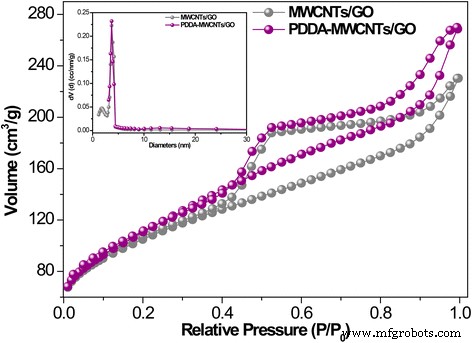
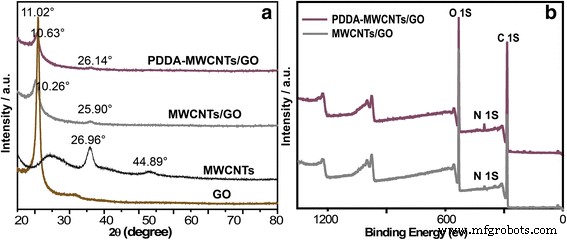
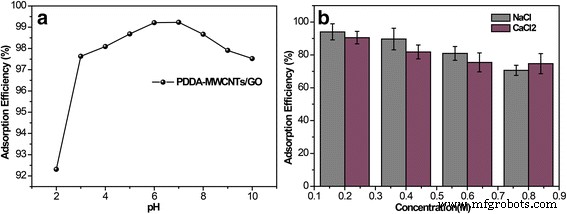
Pharmaceutical contaminants, particularly antibiotics, have become a pressing concern due to their ecological and public‑health impacts even at trace levels. Conventional removal techniques—oxidation (photocatalysis, sonolysis, Fenton) and adsorption—are often complex and energy‑intensive. Membrane separation offers a simpler alternative, yet most existing membranes rely solely on size‑exclusion and struggle with low‑molecular‑weight antibiotics. Carbon‑based materials, especially graphene and graphene oxide (GO), are attractive adsorbents owing to their atomically thin structure, high surface area, and mechanical strength. GO’s oxygenated functional groups confer hydrophilicity and anti‑fouling properties, while its tendency to restack can be mitigated by intercalating one‑dimensional fillers such as multi‑walled carbon nanotubes (MWCNTs). Compared with single‑walled variants, MWCNTs offer superior stability under flow and can effectively widen interlayer spacing, enhancing water flux. To address dispersion challenges, we graft the cationic polyelectrolyte poly diallyldimethylammonium chloride (PDDA) onto MWCNTs. This cationization promotes strong electrostatic attraction with anionic GO sheets, yielding a uniformly dispersed hybrid material that can be cast into a free‑standing, flexible NF membrane via vacuum filtration. The PDDA‑modified MWCNT/GO hybrid was assembled into an ultrathin, flexible membrane (Fig. 1a). The PDDA imparts positive charge, enabling robust electrostatic assembly with GO’s negative charges and preventing CNT aggregation. The resulting membrane exhibits abundant nanochannels that trap antibiotic molecules through steric hindrance and covalent interactions with surface functional groups. Optical imaging (Fig. 2a) confirms the membrane’s cloth‑like appearance and exceptional mechanical flexibility (Fig. 2b). Contact‑angle measurements demonstrate hydrophilicity, while long‑term immersion tests (Fig. 2c) show structural stability. The membrane remains crack‑free after more than seven filtration cycles (Fig. 2d). SEM (Fig. 3a) and TEM reveal a porous, uniformly dispersed structure. Adjusting PDDA concentration (0–20 wt%) influences CNT dispersion and membrane porosity. An optimal 10 wt% PDDA yields the thinnest, most uniform membrane, as evidenced by SEM images (Fig. 3b–e) and nitrogen adsorption–desorption isotherms (Fig. 4). The PDDA‑MWCNT/GO membrane presents a specific surface area of 402.96 m2 g-1, slightly higher than the 378.45 m2 g-1 of the unmodified hybrid, confirming enhanced mesoporosity (pore size 3–10 nm). X‑ray diffraction (Fig. 5a) shows an interlayer spacing increase from 0.81 nm to 0.87 nm upon PDDA functionalization, indicating successful intercalation and reduced restacking. XPS confirms increased nitrogen content, corroborating PDDA grafting. UV/Vis spectroscopy (Fig. 6a) demonstrates superior TCH removal: after a single filtration, residual concentrations drop to 6.74 µM (99.23 % removal) with the PDDA‑MWCNT/GO membrane, compared to 18.78 µM (95.04 %) with the unmodified hybrid. Water permeation reaches 16.12 L m-2 h-1 bar-1, nearly double that of the base membrane. Static adsorption tests yield an uptake of 436.13 mg g-1 for TCH, underscoring the membrane’s high capacity. Stability studies reveal minimal performance loss across a pH range of 2–10, with optimal adsorption at neutral pH. The membrane tolerates high ionic strength (NaCl/CaCl2 up to 0.9 M) with only modest reduction in TCH capture, indicating resilience to saline conditions. For the cationic dye MB, adsorption reaches 88.23 %, confirming that both size exclusion and electrostatic interactions contribute to removal. We have engineered a 3‑D, all‑carbon NF membrane that combines ultrathin GO nanosheets, PDDA‑functionalized MWCNTs, and electrostatic assembly to achieve high‑performance antibiotic removal. The membrane’s structural integrity, hydrophilicity, and anti‑fouling properties enable rapid water permeation and >99 % TCH adsorption in a single pass, while maintaining durability in harsh chemical environments. Its scalable, environmentally friendly synthesis positions it as a promising platform for advanced water treatment and chemical separation applications. GO (2 mg mL-1) was sourced from Nanjing XFNANO Materials Tech Co. Pure MWCNTs (diameter 20–30 nm, length 10–30 µm) were obtained from Beijing Boyu High‑Tech Novel Materials Technology Co. PDDA (MW 200,000–350,000, 20 wt% in H2O), tetracycline hydrochloride, NaCl, CaCl2, HCl, and anhydrous ethanol were purchased from Aladdin Chemical Co. Deionized water (18 M Ω cm-1) was produced by a Billerica water purification system. SEM (FESEM, Ultra 55, Carl Zeiss), TEM (Hitachi H‑800), UV/Vis (Lambda‑25, Perkin‑Elmer), BET (Autosorb‑iQ‑C, Quantachrome), XRD (Shimadzu XD‑3A), XPS (PHI 5000 Versa Probe), contact‑angle meter (Rame‑Hart‑100), zeta‑potential (SurPASS Electrokinetic Analyzer), and AFM (Bruker Multimode 8) were employed for characterization. MWCNTs (4.0 mg) were ultrasonicated in 1 mL deionized water, then PDDA (10 wt%) was added dropwise. The suspension was centrifuged, washed, and dried at 70 °C under vacuum for 24 h. 4.0 mg MWCNTs were dispersed in 24 mL of 0.5 mg mL-1 GO under stirring and sonication. The homogeneous mixture was vacuum‑filtered onto a 0.22 µm PVDF membrane, dried at 60 °C for 3 min, and peeled off after ethanol soaking. 20 mL of 500 µM TCH solution was vacuum‑filtered at 0.9 bar through the membrane. Filtrate concentration was measured by UV/Vis, and rejection rate calculated via R = (1 – Cp/Co) × 100 %. TCH solutions were prepared at pH 2–10 using HCl or NaOH, and at varying NaCl/CaCl2 concentrations (0.1–0.9 M). Filtration performance under these conditions was evaluated by UV/Vis spectroscopy.Abstract
Background
Results and Discussion


Conclusions
Methods/Experimental
Materials
Apparatus
Synthesis of PDDA‑Functionalized MWCNTs
Assembly of the MWCNT‑Interposed GO Membrane
Antibiotic Adsorption Experiments
Stability Experiments
Abbreviations
Nanomaterials
- Build a Headless Raspberry Pi 2 Water Alarm with Adafruit T‑Cobbler Plus
- Craig Trevor of Persuasion Inc. Discusses Growth, Investments, and Careers in the Trades
- Efficient Photocatalytic Removal of Cationic Dyes Using Calcined Zn‑Al Layered Double Hydroxides
- Highly Efficient Magnetic Carbon Microspheres for Reusable Sulfonamide Removal from Water
- Nanoporous Silica Tablets for Quantifying Biomolecules and Nanoparticles via Water Self‑Diffusivity in Bio‑Inspired Pores
- Co‑Doped Boron Nitride Nanosheets: Rapid, Eco‑Friendly Removal of Textile Water Pollutants
- Capture Water Droplets in Action Using Arduino Nano – DIY High-Speed Photography
- Water‑Powered Fire Effect: Arduino UNO + Neopixel Stick Project
- Streamlining Heavy Equipment Production: Motoman Robots for Precision Material Removal
- How to Eliminate Moisture in Compressed Air Systems



