One‑Pot Synthesis of Cu₂ZnSnSe₄ Nanoplates and Their Efficient Visible‑Light Photocatalytic Degradation of Rhodamine B
Abstract
A facile SeO₂‑ethanol precursor has enabled the one‑pot synthesis of monodispersed Cu₂ZnSnSe₄ (CZTSe) nanoplates. The resulting quaternary nanoplates exhibit a uniform morphology and a bandgap of ~1.4 eV, making them highly responsive to visible light. When tested as a photocatalyst, the CZTSe nanoplates achieved >90 % degradation of Rhodamine B within 120 min and retained their activity over five consecutive cycles, underscoring their stability and practical potential for wastewater treatment.
Background
Environmental contamination by chemical pollutants in natural water bodies has prompted intensive research into sustainable remediation strategies. Photocatalytic degradation, powered by solar energy, offers a compelling solution, yet conventional photocatalysts such as TiO₂ and ZnO are limited to ultraviolet absorption, which constitutes only ~4 % of the solar spectrum [1]. In contrast, visible light accounts for roughly 50 % of solar irradiance [2], necessitating the development of semiconductors with suitable bandgaps (1.0–1.5 eV) and high absorption coefficients.
Copper‑based quaternary chalcogenides—including Cu₂ZnSnS₄ and Cu₂ZnSnSe₄—have emerged as promising candidates due to their non‑toxic, earth‑abundant composition and excellent optoelectronic properties [4–9]. While extensive studies have focused on CZTSe nanocrystals and thin films, reports on nanoplate morphologies remain scarce [16–17]. Conventional synthesis routes such as hot‑injection or one‑pot thermal methods rely on expensive, toxic or unstable selenium precursors [18–21]. Here, we introduce a simple SeO₂‑ethanol precursor that dissolves readily in ethanol, enabling a streamlined, cost‑effective synthesis of CZTSe nanoplates.
We present a one‑pot thermal chemical method to produce CZTSe nanoplates, evaluate their visible‑light photocatalytic performance, and assess recyclability, highlighting their suitability for environmental remediation applications.
Methods/Experimental
Synthesis of CZTSe Nanoplates
All reagents were sourced from Aladdin and used without further purification. In a typical synthesis, 1.0 mmol Cu(acac)₂, 0.5 mmol Zn(OAc)₂·2H₂O, 0.5 mmol SnCl₂·2H₂O, and 2.0 mmol SeO₂ dissolved in 4 mL ethanol were added to 20 mL oleylamine (OLA) in a 100 mL three‑neck flask. The mixture was degassed at 130 °C for 1 h, purged with Ar for 30 min, then heated to 280 °C for 1 h. The resulting black precipitate was washed three times each with hexane and ethanol by centrifugation (8000 rpm, 5 min) and dried at 60 °C under vacuum. Prior to photocatalytic testing, the nanoplates were rendered hydrophilic by Na₂S treatment to remove residual OLA ligands [8].
Characterizations
Phase purity was confirmed by powder X‑ray diffraction (XRD, Rigaku D/max 2200, Cu Kα) and Raman spectroscopy (Renishaw InVia, 514 nm). Morphology and crystallinity were examined by transmission electron microscopy (TEM, JEOL JEM‑2100F) and scanning electron microscopy (SEM, FEI Quatan 250FEG). UV‑vis absorption spectra of the CZTSe powders and Rhodamine B (RhB) solutions were recorded using a Perkin Elmer Lambda spectrometer with an integrating sphere and cuvette, respectively.
Photocatalytic Activity Measurements
The visible‑light photocatalytic performance was evaluated by RhB degradation (10 mg L⁻¹) under ambient conditions. A 300‑W Xe lamp equipped with a 420 nm cutoff filter served as the illumination source. In a 100 mL solution, 50 mg of the CZTSe photocatalyst was added and stirred in the dark for 12 h to reach adsorption–desorption equilibrium. Residual RhB concentrations were monitored every 20 min at 554 nm via UV‑vis spectroscopy, and degradation rates were calculated using the Beer–Lambert law.
Results and Discussion
Figure 1a shows the XRD pattern of the as‑synthesized CZTSe, with all peaks matching the tetragonal kesterite structure (JCPDS No. 70‑8930). Key reflections at 27.1°, 45.1°, 53.5°, 65.8°, and 72.5° correspond to the (112), (204), (312)/(116), (400)/(008), and (332) planes, respectively. Raman analysis (Figure 1b) further confirms the single‑phase nature of the nanoplates, with no detectable secondary peaks from CuₓSe, ZnSe, or Cu₂SnSe₃ [16–17].
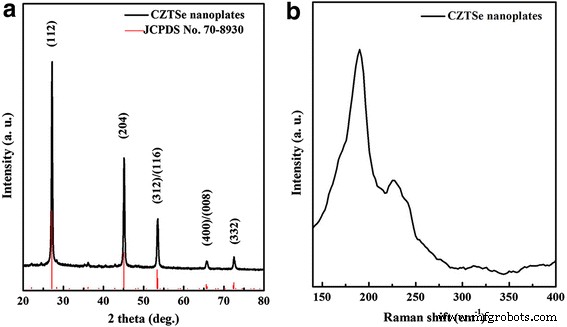
a XRD pattern and b Raman spectrum of CZTSe nanoplates
SEM and TEM images (Figure 2) reveal a well‑defined platelet morphology with an average lateral size of ~210 nm, corroborated by SAED patterns that confirm high crystallinity. HRTEM imaging shows a clear 0.33‑nm lattice spacing, attributable to the (112) planes of CZTSe.

a SEM image. b TEM image (inset: SAED pattern). c HRTEM image of CZTSe nanoplates
UV‑vis absorption (Figure 3a) demonstrates strong absorption across the entire visible spectrum. The Tauc plot (Figure 3b) yields a bandgap of ~1.4 eV, slightly larger than bulk CZTSe due to quantum confinement effects [9].
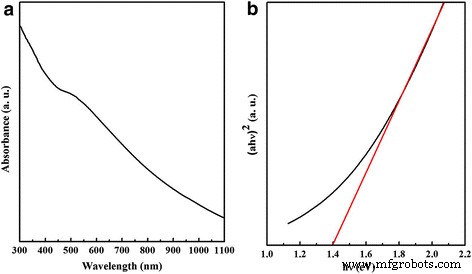
a UV‑vis absorption spectrum and b bandgap of CZTSe nanoplates
Photocatalytic tests reveal that ~90 % of RhB is degraded within 120 min under visible light (Figure 4a). Reusability studies over five cycles show negligible loss in activity, confirming the structural robustness of the nanoplates (Figure 4b). Radical trapping experiments using Ar, ammonium oxalate (AO), tert‑butanol (TBA), and benzoquinone (BQ) indicate that superoxide radicals (•O₂⁻) and photogenerated holes (h⁺) are the primary oxidative species, whereas hydroxyl radicals play a minor role (Figure 4c). The proposed mechanism (Figure 4d) illustrates electron transfer from the valence to the conduction band, followed by •O₂⁻ generation and subsequent dye degradation.
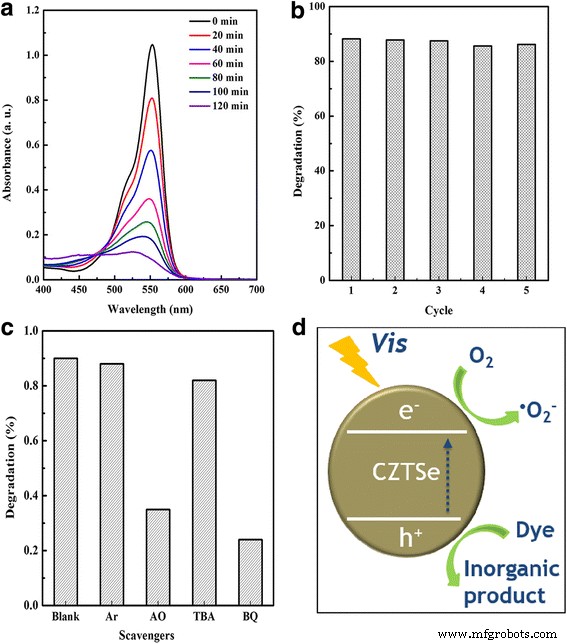
a RhB degradation. b Cycle test. c Effects of various quenchers on the degradation efficiency of RhB. d Scheme of the photocatalytic degradation process
Conclusions
We have demonstrated a straightforward SeO₂‑ethanol precursor for the one‑pot synthesis of monodisperse CZTSe nanoplates with a ~1.4 eV bandgap. The resulting nanoplates exhibit high visible‑light photocatalytic activity, achieving >90 % Rhodamine B degradation in 120 min and maintaining performance over multiple cycles. These findings confirm that CZTSe nanoplates are promising, stable photocatalysts for environmental remediation under solar‑sized visible illumination.
Nanomaterials
- High‑Performance Au/Ce‑La Nanorod Catalysts for Low‑Temperature CO Oxidation: Synthesis, Characterization, and Mechanistic Insights
- Water‑Soluble Antimony Sulfide Quantum Dots: Efficient Synthesis and Superior Photo‑Electric Performance
- Zinc Oxide Nanoparticles: Antimicrobial Properties, Mechanisms, and Applications
- Ligand‑Free Iridium Nanoparticles: A Simple Aqueous Synthesis and Demonstrated In‑Vitro Biocompatibility
- Rapid One‑Pot Synthesis of Monodisperse CoFe₂O₄@Ag Core–Shell Nanoparticles with Exceptional Plasmonic and Magnetic Properties
- Ag3PO4/BiFeO3 Heterojunctions: Superior Visible‑Light Photocatalytic Degradation of Acid Orange 7
- Optimizing ZnO‑Based Nanohybrids: How Materials, Heterojunctions, and Crystal Orientation Enhance Methyl Orange Degradation
- Hierarchical Au@CdS‑CdS Nanoflowers: Stepwise Fabrication and Superior Photocatalytic Performance
- Synthesis and Enhanced Photocatalytic Degradation of Rhodamine B by Ag3PO4/T‑ZnO Whisker Heterostructures
- Anion‑Controlled Fabrication of ZnO 1D Necklace‑Like Nanostructures for Superior Photocatalytic Performance



