Enhanced Power Conversion in Flexible Fibrous Dye‑Sensitized Solar Cells via Multilayer TiO₂ Photoanodes and Composite Pt Counter Electrodes
Abstract
We report a flexible fibrous dye‑sensitized solar cell (FFDSSC) that achieves a record 6.35 % photoelectric conversion efficiency. A multilayer TiO₂ photoanode, fabricated by a brush‑coating technique and successive TiCl₄ treatments, suppresses electron recombination and boosts charge capture. The counter electrode is a composite Pt layer deposited on an Al core, formed through a two‑step electrochemical–thermal decomposition route that creates a microporous surface and a low charge‑transfer resistance of 3.11 Ω cm². Comparative tests show that the composite Pt electrode matches the performance of a pure Pt wire counter electrode while using far less platinum, making the device inexpensive and scalable. The resulting FFDSSC combines high elasticity, flexibility, and stretchability, positioning it as a promising candidate for future wearable electronics.
Background
Dye‑sensitized solar cells (DSSCs) offer a compelling alternative to silicon photovoltaics, combining low cost, high power‑conversion efficiency, and environmental friendliness.1,2 However, conventional DSSCs rely on rigid glass substrates, limiting their integration into portable or textile‑based systems.3–5 Flexible, fiber‑shaped DSSCs address these constraints by providing lightweight, conformable power sources that can adhere to curved surfaces and withstand mechanical deformation.6–8
Previous work has demonstrated fiber DSSCs using modified titanium wires as photoanodes and pure platinum wires as counter electrodes.9,10 While platinum provides excellent electrocatalysis for the I⁻/I₃⁻ redox couple, its high cost hinders large‑scale adoption. Low‑cost alternatives—such as platinum films prepared by thermal decomposition or electrochemical reduction—have emerged, reducing platinum usage without sacrificing performance.11–14 The photoanode’s efficiency also suffers from limited dye loading and electron recombination. Multilayer TiO₂ structures, surface modifications, and particle‑size control have been employed to enhance light absorption and charge transport. The goal of this study is to integrate a multilayer TiO₂ photoanode with a composite Pt counter electrode to maximize the efficiency of FFDSSCs.
Methods
Materials
Analytical‑grade reagents were sourced from Shanghai Chemical Agent Ltd. (NiCl₂·6H₂O, thiourea, CoCl₂·6H₂O, ethanol, chloroplatinic acid, TiCl₄, tetra‑n‑butyl titanate). Aluminum and titanium wires (0.2 mm, 99.999 %) were obtained from Shengshida Metallic Material Co. N719 dye was purchased from Solaronix SA. TiO₂ paste (20 nm) was supplied by Wuhan Geao Co. Ltd.
Preparation of Flexible Fibrous TiO₂ Photoanode
Ti wires (15 cm) were polished, ultrasonically cleaned, and stored in isopropanol. TiCl₄ solutions (0.03 M and 0.05 M) were prepared and refrigerated. The photoanode was built in three stages:
- Barrier layer: immersion in 0.03 M TiCl₄ at 70 °C for 1 h, followed by 450 °C sintering for 30 min, repeated five times.
- Coating: brush application of 20 nm TiO₂ paste onto the barrier, sintered at 450 °C for 30 min, repeated three times to create a smooth surface.
- Pore filling: immersion in 0.05 M TiCl₄ at 70 °C for 1 h, sintered again at 450 °C for 30 min, repeated twice.
Finally, the anode was sensitized by dipping in a 0.3 mM N719 solution (tert‑butanol/acetonitrile) for 12 h, yielding the dye‑sensitized TiO₂ fiber.
Preparation of Pt Fibrous CE and FFDSSC Fabrication
The counter electrode was fabricated via a two‑step process:
- Electrodeposition: aluminum wire was immersed in 0.01 M H₂PtCl₆/LiClO₄ ethanol solution, forming a Pt‑1 CE.
- Thermal decomposition: Pt‑1 CE was heated to 250 °C, then 0.5 mL of a 1.0 wt % H₂PtCl₆/isopropanol:n‑butyl alcohol (1:1) solution with 1.0 wt % OP emulsifier was sprayed onto the surface. The composite Pt‑2 CE was sintered at 450 °C for 30 min, producing a microporous platinum film.
The Pt‑2 CE was twisted around the TiO₂ photoanode with a 0.5 mm pitch to form the FFDSSC. The assembly was sealed in a 0.5 mm plastic tube, filled with a redox electrolyte (0.05 M I₂, 0.1 M LiI, 0.6 M TBAB, 0.5 M TBP in acetonitrile), and UV‑cured with HT8803 adhesive to prevent leakage. Comparative devices using Pt‑1 or pure Pt CEs and unmodified TiO₂ photoanodes were also fabricated for benchmarking.
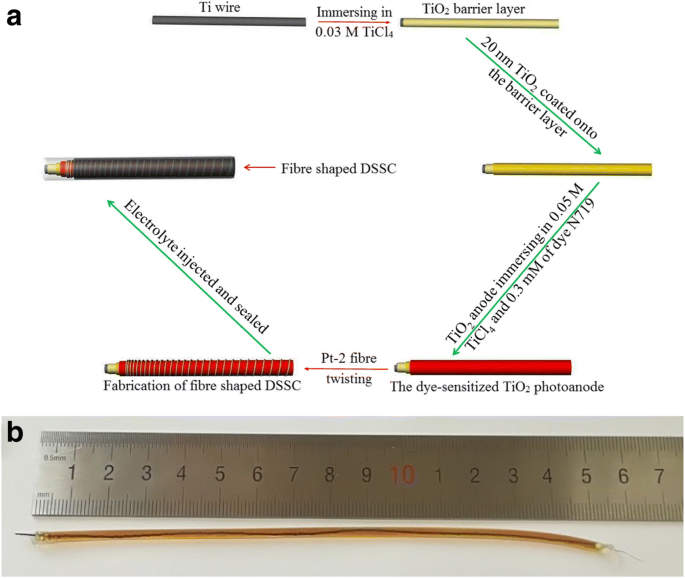
Schematic illustration of the fiber‑shaped DSSC fabrication. a Fabrication process of the fiber‑shaped DSSC. b Photograph of the fiber‑shaped DSSC.
Characterization
Surface morphologies were examined by JSM‑7001F field‑emission SEM, and elemental composition was analyzed via EDS (Bruker‑ASX Quan‑Tax 200). Cyclic voltammetry (CV) employed a three‑electrode cell (working: Pt wire, counter: 1.5 cm² Pt sheet, reference: Ag/AgCl) in an acetonitrile solution containing 10 mM LiI, 1 mM I₂, and 0.1 M LiClO₄. Electrochemical impedance spectroscopy (EIS) was performed at 20 °C with a 20 mV AC amplitude over 0.1–10⁵ Hz under dark, 0 V bias. Photovoltaic performance was assessed under 100 mW cm⁻² (AM 1.5 G) using a solar simulator (CEL‑S500). Efficiency (η) and fill factor (FF) were calculated from J–V curves:
η (%) = (V_max × J_max / P_in) × 100 = (V_oc × J_sc × FF / P_in) × 100
FF = (V_max × J_max / V_oc × J_sc)
Results and Discussion
Surface Morphology and Composition
Figure 2a,b show the TiO₂ photoanode’s smooth, porous surface, confirming uniform nanoparticle distribution. The dual TiCl₄ barrier layers effectively block electron recombination between the electrolyte and Ti wire. Figures 2c,d reveal the composite Pt CE’s uniformly distributed micropores—formed by rapid evaporation of the isopropanol/n‑butyl alcohol mixture—greatly increasing the platinum surface area and facilitating electrolyte adsorption. EDS maps (Figures 2e,f) confirm successful dye loading via the prominent Ru signal. The strong Al and Pt peaks in Figure 2g confirm the composite CE’s construction on an Al core.
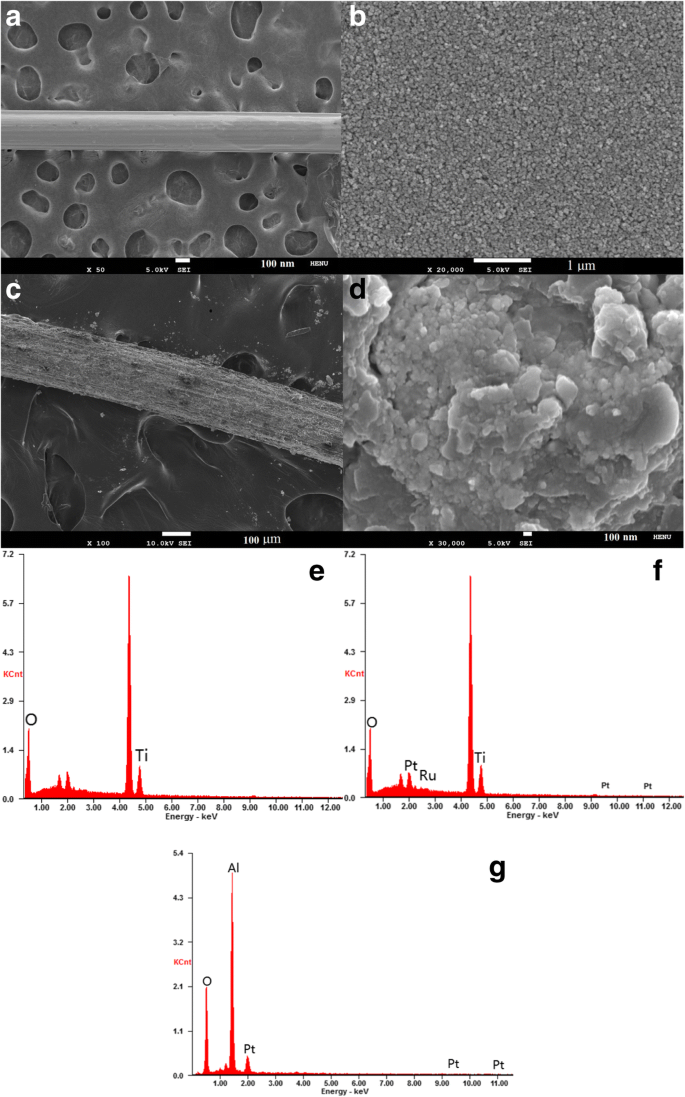
The SEM images of the TiO₂ photoanode (a,b) and fibrous Pt CE (c,d) with different resolutions, the EDS images of the TiO₂ photoanode before (e) and after (f) sensitizing, and the fibrous Pt CE (g).
Electrochemical Properties
CV curves (Figure 3) show that the Pt‑2 CE exhibits a cathodic peak current density of 2.87 mA cm⁻²—higher than Pt‑1’s 2.10 mA cm⁻²—indicating superior catalytic activity due to the microporous surface. Both electrodes share similar peak‑to‑peak separations, but Pt‑2’s larger peak current demonstrates lower overpotential for the I⁻/I₃⁻ reaction. Table 1 (not shown) lists the R_ct values, with Pt‑2’s 3.11 Ω cm² outperforming Pt‑1 (3.99 Ω cm²) and matching the pure Pt wire (3.10 Ω cm²).
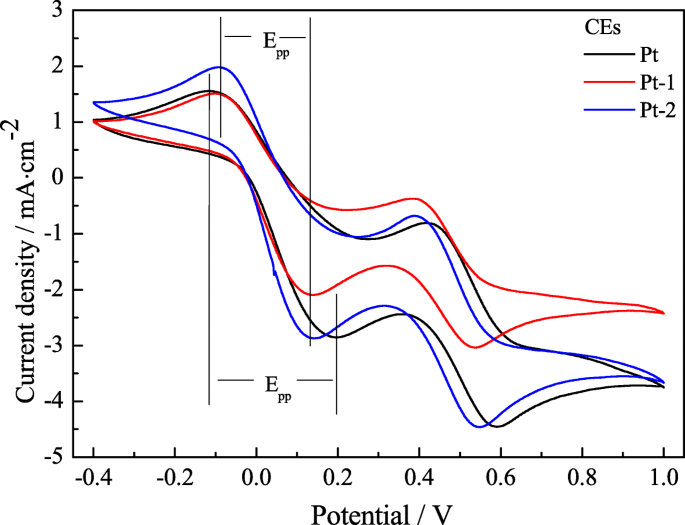
The cyclic voltammograms for the Pt‑1, Pt‑2, and pure Pt CEs at a scan rate of 50 mV s⁻¹.
Stability tests (Figure 4) confirm that Pt‑2’s peak currents remain unchanged over 50 cycles, evidencing excellent chemical durability.
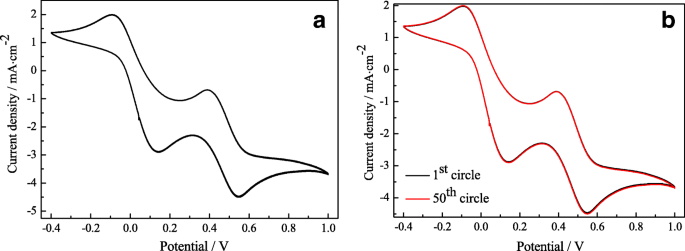
Cyclic voltammograms for the Pt‑2 CE at 50 mV s⁻¹. 50 continuous cycles (a); first and 50th cycles (b).
EIS results (Figure 5) further support the superior performance: Pt‑2’s series resistance (R_s = 3.57 Ω cm²) and charge‑transfer resistance (R_ct = 3.11 Ω cm²) are lower than those of Pt‑1 and comparable to the pure Pt CE. The reduced R_ct translates to faster electron transfer at the CE/electrolyte interface.
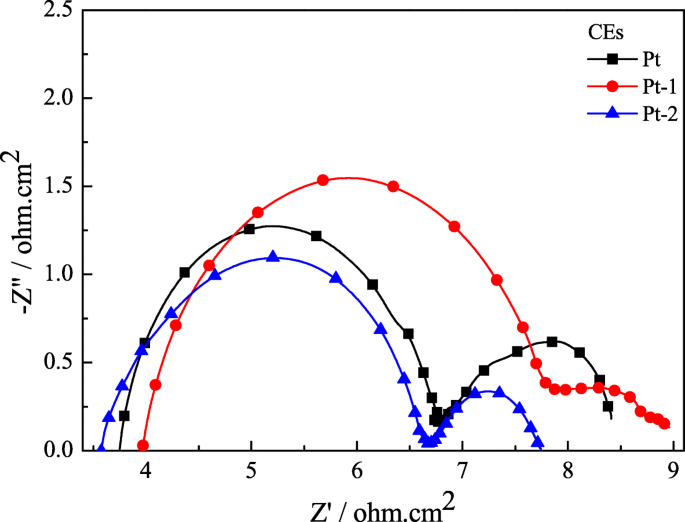
EIS of the Pt‑1, Pt‑2, and pure Pt CEs for the I⁻/I₃⁻ redox couple.
Tafel analysis (Figure 6) corroborates these findings: Pt‑2 exhibits an exchange current density (J₀) of 1.48 mA cm⁻² and a limiting diffusion current (J_lim) of 2.18 mA cm⁻², surpassing Pt‑1 (J₀ = 1.28 mA cm⁻², J_lim = 1.89 mA cm⁻²). The inverse relationship between J₀ and R_ct (J₀ = RT / (nFR_ct)) confirms Pt‑2’s lower interfacial resistance and higher catalytic efficiency.
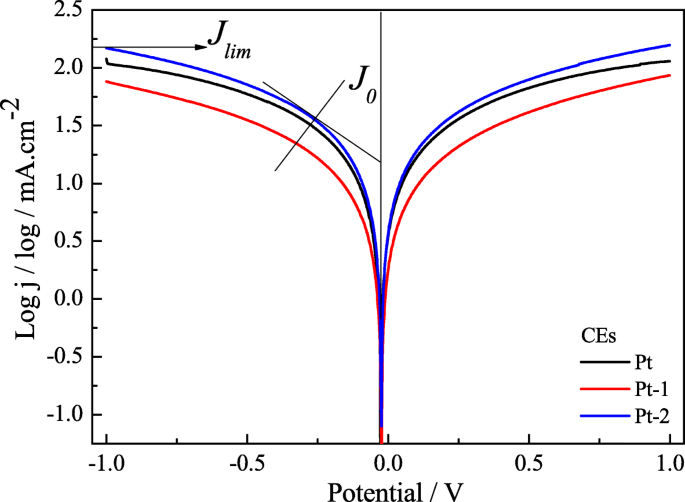
Tafel curves of the Pt‑1, Pt‑2, and pure Pt CEs for the I⁻/I₃⁻ redox couple.
Photovoltaic Performance of the FFDSSCs
J–V measurements (Figure 7 and Table 2) demonstrate that the FFDSSC with Pt‑2 CE and TiCl₄‑modified TiO₂ photoanode (device e) achieves V_oc = 0.860 V, J_sc = 12.45 mA cm⁻², FF = 0.70, and η = 6.35 %. This represents a 69.8 % improvement over the monolayer TiO₂/Ft‑1 device (η = 3.72 %) and matches the performance of the pure Pt CE device (η = 6.32 %). The enhanced efficiency is attributed to the reduced R_ct of Pt‑2, increased electrolyte contact area, and the TiCl₄ barrier layers that suppress recombination and boost electron injection.
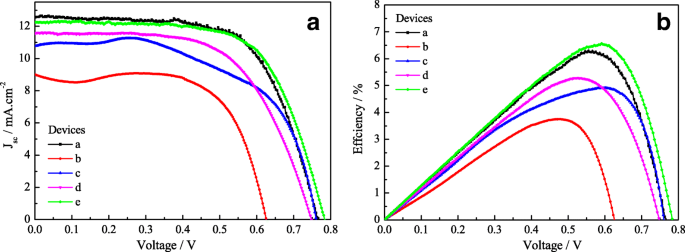
Photovoltaic performance curves for the FFDSSCs. a) J–V characteristics; b) Relationship between power‑conversion efficiency and V_oc.
EIS under illumination (Figure 8) shows that devices with TiCl₄‑modified photoanodes exhibit lower R_s and R_ct values, reflecting improved electron transport and reduced recombination. The FFDSSC‑e device displays the smallest resistances, corroborating its superior efficiency.
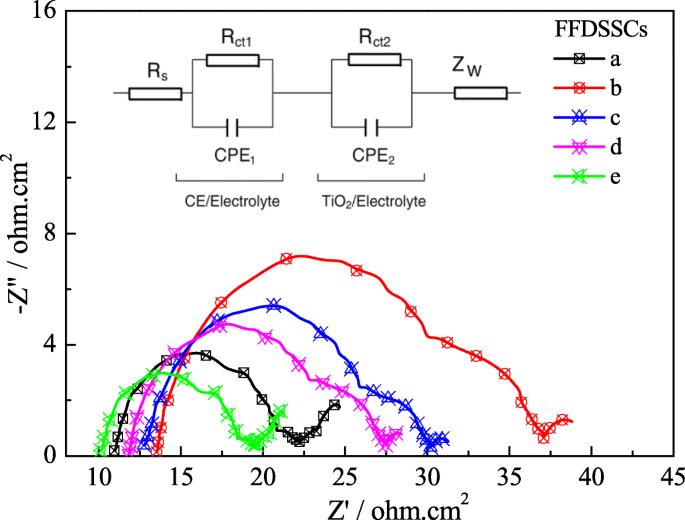
EIS for the FFDSSCs fabricated with various photoanodes and CEs under standard illumination.
External quantum efficiency (IPCE) spectra (Figure 9) peak around 520 nm—coincident with N719’s absorption maximum—and follow the order e > a > d > c > b. The higher IPCE for device e confirms the enhanced light harvesting and charge collection enabled by the multilayer TiO₂ and composite Pt CE.
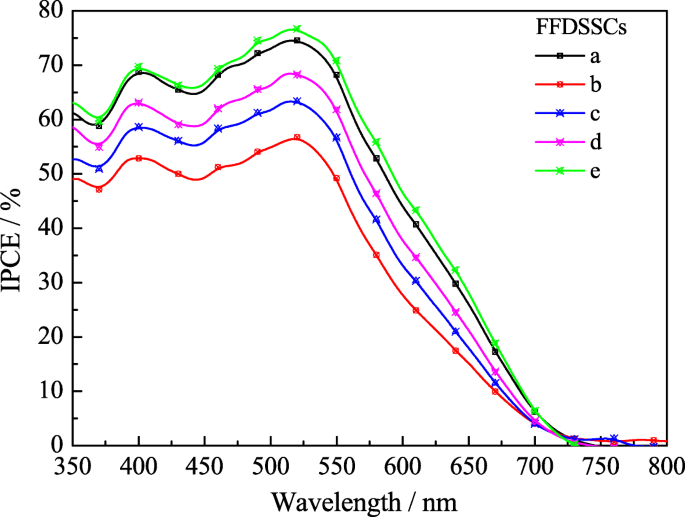
The IPCE of the various FFDSSCs.
Conclusions
We have demonstrated a high‑performance, low‑cost FFDSSC that combines a multilayer TiO₂ photoanode—modified with TiCl₄—and a composite Pt counter electrode with a double‑layer, microporous structure. The Pt‑2 CE delivers electrocatalytic activity comparable to a pure Pt wire while requiring significantly less platinum. The resulting device achieves a photo‑electric conversion efficiency of 6.35 %, a 69.8 % improvement over monolayer TiO₂/Ft‑1 devices, and is fully compatible with flexible, wearable applications due to its excellent elasticity and stretchability. This platform offers a scalable route toward high‑performance, wearable micro‑solar cells for future energy‑harvesting systems.
Abbreviations
- CE:
- Counter electrode
- CV:
- Cyclic voltammetry
- FFDSSC:
- Flexible fibrous dye‑sensitized solar cell
- I⁻/I₃⁻:
- Iodide/triiodide
- J₀:
- Exchange current density
- J_lim:
- Limiting current density
- J_max:
- Maximum current density
- J_sc:
- Short‑circuit current density
- J‑V:
- Photocurrent‑photovoltage
- PCE:
- Power conversion efficiency
- P_in:
- Incident light power
- R_ct:
- Charge transfer resistance
- R_s:
- Series resistance
- SEM:
- Scanning electron microscopy
- V_max:
- Maximum voltage
- V_oc:
- Open‑circuit voltage
Nanomaterials
- Nano‑Tree ZnO Nanowires Boost Dye‑Sensitized Solar Cell Efficiency
- High‑Efficiency Graphene Solar Cells: 9% Power Conversion with TFSA Doping
- Nano‑Heterojunctions: Boosting Solar Cell Efficiency with Colloidal Quantum Dots
- Optimizing Titanium Precursors for High‑Performance Compact TiO₂ Layers in Perovskite Solar Cells
- Boosting Perovskite Solar Cell Efficiency Using Er3+-Yb3+-Li+ Tri‑Doped TiO₂ Up‑Conversion Layer
- Optimizing Perovskite Solar Cell Efficiency with Size‑Controlled Ag Nanoparticles in a TiO₂ Compact Layer
- Dye‑Sensitized Solar Cells: Fundamentals, Advances, and Commercial Outlook
- Enhancing Solid‑State CuInS₂ Quantum‑Dot Solar Cells: Optimizing Charge Recombination via Controlled SILAR Deposition and Annealing
- High-Performance MoIn₂S₄@CNT Counter Electrodes for Dye‑Sensitized Solar Cells
- Climate Change Threatens Solar Panel Efficiency: MIT Study Reveals Impact



