Hierarchical Au@CdS‑CdS Nanoflowers: Stepwise Fabrication and Superior Photocatalytic Performance
Abstract
Hybrid nanostructures with intricate morphologies and high photocatalytic activity are notoriously difficult to fabricate, often requiring specialised skills and costly processes. Here we report a stepwise synthesis of flower‑shaped Au@CdS‑CdS nanoflowers that combine a gold core, a cadmium sulfide (CdS) shell, and epitaxially grown CdS nanorods. The resulting nanoflowers absorb across the entire visible spectrum (up to 850 nm) and exhibit markedly enhanced photo‑induced charge transfer, as confirmed by photoluminescence studies. Compared with both CdS alone and Au@CdS core‑shell nanoparticles, the Au@CdS‑CdS nanoflowers show the highest photocatalytic degradation rates under irradiation of 400–780 nm and 600–780 nm. We also propose a plausible growth mechanism that can guide the rational design of other metal‑semiconductor hybrids with complex architectures.
Introduction
Solar energy is the cleanest and most abundant renewable resource available today. Converting sunlight into chemical or electrical energy through semiconductor photocatalysis is widely regarded as a promising strategy for both energy production and environmental remediation [1, 2]. Over the past decade, researchers have engineered numerous semiconductor photocatalysts, with CdS emerging as a standout candidate because its 2.4 eV bandgap enables efficient visible‑light absorption [5]. Unfortunately, CdS suffers from rapid electron–hole recombination and photocorrosion, which limit its practical performance.
Hybrid nanostructures, especially metal‑semiconductor composites, have attracted significant attention because they can broaden light absorption, improve charge separation, and enhance catalytic activity [6–9]. Recent examples include ZnS‑CdS‑Cu₂‑xS ternary heteronanorods [10], CdS@TiO₂ core‑shell particles [11, 12], and CdS–Au–TiO₂ Z‑scheme junctions [13]. Despite these advances, the precise control of composition, crystal phase, and morphology in CdS‑based hybrids remains a formidable challenge.
Gold (Au) nanoparticles provide localized surface plasmon resonance (LSPR) that can concentrate and scatter light at the nanoscale [20, 21]. When coupled with CdS, the plasmon–exciton interaction can significantly boost photo‑excitation efficiency [22–29]. While various Au–CdS morphologies have been explored, three‑dimensional hierarchical structures with well‑defined architectures are rare, primarily because their synthesis demands meticulous control over nucleation and growth steps.
In this study, we present a facile stepwise approach to fabricate Au@CdS‑CdS nanoflowers. The nanoflowers consist of an Au core, a thin CdS shell, and dense arrays of CdS nanorods grown epitaxially on the shell. They exhibit an extended absorption band up to 850 nm and superior photocatalytic performance under visible‑light irradiation. We also elucidate a growth mechanism that can inform future designs of complex metal‑semiconductor nanohybrids for clean‑energy and environmental applications.
Methods
Synthesis of Au@CdS‑CdS Nanoflowers
All reagents were analytical grade (Aladdin) and used as received. Au colloids (~50 nm) were prepared by the Frens method [31]. Au@CdS core‑shell nanoparticles (CSNs) were synthesized via a hydrothermal route [32]. In a typical step, an aqueous l‑cysteine (Cys) solution (2:1 M ratio of Cys to Cd²⁺) was mixed with Au colloids, stirred for 15 min, diluted to 50 mL, and sealed in a 100 mL Teflon‑lined autoclave. The autoclave was heated to 130 °C for 6 h to grow the initial CdS shell. For a thicker shell, 10 mL of the Cys/Cd²⁺ mixture was added to 10 mL Au colloids before the hydrothermal step.
To grow the CdS nanorods, 10 mL of Au@CdS CSN colloid, 1 mL of 10 mM Cys/Cd²⁺, and 20 mL ethylenediamine (En, >99%) were combined, stirred for 15 min, and transferred to a 50 mL Teflon‑lined autoclave. The mixture was heated to 180 °C and maintained for 10 h, then cooled to room temperature. The product was washed repeatedly with deionised water and ethanol to remove unreacted ions, and finally dispersed in absolute ethanol.
For comparison, CdS nanoparticles were prepared by heating 4 mL Cys/Cd²⁺ with 20 mL water to 130 °C for 6 h under identical conditions.
Material Characterisations
X‑ray diffraction (XRD) was performed on a Bruker D8 diffractometer. UV‑Vis absorption spectra were recorded with a PerkinElmer Lambda 35 photometer. Field‑emission scanning electron microscopy (FE‑SEM) and energy‑dispersive X‑ray spectroscopy (EDS) were carried out on a JSM‑7001F. Transmission electron microscopy (TEM) and high‑resolution TEM (HR‑TEM) images were obtained on a JEM‑2010 operating at 200 kV. Photoluminescence (PL) spectra were measured on a Perkin‑Elmer LS‑55 (excitation 400 nm). Photocurrent responses were recorded in a three‑electrode cell (ITO working electrode, Pt counter, Ag/AgCl reference) under a 300 W Xe lamp without bias, using 0.2 M Na₂SO₄ as the electrolyte.
Photocatalytic Activity Measurement
Photocatalytic degradation of Rhodamine 6G (R6G) was performed under visible‑light irradiation using a 300 W Xe lamp coupled with two band‑pass filters (400–780 nm and 600–780 nm). In each experiment, 6 mg of catalyst was added to 20 mL of 1.0 × 10⁻⁵ M R6G, stirred for 30 min in the dark to reach adsorption equilibrium, and then illuminated. Aliquots (2.5 mL) were withdrawn every 10 min, centrifuged to remove the catalyst, and analysed by UV‑Vis spectroscopy. Degradation (%) was calculated as [(C₀–Cₜ)/C₀] × 100 %, where C₀ is the initial concentration and Cₜ is the concentration at time t.
Results and Discussion
Synthesis Mechanism of Nanoflowers
The stepwise synthesis is illustrated in Fig. 1. First, 50 nm Au nanospheres are formed. A hydrothermal CdS deposition then yields Au@CdS core‑shell nanoparticles (CSNs). Finally, a mixed solvothermal process grows CdS nanorods on the CSN surfaces, producing hierarchical Au@CdS‑CdS nanoflowers. l‑Cysteine, with SH, COOH, and NH₂ groups, acts as both a sulfur source and a ligand that mitigates lattice mismatch between Au and CdS [32]. Ethylenediamine guides the anisotropic growth of the CdS rods [33]. Direct co‑precipitation of Au, Cys/Cd²⁺, and En fails to produce the desired morphology, underscoring the necessity of the intermediate CSN step.
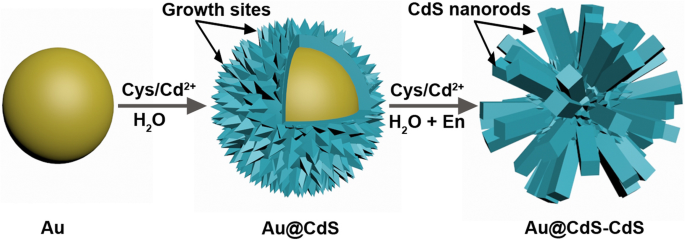
Schematic illustration of the synthesis processes of Au@CdS‑CdS nanoflowers
Sample Morphology and Structure
SEM images in Fig. 2a–c confirm the evolution of the particles. Au nanospheres appear uniform and well‑dispersed (Fig. 2a). The Au@CdS CSNs show a serrated CdS shell with pronounced bulges (Fig. 2b, inset). The final nanoflowers exhibit well‑defined, flower‑like shapes with dense CdS nanorods (Fig. 2c). TEM images (Fig. 2d–e) reveal that the nanorods have a diameter of ~16 nm and a length of ~40 nm, growing preferentially along the [001] direction. HR‑TEM (Fig. 2f) displays a lattice spacing of 0.34 nm, matching the (002) planes of wurtzite CdS. EDS analysis (Fig. 2g) confirms the presence of Au, Cd, and S. Photographs of the colloidal solutions (Fig. 2i) show distinctive colors: red wine for Au colloids, purple for thin‑shell Au@CdS, green for nanoflowers, yellow for CdS, and green for thick‑shell Au@CdS.

a, b, h SEM images of Au nanoparticles, Au@CdS with thin shell, and CdS counterparts, respectively. d TEM images of Au@CdS with thick shell. c, e, f, g SEM, TEM, HR‑TEM image, and EDS profile of Au@CdS‑CdS nanoflowers. i Photographs of their solution colors
X‑ray Diffraction Analysis
Fig. 3 displays the XRD patterns. The Au nanospheres show fcc peaks at 2θ = 38.2°, 44.2°, and 64.75° (111, 200, 220). CdS exhibits wurtzite peaks at 25°, 26.5°, 28.2°, 43.8°, 47.8°, and 51.9° (100, 002, 101, 110, 103, 112). Both Au and CdS peaks are present in the CSNs and nanoflowers, confirming the composite nature. The dominant (002) reflection in the nanoflowers indicates that the CdS rods grow along [001], consistent with HR‑TEM results.
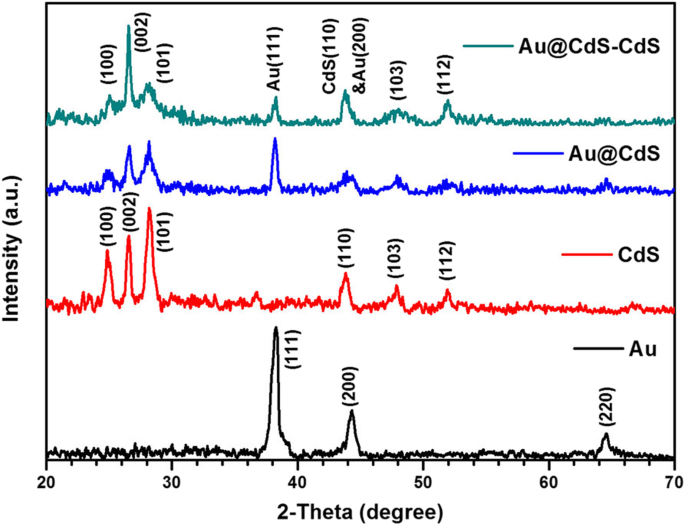
XRD patterns of Au, CdS, Au@CdS CSNs, and Au@CdS‑CdS nanoflowers
Optical Properties
UV‑Vis spectra (Fig. 4) show that pure CdS absorbs below 500 nm. Au nanospheres exhibit an LSPR peak at 530 nm, which red‑shifts to 568 nm after coating with a thin CdS shell, and further to 623 nm and 635 nm for thick‑shell CSNs and nanoflowers, respectively. The shift reflects the increasing dielectric environment and shell thickness. Importantly, the nanoflowers extend absorption up to 850 nm, effectively harvesting the full visible spectrum (400–760 nm) and thereby enhancing photocatalytic activity.
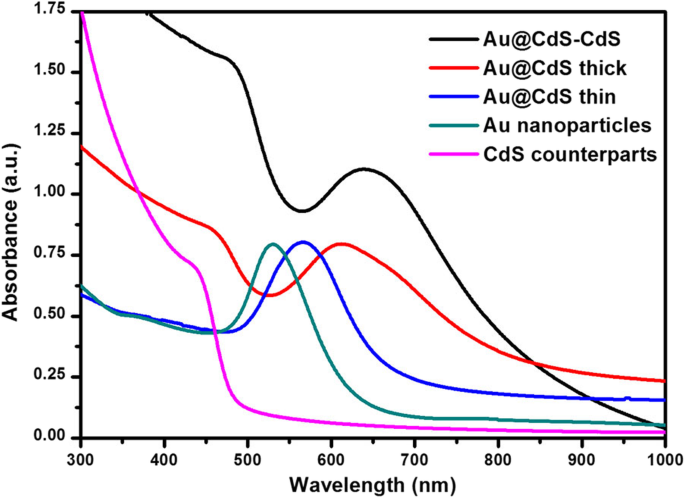
The UV‑Vis absorption spectra of CdS counterparts, Au nanoparticles, and Au@CdS CSNs and Au@CdS‑CdS nanoflowers, respectively
Charge‑Transfer Dynamics
PL spectra (Fig. 5a) reveal strong quenching of the CdS emission in the hybrid structures, indicating efficient electron transfer from CdS to Au (Fermi level of Au ≈ +0.5 V vs. NHE, below the CdS conduction band ≈ –1.0 V). The nanoflowers exhibit the greatest quenching, reflecting superior charge separation. Transient photocurrent measurements (Fig. 5b) further confirm that the nanoflowers generate the highest photocurrent among the tested samples, signifying prolonged carrier lifetimes conducive to photocatalysis.
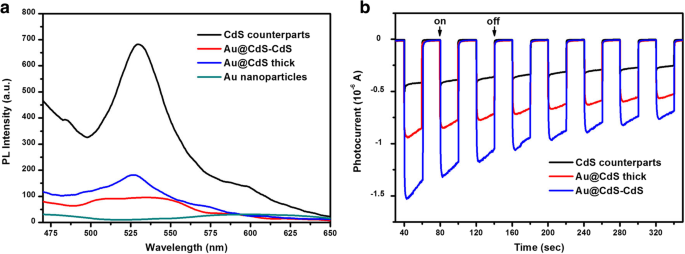
a The PL spectra of Au colloids, Au@CdS thick, Au@CdS‑CdS, and CdS counterparts, respectively. b Comparison of transient photocurrent response of the Au@CdS‑CdS nanoflowers, Au@CdS thick, and CdS counterparts irradiated with visible light (λ ≥ 400 nm) in 0.2 M Na₂SO₄ without bias versus Ag/AgCl
Photocatalytic Performance
Under 400–780 nm irradiation, the nanoflowers achieve 86% degradation of R6G in 40 min, outperforming Au@CdS thick (71%), CdS (46%), and Au (12%) (Fig. 6b). The superior activity is attributed to (i) enhanced light harvesting, (ii) efficient charge separation at the Au/CdS interface, and (iii) a larger specific surface area—more than 4.67 times that of the nanospheres (see Additional file 1).
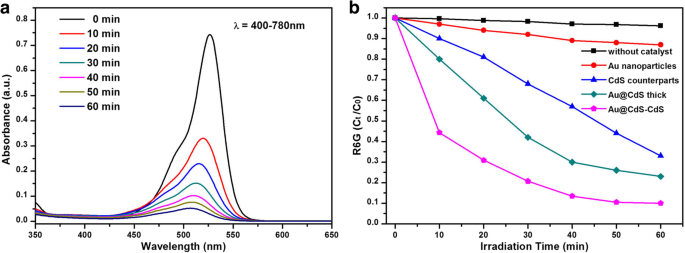
a Absorption spectra of R6G solutions at different irradiation times using Au@CdS‑CdS nanoflowers (400–780 nm). b Normalised concentration (Cₜ/C₀) versus time for the four catalysts
When irradiated with 600–780 nm light, the nanoflowers still outperform the others (45% vs. 34% for Au@CdS thick, 15% for CdS, 9.8% for Au) (Fig. 7b). This indicates that hot electrons generated in Au at wavelengths above 516 nm are efficiently injected into the CdS conduction band, driving the photocatalytic reaction. Repeated cycling (Fig. 8a) demonstrates remarkable stability, with negligible loss of activity over three cycles.
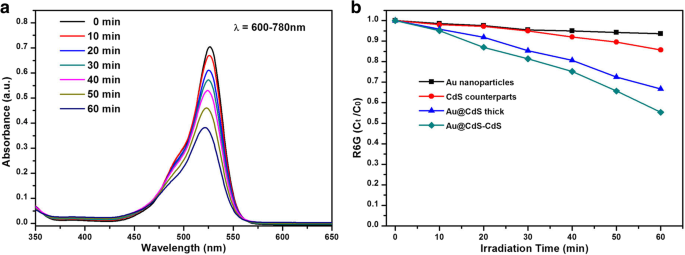
a Absorption spectra of R6G solutions at different irradiation times under 600–780 nm using Au@CdS‑CdS nanoflowers. b Normalised concentration (Cₜ/C₀) versus time for the four catalysts
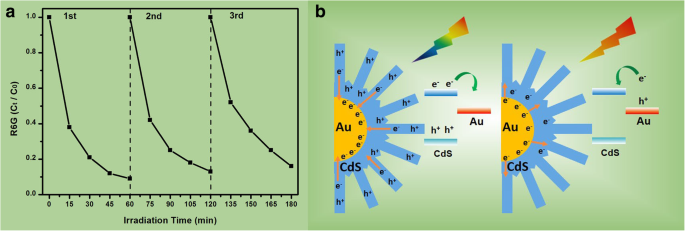
a Photocatalytic cycling for R6G degradation over Au@CdS‑CdS nanoflowers (400–780 nm). b Schematic of the two charge‑separation pathways under 400–780 nm and 600–780 nm excitation
Mechanistic Insights
The Au@CdS‑CdS nanoflowers offer two distinct photo‑induced charge‑transfer routes. When the CdS bandgap is excited (λ < 516 nm), electrons migrate from CdS to Au. When the Au LSPR is excited (λ > 516 nm), hot electrons transfer to the CdS conduction band. Simultaneous excitation enhances the probability of charge separation, reducing recombination and boosting photocatalytic reactions that generate ·OH and O₂⁻ radicals.
Conclusion
We have demonstrated a simple, stepwise route to produce hierarchical Au@CdS‑CdS nanoflowers with epitaxial CdS nanorods on a gold core. These nanoflowers display a broad visible‑light absorption up to 850 nm and exhibit the highest photocatalytic degradation rates among the studied systems for Rhodamine 6G under both 400–780 nm and 600–780 nm irradiation. The enhanced performance stems from improved charge separation, increased surface area, and an extended absorption range. The proposed growth mechanism provides a blueprint for designing other metal‑semiconductor hybrids with complex architectures for clean‑energy and environmental remediation applications.
Abbreviations
- CSNs
Core‑shell nanoparticles
- Cys
l‑cysteine
- Cys/Cd2+
l‑cysteine‑Cd2+
- EDS
Energy‑dispersive spectrometer
- En
Ethylenediamine
- FE‑SEM
Field‑emission scanning electron microscope
- HR‑TEM
High‑resolution transmission electron microscope
- NPs
Nanoparticles
- PL
Photoluminescence
- TEM
Transmission electron microscope
- UV‑Vis
Ultraviolet‑visible
- XRD
X‑ray diffraction
Nanomaterials
- Cellulose–POSS–Silica/Gold Core–Shell Hybrid Nanocomposites: One‑Step Sol‑Gel Synthesis and Multifunctional Properties
- Enhanced Photocatalytic Performance of ZnO/In₂O₃ Hybrid Nanostructures via Hydrothermal Synthesis
- Water‑Soluble Antimony Sulfide Quantum Dots: Efficient Synthesis and Superior Photo‑Electric Performance
- Easily Synthesized Wormhole‑Like Mesoporous SnO₂ via Evaporation‑Induced Self‑Assembly: Superior Ethanol Gas‑Sensor Performance
- One‑Pot Synthesis of Cu₂ZnSnSe₄ Nanoplates and Their Efficient Visible‑Light Photocatalytic Degradation of Rhodamine B
- Efficient One‑Step Photo‑Ultrasonic Synthesis of rGO/Ag₃PO₄ Quantum‑Dot Composites for Enhanced Visible‑Light Photocatalysis
- PVP‑Enhanced SnO₂ Nanoflowers Deliver Ultra‑Fast, Highly Selective H₂S Sensing
- Hybrid TiO₂ Nanocomposite Coating Achieves 80% Diffuse Reflectance and Suppressed Specular Glare
- Enhanced Visible‑Light Photocatalysis and Self‑Cleaning via CuS‑Decorated TiO₂/PVDF Nanofibers
- Enhanced Hydrogen Evolution Catalysts: MoSe₂–Ni₃Se₄ Hybrid Nanostructures with Superior Electrocatalytic Activity



