Magnetic ZnO/Fe3O4/g‑C3N4 Nanocomposite: High‑Efficiency Visible‑Light Photocatalyst for Monoazo Dye Degradation
Abstract
We report a magnetically recoverable ternary nanocomposite (ZnO/Fe3O4/g‑C3N4) that demonstrates superior photocatalytic performance for the visible‑light degradation of monoazo dyes. Structural and optical properties were characterized by XRD, TEM, EDS, XPS, PL, UV‑vis DRS, and photo‑electrochemical measurements. The heterojunction between g‑C3N4 and ZnO expands the light‑absorption window and suppresses charge‑carrier recombination, leading to a pronounced photocatalytic activity that outperforms pure ZnO and g‑C3N4. The 50 wt % g‑C3N4 composite (ZnO/Fe3O4/g‑C3N4‑50%) retained 95.3 % of its activity after five recycling cycles and achieved degradation efficiencies of 97.87 %, 98.05 %, and 83.35 % for methyl orange (MO), alizarin yellow R (AYR), and orange G (OG), respectively. The catalyst’s magnetic recoverability, high stability, and rapid de‑coloration make it a promising candidate for industrial wastewater treatment.
Introduction
Industrial effluents contain high concentrations of organic dyes, which are carcinogenic and mutagenic, posing a significant environmental threat. Conventional treatment methods often fail to remove these recalcitrant compounds efficiently. Photocatalysis has emerged as a clean, energy‑efficient technology for dye degradation, especially when driven by visible light. ZnO is an inexpensive, environmentally benign semiconductor, yet its wide bandgap (3.37 eV) limits absorption to UV light and promotes rapid electron–hole recombination. Coupling ZnO with a narrow‑bandgap, metal‑free semiconductor such as g‑C3N4 (Eg = 2.70 eV) has been shown to enhance visible‑light absorption and charge separation. However, ZnO/g‑C3N4 composites often suffer from low catalytic activity and difficulty in recovery. Introducing Fe3O4 imparts magnetic properties, enabling easy separation and recyclability. This study synthesizes a ternary ZnO/Fe3O4/g‑C3N4 nanocomposite and investigates its structural, optical, and photocatalytic properties, with a focus on the degradation of monoazo dyes.
Materials and Methods
Materials
All reagents were analytical grade: zinc acetate, ethanol, urea, oxalic acid, and monoazo dyes (MO, AYR, OG). Detailed properties are listed in Table 1.
Preparation of Fe3O4
Fe3O4 nanoparticles were synthesized via a hydrothermal route using FeCl3·6H2O and FeSO4·7H2O (2:1 molar ratio) in aqueous ammonia. The brownish suspension was aged at 70 °C, washed, and dried at 40 °C under vacuum.
Preparation of ZnO and ZnO/Fe3O4
ZnO was obtained by dissolving zinc acetate in ethanol, adding oxalic acid, aging the sol, drying, and calcining at 400 °C. Fe3O4 was dispersed into the precursor solution to produce ZnO/Fe3O4.
Preparation of ZnO/Fe3O4/g‑C3N4‑x
ZnO/Fe3O4 (1 g) was ground with melamine (g‑C3N4 precursor) at a 1:1 mass ratio, dispersed in water, ultrasonicated, dried, and annealed at 550 °C to yield composites with 20 %, 50 %, and 70 % g‑C3N4 content (denoted ZnO/Fe3O4/g‑C3N4‑20%, ‑50%, ‑70%).
Characterization
XRD (Rigaku Giegerflex D/Max B), TEM (Tecnai G2F20), EDS, XPS (250XI ESCA), PL (FLsp920), UV‑vis DRS (Hitachi UV‑4100), VSM, and photo‑electrochemical tests (three‑electrode quartz cell) were performed to assess crystal structure, morphology, composition, surface states, optical absorption, magnetic properties, and charge‑carrier dynamics.
Photocatalytic Activity
Visible‑light (420 nm cutoff) degradation of 30 mg L‑1 dye solutions was monitored with a 500‑W Xe lamp. 10 mg of catalyst was added to 50 mL dye solution, pre‑adsorbed in the dark for 30 min, then irradiated. Dye concentrations were measured by UV‑Vis spectrophotometry at λmax of 466 nm (MO), 373 nm (AYR), and 475 nm (OG). Degradation efficiency (η) was calculated as η = (C0 – Ct)/C0 × 100 %.
Results and Discussion
XRD
ZnO exhibited characteristic wurtzite peaks at 2θ = 31.81°, 34.44°, etc. g‑C3N4 showed (002) at 27.3°. Composite patterns displayed all ZnO, Fe3O4, and g‑C3N4 peaks, confirming phase purity. Peak intensities varied with g‑C3N4 loading.
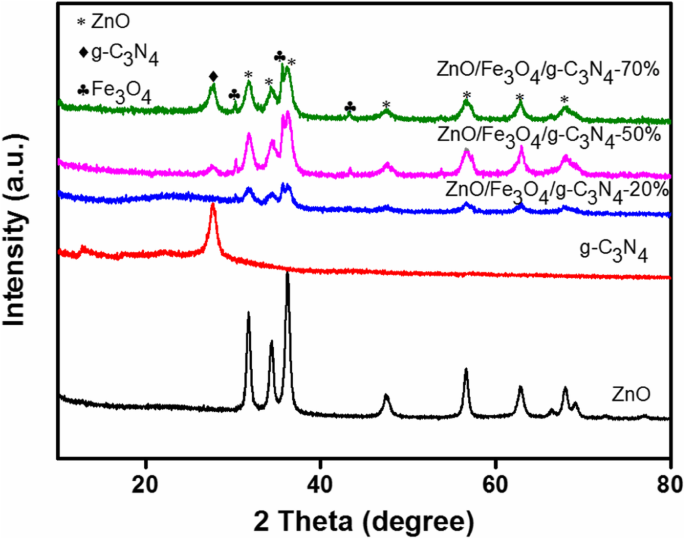
XRD patterns of ZnO, g‑C3N4, and composites (20 %, 50 %, 70 %)
TEM and EDS
ZnO displayed hexagonal particles; g‑C3N4 formed layered sheets. In ZnO/Fe3O4/g‑C3N4‑50%, ZnO/Fe3O4 nanoparticles were anchored on g‑C3N4 sheets. EDS confirmed Zn, Fe, C, N, and O signals.
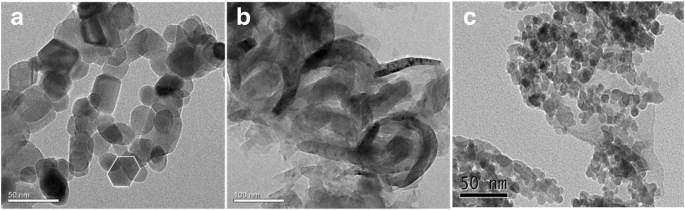
TEM images of ZnO (a), g‑C3N4 (b), and ZnO/Fe3O4/g‑C3N4‑50% (c)
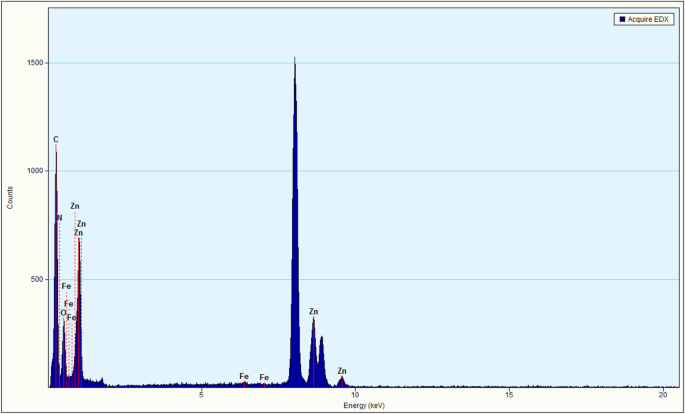
EDS mapping of ZnO/Fe3O4/g‑C3N4‑50%
XPS
Survey spectra showed C, N, O, Zn, and Fe. C 1s peaks at 284.6, 286.5, and 287.8 eV correspond to C–N=C, C–(N)3, and C–N–C. N 1s peaks at 397.9 and 399.2 eV indicate C=N–C and N–(C)3. Zn 2p binding energies confirmed Zn2+. Fe 2p peaks confirmed Fe3O4. The data confirm successful compositing and surface integrity.
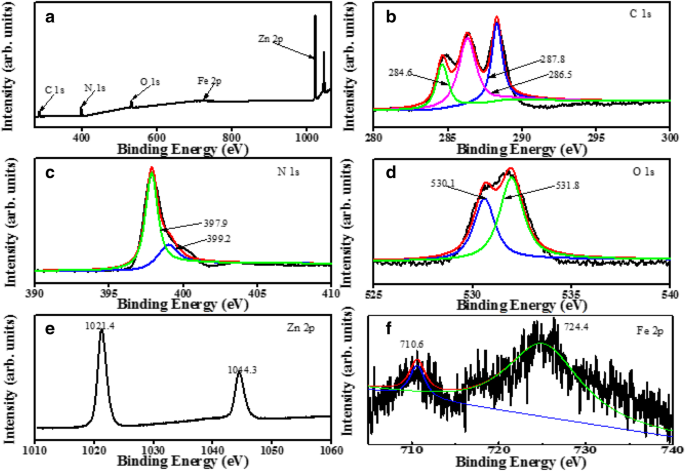
XPS spectra of ZnO/Fe3O4/g‑C3N4‑50%
UV‑vis DRS
Pure ZnO absorbed up to 388 nm (Eg = 3.20 eV). g‑C3N4 shifted to 460 nm (Eg = 2.70 eV). The composite exhibited a pronounced red‑shift, with the 50 % loading showing the broadest visible‑light absorption, indicating effective band‑gap tuning.
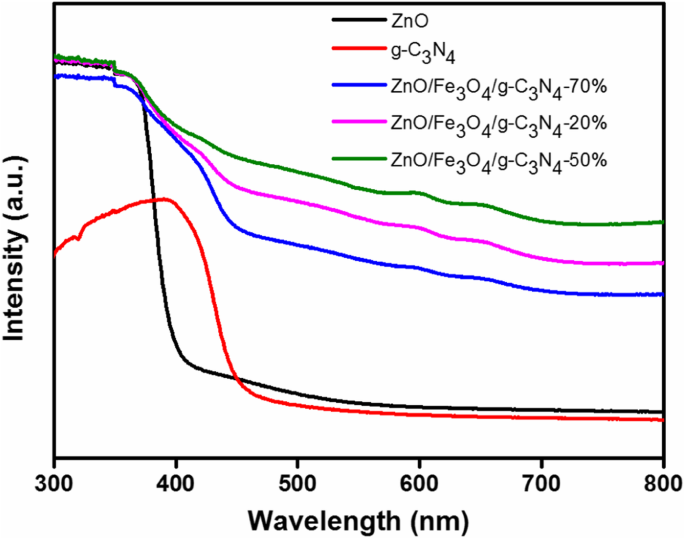
UV‑vis DRS of ZnO, g‑C3N4, and composites (20 %, 50 %, 70 %)
PL
PL intensity decreases with composite formation, reaching the lowest for ZnO/Fe3O4/g‑C3N4‑50%, implying suppressed electron–hole recombination due to effective heterojunctions.
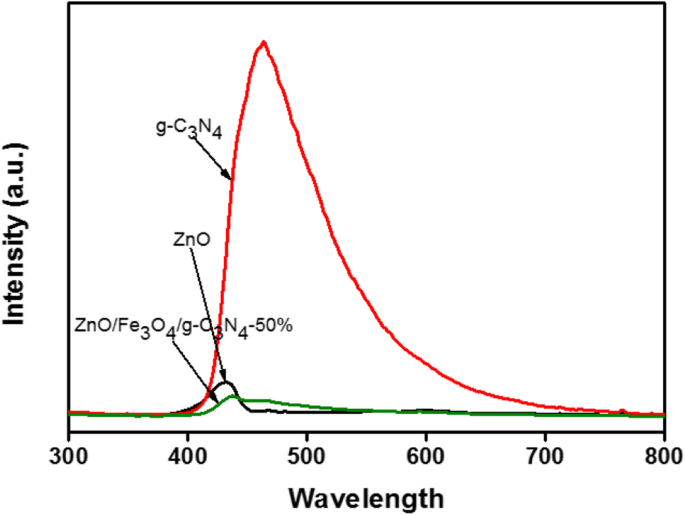
PL spectra of ZnO, g‑C3N4, and ZnO/Fe3O4/g‑C3N4‑50%
Electrochemical Analysis
Transient photocurrent under visible light revealed the highest current for the 50 % composite, indicating superior charge separation. EIS Nyquist plots showed the smallest semicircle radius for the 50 % sample, confirming low interfacial resistance.
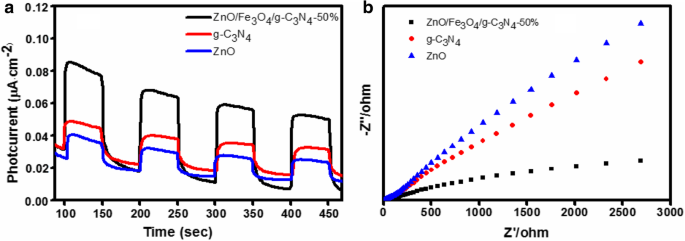
Transient photocurrent (a) and EIS (b) of ZnO, g‑C3N4, and ZnO/Fe3O4/g‑C3N4‑50% under visible light
Magnetic Properties
Fe3O4 exhibited the highest saturation magnetization; the 50 % composite retained sufficient magnetic response for rapid magnetic separation, while remaining superparamagnetic.
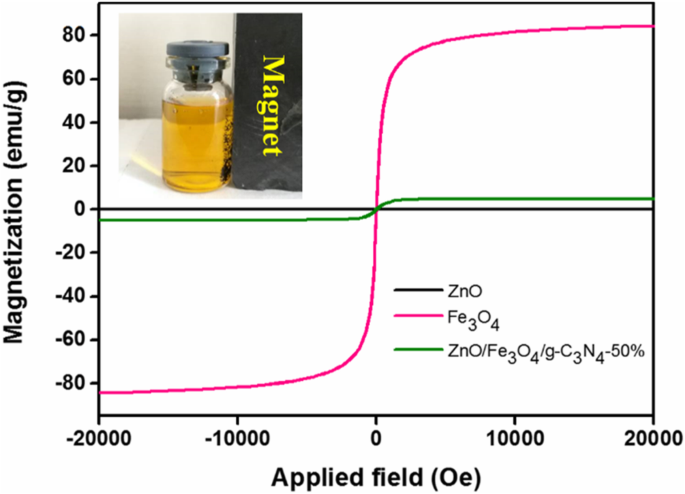
Hysteresis loops of ZnO, Fe3O4, and ZnO/Fe3O4/g‑C3N4‑50%
Photocatalytic Performance
Under visible light, ZnO/Fe3O4/g‑C3N4‑50% achieved 97.87 % MO degradation in 150 min, surpassing pure ZnO and g‑C3N4. First‑order kinetics yielded a rate constant of 0.02430 min‑1, higher than reported TiO2‑based composites.
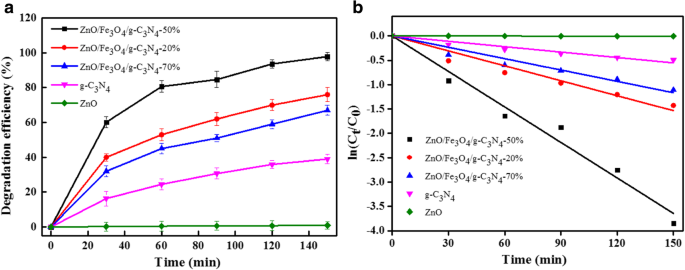
Photocatalytic degradation of MO: efficiency (a) and kinetic curves (b)
Stability and Recyclability
After five magnetic recoveries and re‑use cycles, the composite retained 95.3 % of its initial activity. XRD before and after cycling showed no structural changes, confirming robustness.
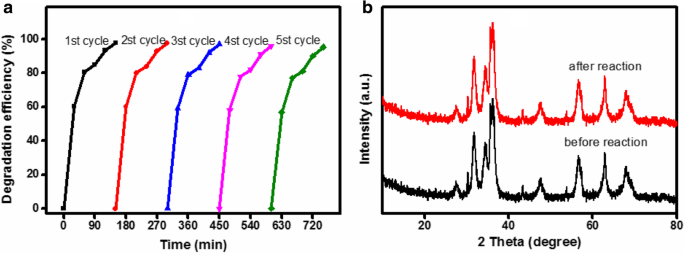
Recycling efficiency (a) and XRD comparison (b) of ZnO/Fe3O4/g‑C3N4‑50%
Degradation of Various Monoazo Dyes
MO, AYR, and OG were degraded to 97.87 %, 98.05 %, and 83.35 % respectively. Lower OG degradation is attributed to its larger aromatic structure and higher sulfonic acid content, which reduce adsorption and reaction rates. A strong negative correlation (R² = 0.9776) exists between dye molecular weight and degradation efficiency.
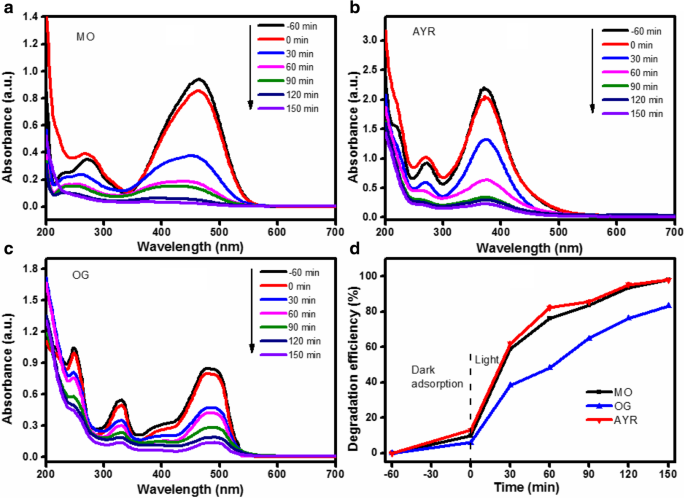
UV‑vis spectra of MO (a), AYR (b), OG (c) and degradation curves (d)
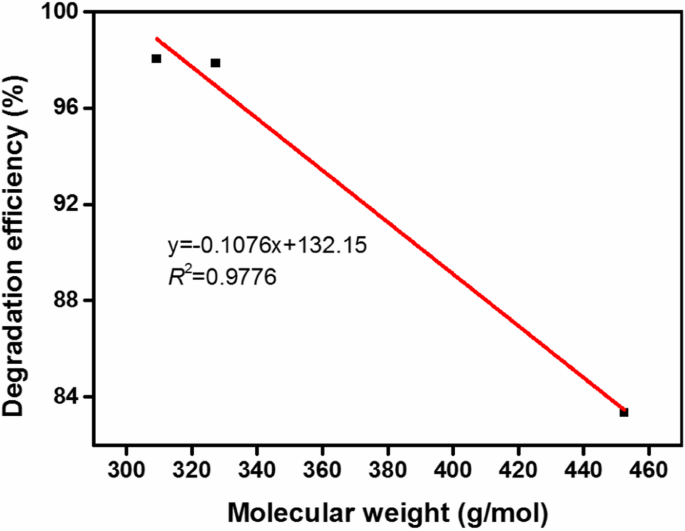
Correlation between dye molecular weight and degradation efficiency
Mechanistic Insight
Scavenger studies reveal that hydroxyl radicals (·OH) and superoxide anions (·O2–) are the dominant reactive species. An n–n heterojunction facilitates electron transfer from the g‑C3N4 conduction band to ZnO, while holes remain in g‑C3N4, generating ·OH from water and ·O2– from dissolved oxygen. The proposed pathway is: ZnO/Fe3O4/g‑C3N4‑50% + hν → e−(CB, ZnO) + h+(VB, g‑C3N4) → ·O2– + ·OH → MO → CO2 + H2O.
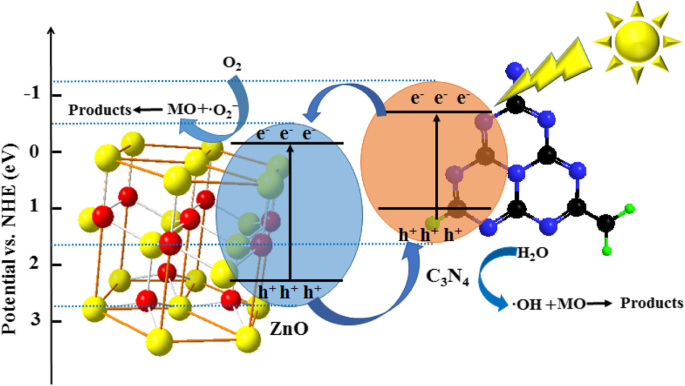
Proposed mechanism for monoazo dye degradation by ZnO/Fe3O4/g‑C3N4‑50%
Conclusions
We successfully fabricated a magnetic ZnO/Fe3O4/g‑C3N4 nanocomposite that combines visible‑light absorption, efficient charge separation, and magnetic recoverability. The 50 % g‑C3N4 composite delivers >95 % dye degradation after five cycles and achieves up to 98 % removal of monoazo dyes. Its robust performance and facile separation position it as a viable candidate for large‑scale wastewater remediation.
Abbreviations
- AO
- Ammonium oxalate
- AYR
- Alizarin yellow R
- CB
- Conduction band
- DRS
- Diffuse reflectance spectroscopy
- EDS
- Energy‑dispersive X‑ray spectroscopy
- EIS
- Electrochemical impedance spectroscopy
- EtOH
- Ethanol
- Fe3O4
- Ferroferric oxide
- g‑C3N4
- Graphite‑like carbon nitride
- MO
- Methyl orange
- OG
- Orange G
- p‑BQ
- p‑Benzoquinone
- PL
- Photoluminescence spectra
- t‑BuOH
- Tert‑butanol
- TEM
- Transmission electron microscopy
- UV
- Ultraviolet
- VB
- Valence band
- VSM
- Vibrating sample magnetometry
- XRD
- X‑ray diffraction
- ZnO
- Zinc oxide
Nanomaterials
- Nanodiamonds: Ultra‑Precise Magnetic Sensors for Biomedical and Quantum Applications
- High-Capacity Few-Layered MoS₂/Acetylene Black Anode for Li‑Ion Batteries
- Pd‑Loaded Zr‑MOF for Visible‑Light Photocatalytic Hydrogen Generation
- Core/Shell CoFe₂O₄/Fe₃O₄ Nanoparticles: Interfacial Magnetism and Tunable Anisotropy
- Efficient Visible‑Light Hydrogen Production with Eosin Y‑Sensitized g‑C3N4/GO Hybrid Loaded with PtNi Alloy Cocatalyst
- High‑Performance Dye‑Sensitized Solar Cells Using Screen‑Printed Multi‑Walled Carbon Nanotube Counter Electrodes
- Carbon Quantum Dot–Platinum Porphyrin Composite: A Dual‑Function Theranostic Agent for Enhanced Photodynamic Cancer Therapy
- High‑Performance Flexible Photocatalytic Paper: Cu2O and Ag Nanoparticle‑Decorated ZnO Nanorods for Visible‑Light Degradation of Organic Dyes
- Magnetite Nanocluster-Based Theranostic Agents for T2‑Weighted MRI and pH‑Responsive Doxorubicin Delivery
- Enhanced Dye Degradation & Antibacterial Performance of Graphene Oxide‑Doped MgO Nanostructures



