Boosting Perovskite Solar Cell Efficiency Using Er3+-Yb3+-Li+ Tri‑Doped TiO₂ Up‑Conversion Layer
Abstract
We fabricated Er3+-Yb3+-Li+ tri‑doped TiO2 (UC‑TiO2) by introducing Li+ into Er3+-Yb3+ co‑doped TiO2. UC‑TiO2 displays markedly stronger up‑conversion emission than the co‑doped counterpart. When integrated into perovskite solar cells (PSCs), the power conversion efficiency (PCE) rose from 14.0% to 16.5%—a 19% improvement—demonstrating UC‑TiO2 as an effective light‑harvesting enhancer that extends absorption from visible to near‑infrared (NIR) wavelengths.
Background
Organolead halide perovskite solar cells have attracted significant attention due to their rapid efficiency gains, low cost, and straightforward fabrication. While PSCs now reach efficiencies above 22%, their narrow bandgap limits absorption mainly to the UV–visible spectrum, leaving a large portion of NIR light unutilized.
Up‑conversion materials that convert NIR photons into visible light can bridge this gap. Previous work has employed β‑NaYF4 doped with Er3+/Yb3+ ions; however, such systems can impede electron transport in the TiO2 layer. Recent studies have shown that tri‑doping TiO2 with Er3+, Yb3+, and Li+ enhances up‑conversion emission and improves dye‑sensitized solar cell performance. Moreover, Li‑doping has been reported to boost PSC efficiency relative to undoped TiO2. These findings motivated the present investigation of Er3+-Yb3+-Li+ tri‑doped TiO2 in PSCs.
In this study, we synthesized UC‑TiO2 by adding Li+ to Er3+-Yb3+ co‑doped TiO2, confirmed its enhanced up‑conversion emission, and incorporated it into mesoporous TiO2 layers for perovskite solar cells. The resulting devices exhibited a 19% increase in PCE.
Methods
Synthesis of Er3+-Yb3+-Li+ Tri‑Doped TiO2
TiO2 nanocrystals were prepared by a modified sol‑gel route. Titanium(IV) n‑butoxide (Ti(OBu)4) was first complexed with acetylacetone (AcAc) at room temperature for 1 h under agitation. The mixture was then hydrolyzed by adding isopropanol, deionized water, and concentrated nitric acid, yielding a light‑yellow TiO2 sol after 6 h of stirring. Er(NO3)3·5H2O, Yb(NO3)3·5H2O, and LiNO3 were subsequently introduced to achieve a molar ratio of Er:Yb:Li:Ti = 0.5:10:x:100 (x = 0, 10, 15, 20, 25). The solvent was removed by drying at 100 °C for 8 h, and the resulting powder was calcined at 500 °C for 30 min.
Fabrication of Perovskite Solar Cells
Fluorine‑doped tin oxide (FTO) glass was sequentially cleaned with acetone, isopropanol, and ethanol, followed by 15 min of UV‑O3 treatment. A compact TiO2 layer was spin‑coated from a 0.1 M titanium diisopropoxide bis(acetylacetonate) solution in 1‑butanol and annealed at 500 °C for 30 min. A mesoporous TiO2 film was then deposited at 4000 rpm for 30 s, followed by annealing at 100 °C (10 min) and 500 °C (30 min). The mesoporous layer was either pure commercial TiO2 (30NR‑D) or a mixture of UC‑TiO2 sol and diluted TiO2 paste (UC‑TiO2:TiO2 = x:100 v/v, x = 10–40).
A perovskite precursor comprising PbI2 (1.1 M), FAI (1 M), PbBr2 (0.22 M), and MABr (0.2 M) in a DMF/DMSO (4:1 v/v) mixture was spin‑coated in two steps (1000 rpm, 10 s; 4000 rpm, 30 s) with 200 µL chlorobenzene added during the second step. The film was then annealed at 100 °C for 1 h. A CsI stock solution (1.5 M in DMSO) was added to the perovskite precursor to improve thermal stability. The hole‑transport layer was formed by spin‑coating spiro‑MeOTAD (4000 rpm, 30 s), and a 80 nm gold electrode was evaporated on top.
Characterization
Up‑conversion photoluminescence was measured with a FLS 980 E fluorometer. Structural analysis employed XRD (DX‑2700) and XPS (THS‑103, Al Ka). UV‑vis‑NIR absorption was recorded on a Varian Cary 5000 spectrophotometer. Morphology was examined by SEM (JEM‑7001F). Photovoltaic performance was evaluated by J‑V curves under AM 1.5 G illumination using a Keithley 2440 sourcemeter. Electrochemical impedance spectroscopy (EIS) was performed with a CHI660e workstation.
Results and Discussion
Figure 1a shows that UC‑TiO2 with Li content x = 20 exhibits the strongest up‑conversion emission under 980‑nm excitation, with green (525/545 nm) and red (658 nm) bands attributable to Er3+ transitions. Increasing Li beyond this level reduces emission intensity.
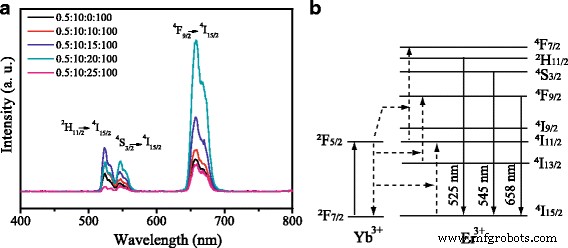
a Up‑conversion spectra of Er3+-Yb3+-Li+ tri‑doped TiO2 (x = 0–25). b Energy‑transfer scheme of Er3+.
XRD patterns confirm that UC‑TiO2 retains the anatase phase (JCPDS 21‑1272). XPS spectra (Figure 3) detect Ti 2p, Er 4d, Yb 4d, and Li 1s peaks, verifying successful doping.
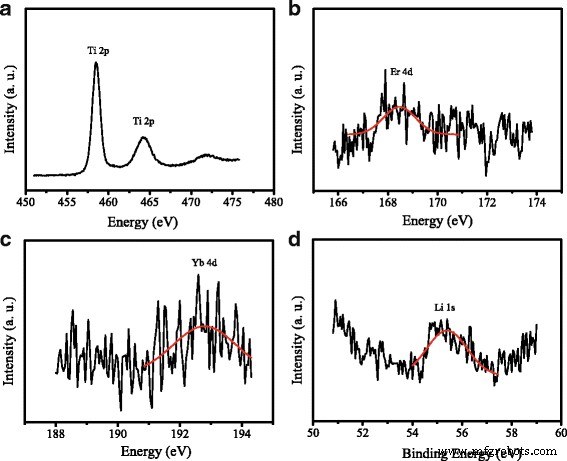
XPS spectra of UC‑TiO2: a Ti 2p, b Er 4d, c Yb 4d, d Li 1s.
UV‑vis‑NIR absorption (Figure 4a) reveals a pronounced tail extending to 1000 nm for UC‑TiO2, and Tauc analysis shows a reduced bandgap of 3.10 eV versus 3.20 eV for commercial TiO2. SEM images (Figure 5) indicate that the particle size (~30 nm) and morphology are unchanged by Li incorporation.

SEM images of the mesoporous layers: a without UC‑TiO2, b with UC‑TiO2.
Device performance (Figure 6a) shows that adding UC‑TiO2 at 20 % v/v maximizes PCE, rising from 14.0 % (control) to 16.5 % (UC‑TiO2)—a 19 % boost. Table 1 lists the key photovoltaic parameters. Enhanced short‑circuit current (Isc) and open‑circuit voltage (Voc) drive the efficiency gain.
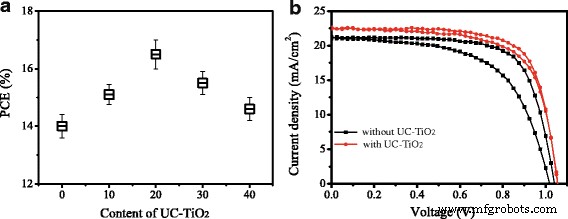
a PCE versus UC‑TiO2 content. b Representative J‑V curves.
Steady‑state PL (Figure 7a) shows reduced intensity for perovskite on UC‑TiO2, indicating more efficient electron extraction. Time‑resolved PL (Figure 7b) reveals a shorter fast decay component (2.8 ns) and a higher fraction (98.2 %) for UC‑TiO2 devices, confirming faster charge transfer.
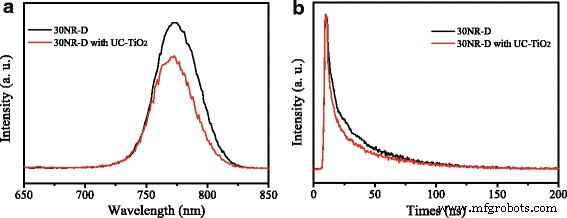
a PL and b TRPL of perovskite on 30NR‑D versus UC‑TiO2‑doped 30NR‑D.
EIS Nyquist plots (Figure 8a) show a reduced series resistance and increased recombination resistance for UC‑TiO2 devices, indicating improved charge transport and suppressed recombination.
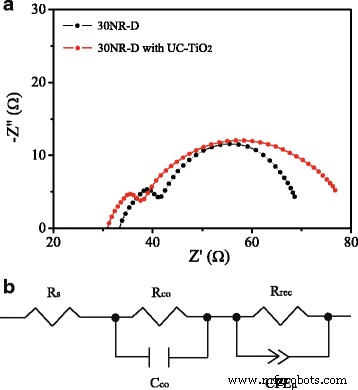
a Nyquist plots. b Equivalent circuit model.
Under 980‑nm filtered illumination, UC‑TiO2 devices produce higher photocurrent (Figure S3), confirming that the up‑conversion layer effectively converts NIR photons into usable visible light.
Band‑diagram analysis (Figure 9) shows that UC‑TiO2 has a lower conduction‑band edge than pure TiO2, creating a larger offset with the perovskite absorber. This barrier enhances Voc by reducing back‑tunneling of carriers.
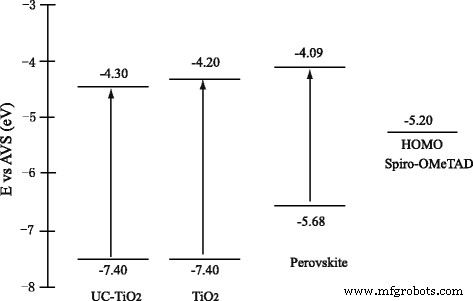
Schematic energy‑band alignment of UC‑TiO2, TiO2, perovskite, and Spiro‑OMeTAD.
In summary, incorporating UC‑TiO2 into the mesoporous scaffold expands the spectral response into the NIR, reduces recombination, and accelerates charge extraction, together yielding a 19 % PCE increase.
Conclusions
Tri‑doping TiO2 with Er3+, Yb3+, and Li+ creates UC‑TiO2 that emits strong up‑conversion light. When applied to PSCs, UC‑TiO2 improves Isc, Voc, and fill factor, raising PCE from 14.0 % to 16.5 % (19 % enhancement). The performance gains arise from extended NIR absorption, enhanced charge transport, and a favorable band‑offset.
Abbreviations
- EIS
Electrochemical impedance spectroscopy
- NIR
Near‑infrared
- PCE
Power conversion efficiency
- PL
Photoluminescence
- PSCs
Perovskite solar cells
- TRPL
Time‑resolved photoluminescence
Nanomaterials
- Optimizing Perovskite Solar Cell Efficiency with Size‑Controlled Ag Nanoparticles in a TiO₂ Compact Layer
- Enhanced Power Conversion in Flexible Fibrous Dye‑Sensitized Solar Cells via Multilayer TiO₂ Photoanodes and Composite Pt Counter Electrodes
- Enhanced Up‑Conversion TiO₂ Nanomaterial Boosts Perovskite Solar Cell Efficiency to 16.3 %
- Significant Efficiency Boost in CdS/CdSe Quantum Dot-Sensitized Solar Cells Using (001)-Oriented TiO2 Nanosheet Photoanodes
- Enhanced Performance of CsPbI₂Br Perovskite Solar Cells via ZnCl₂–MnCl₂ Doping
- High‑Efficiency 22%+ Perovskite Solar Cells Using Ultra‑Compact FA0.1MA0.9PbI3 Films
- In Situ-Formed, Low‑Temperature Deposited Nb‑Doped TiO₂ Compact‑Mesoporous Layer Enhances Hysteresis‑Free, High‑Efficiency Perovskite Solar Cells
- Gradient‑Engineered Light‑Absorbing Layer Boosts Carrier Separation and Efficiency in Perovskite Solar Cells
- Enhancing Quantum Dot Solar Cell Efficiency with Concentrating Photovoltaics and Dual Photoanodes
- Boosting Light Capture in Perovskite‑Silicon Tandem Solar Cells Using Nanohole Arrays



