Rapid One‑Pot Synthesis of Monodisperse CoFe₂O₄@Ag Core–Shell Nanoparticles with Exceptional Plasmonic and Magnetic Properties
Abstract
Monodispersed magnetic core–shell nanoparticles are pivotal for magnetic fluids, catalysis, and biomedical imaging. Conventional syntheses typically involve multiple steps—solvent exchange, ligand replacement, and repeated centrifugation—slowing down progress toward practical applications. We present a streamlined, single‑pot thermal decomposition protocol that produces CoFe₂O₄ cores uniformly coated with an Ag shell, achieving a polydispersity index (PDI) of 0.083 in hexane. The resulting particles display a sharp localized surface plasmon resonance (LSPR) from the Ag shell and exhibit soft‑magnetic behavior at room temperature (H_c ≈ 70 Oe) while retaining hard‑magnetic characteristics (H_c ≈ 11 kOe) at 5 K.
Background
Core–shell magnetic nanoparticles have attracted intense interest across engineering and biomedical fields, enabling advanced magnetic separation, recoverable catalysis, drug delivery, and enhanced MRI contrast (see Refs. [1–11]). Spinel ferrites, particularly cobalt ferrite (CoFe₂O₄), are preferred cores due to their high coercivity, chemical stability, and mechanical hardness (Refs. [12–17]). Thermal decomposition has emerged as a reliable route to produce highly crystalline, size‑controlled ferrite nanoparticles (Refs. [13,17,18]).
While Au‑shells confer both plasmonic and thiol‑binding functionality, Ag‑shells offer superior extinction coefficients, sharper resonance bands, and stronger local fields (Refs. [19–25]). Despite this, Ag‑coated magnetic cores have rarely been synthesized via one‑pot routes. Our study demonstrates that a two‑step thermal decomposition within a single reaction vessel can yield precise CoFe₂O₄@Ag core–shell nanostructures, confirmed by TEM, XRD, XPS, and magnetic measurements.
Method/Experimental
Materials
Fe(acac)₃, Co(acac)₂, diphenyl ether, oleylamine (OAm), silver(I) acetate, and oleic acid (OA) were sourced from Tokyo Chemical Industry, Wako, and Kanto Chemical, respectively.
One‑Pot CoFe₂O₄@Ag Synthesis
The procedure follows Scheme 1. Initially, 1 mmol Fe(acac)₃, 0.5 mmol Co(acac)₂, and 12 mmol OA were dissolved in 30 mL diphenyl ether pre‑heated at 180 °C for 30 min. The mixture was refluxed at 180 °C for 16 h, producing black CoFe₂O₄ cores. After cooling, a second solution containing 5.2 mmol OA, 30.4 mmol OAm, and 3.6 mmol silver acetate in 100 mL diphenyl ether was added, and the temperature was raised to 180 °C for 1.5 h to grow the Ag shell. The reaction mixture turned metallic purple, confirming shell formation. Methanol (400 mL) was introduced to precipitate the particles, followed by sequential centrifugation (5000 rpm, 5 min) and hexane dispersion (60 mL). A final high‑speed spin (14 000 rpm, 20 min) yielded a colloid of ~60 mg nanoparticles (~1 mg mL⁻¹). CoFe₂O₄ cores (reference) were prepared by omitting the second step.
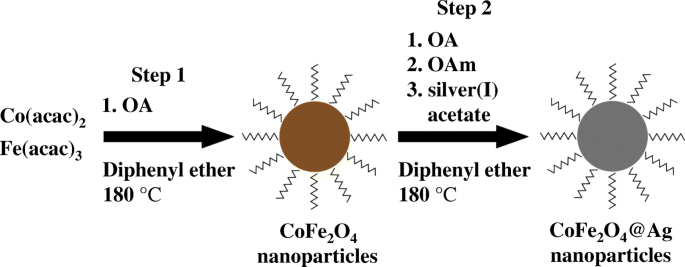
Procedure for synthesizing CoFe₂O₄@Ag nanoparticles
Characterization and Calculations
Transmission electron microscopy (TEM, Hitachi FE 2000) revealed particle morphology. X‑ray diffraction (XRD, PANalytical X’Pert PRO MPD) assessed crystal phases (Cu Kα, 2θ = 20°–80°). Elemental composition was probed by X‑ray photoelectron spectroscopy (XPS, KARATOS ESCA 3400) with Ar ion sputter etching. Magnetization was measured by SQUID (Cryogenic S700X‑R). UV–vis absorption (Jasco V‑670) quantified plasmonic behavior. Dynamic light scattering (DLS, Malvern Zetasizer Nano‑ZS, 633 nm) evaluated colloidal stability. Optical modeling employed Mie theory calculations (Bohren & Huffman) via MATLAB code by Mätzler, using Ag dielectric functions from Ref. [25].
Results and Discussion
Figure 1 shows TEM images and size histograms of CoFe₂O₄ cores and CoFe₂O₄@Ag core–shell particles. The cores average 3.5 ± 0.76 nm, while the shell‑coated particles average 5.5 ± 0.77 nm, implying a ~1 nm Ag shell. Core aggregation was evident, whereas core–shell particles remained dispersed, attributed to the reduced surface energy conferred by the Ag shell (Ref. [26]). No residual cores were detected in the shell‑coated sample, confirming complete coverage.
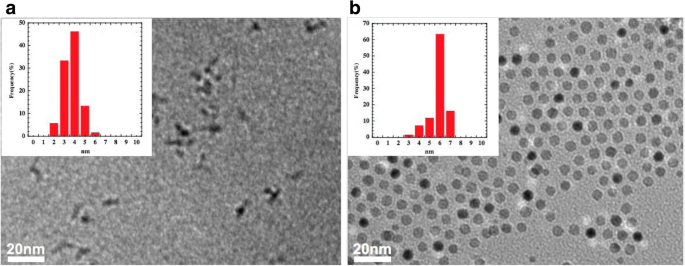
TEM images and particle size histograms for nanoparticles of a CoFe₂O₄ and b CoFe₂O₄@Ag
Figure 2 presents XRD patterns. CoFe₂O₄ peaks at 2θ = 30.50°, 35.75°, 43.50°, 53.8°, 57.5°, 63.0°, and 74.4° confirm a cubic spinel phase (Ref. [17]). The CoFe₂O₄@Ag pattern shows additional fcc Ag peaks at 2θ = 38.42°, 44.50°, 64.91°, 77.75°, and 81.83° (Ref. [10]). Debye–Scherrer analysis yields crystallite sizes of 7.1 nm (core) and 3.6 nm (core–shell), consistent with TEM and indicative of a uniform Ag coating.
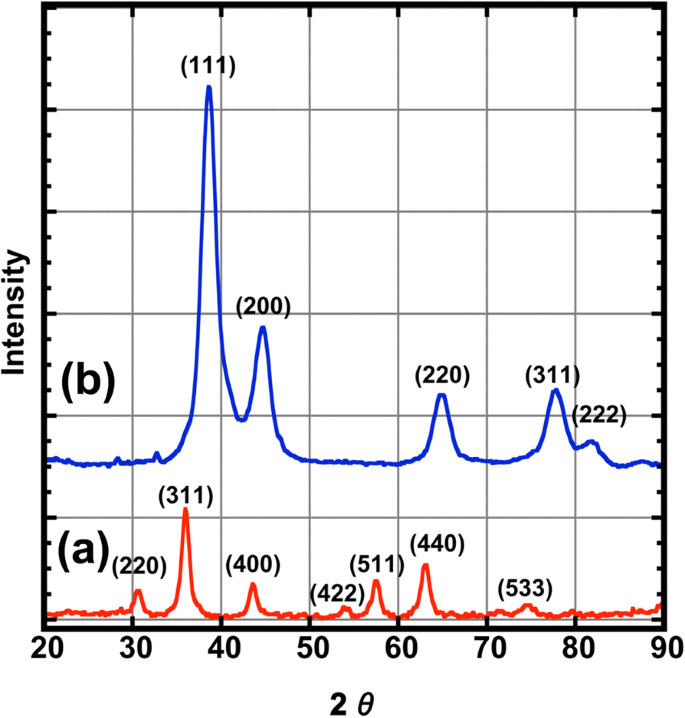
XRD pattern of for nanoparticles, (a) CoFe₂O₄ (red line) and (b) CoFe₂O₄ @Ag (blue line)
XPS spectra before and after Ar ion etching (Fig. 3) confirmed the core–shell architecture. Surface spectra showed only Ag 3d peaks and C 1s from oleic acid, while Fe 2p and Co 2p appeared only after etching, confirming complete core coverage. Deconvolution of Ag 3d revealed distinct surface and subsurface components, further supporting a uniform shell.
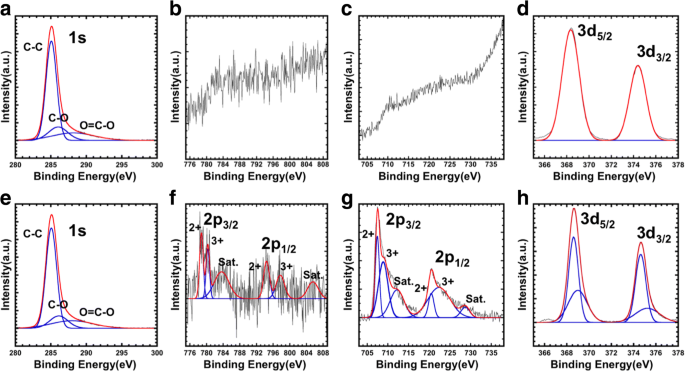
XPS spectra of CoFe₂O₄@Ag by argon ion etching before (a–d) and after (e–h). a, e C 1s. b, f Co 2p. c, g Fe 2p. d, h Ag 3d
Magnetic hysteresis (Fig. 4) shows that CoFe₂O₄ cores are superparamagnetic at 300 K (H_c ≈ 0 Oe) but exhibit a coercivity of 7 kOe at 5 K. In contrast, core–shell particles display a modest soft‑magnetic response at 300 K (H_c ≈ 70 Oe) and a pronounced hard‑magnetic behavior (H_c ≈ 11 kOe) at 5 K. The reduction in saturation magnetization for core–shell particles (M_s = 3.3 emu g⁻¹) relative to cores (M_s = 11 emu g⁻¹) reflects the diamagnetic contribution of the Ag shell (Refs. [8–10,32–34]).
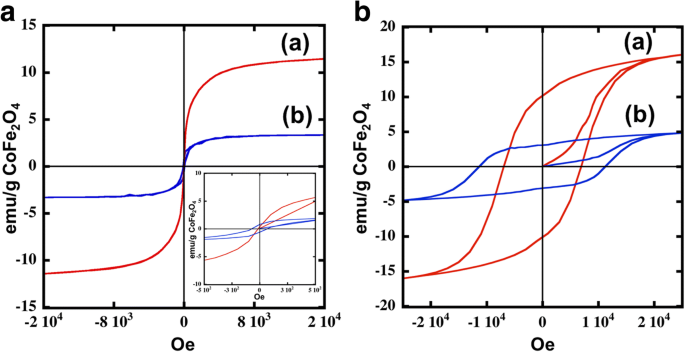
Hysteresis loops for nanoparticles: (a) and (b) are for the CoFe₂O₄ nanoparticles (red line) and CoFe₂O₄ @Ag nanoparticles (blue line), respectively, at a 300 K and b at 5 K
UV–vis spectroscopy (Fig. 5) reveals a pronounced LSPR peak at 416 nm for core–shell particles, absent in the bare cores. Mie theory calculations corroborate this resonance, confirming that the Ag shell is responsible for the optical response (Refs. [24,25]). The optical properties remained stable after one month in air, underscoring the chemical robustness of the particles.
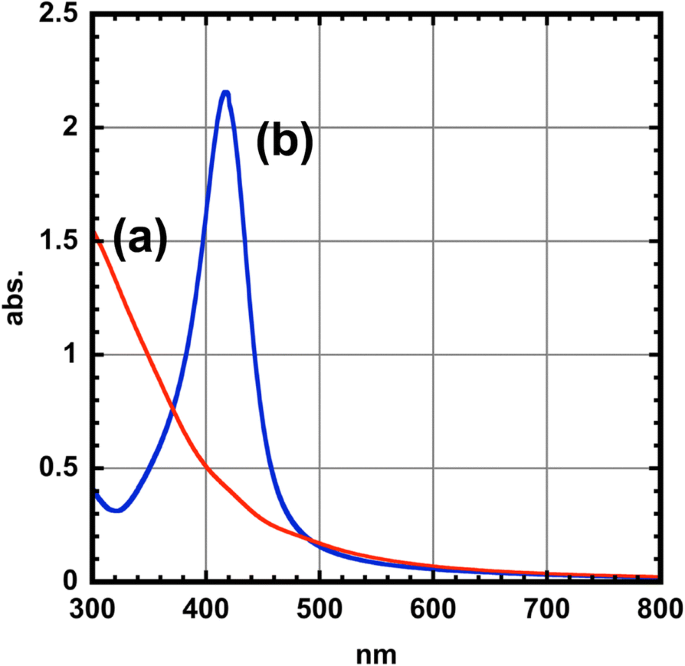
UV-vis spectra for (a) CoFe₂O₄ nanoparticles (red line) and (b) CoFe₂O₄ @Ag nanoparticles (blue line)
Dynamic light scattering (DLS) confirmed colloidal stability. CoFe₂O₄ cores exhibited a hydrodynamic diameter of 19.67 nm, while core–shell particles measured 9.27 nm, indicating that the Ag shell prevents aggregation (Table 2). The low PDI (0.083) for core–shell particles further attests to their monodispersity and enhanced stability.
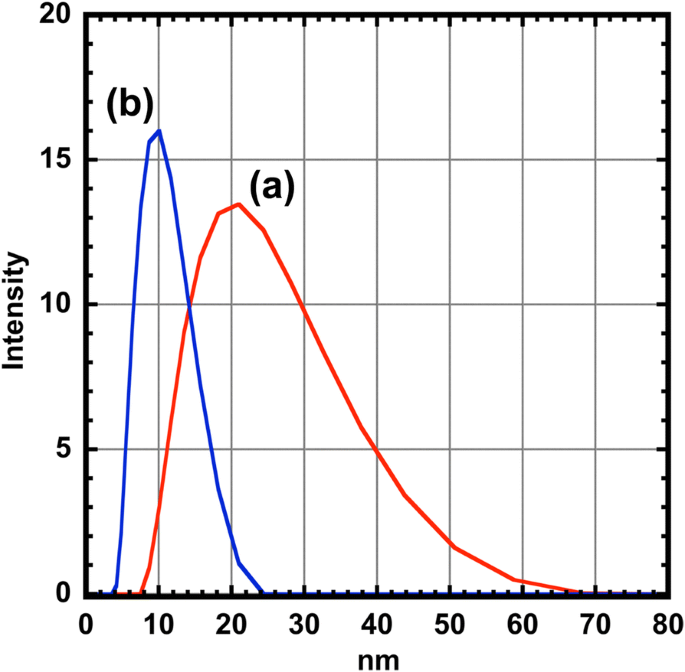
Size distribution (a) of the CoFe₂O₄ (red line) and (b) the CoFe₂O₄@Ag nanoparticles (blue line) measured by DLS
Conclusions
We have demonstrated that a single‑pot thermal decomposition route yields highly monodisperse CoFe₂O₄@Ag core–shell nanoparticles with a uniform ~1 nm Ag shell. These particles combine plasmonic extinction (416 nm) with tunable magnetic behavior—soft at room temperature and hard at cryogenic temperatures—while maintaining excellent colloidal stability. The straightforward synthesis and multifunctionality position these nanoparticles as promising candidates for magnetic separation, catalysis, and biomedical imaging.
Nanomaterials
- Cobalt‑Doped FeMn₂O₄ Spinel Nanoparticles: Size‑Controlled Synthesis and Magnetic Behavior
- Eco‑Friendly Copper Oxide Nanoparticles Doped with Ginger and Garlic Extracts Exhibit Potent Antibacterial Activity Against Escherichia coli
- How Synthesis Route Shapes the Structure, Magnetism, and Hyperthermia Performance of La₁₋ₓSrₓMnO₃ Nanoparticles
- One‑Pot Synthesis of Cu₂ZnSnSe₄ Nanoplates and Their Efficient Visible‑Light Photocatalytic Degradation of Rhodamine B
- Optimized Fe³⁺‑Grafted BiOCl for Rapid Adsorption of Trace Cationic and Anionic Dyes in Aqueous Wastewater
- Green Synthesis of Metal‑Oxide Nanoparticles with Gum Karaya and Their Ecotoxicological Impact on *Chlamydomonas reinhardtii*
- Ligand‑Free Iridium Nanoparticles: A Simple Aqueous Synthesis and Demonstrated In‑Vitro Biocompatibility
- Comprehensive Review of Luminescent Silica Nanoparticles: Classification, Synthesis, and Practical Applications
- Bibliometric Trends in Drug Delivery and Magnetic Nanoparticles (1980‑2017)
- Eco‑Friendly, Cost‑Effective Synthesis of Tin Oxide Nanoparticles: A Comprehensive Review of Methods, Formation Mechanisms, and Applications



