High‑Quality In₂S₃ Quantum Dots Synthesized at Room Temperature for Ultra‑Sensitive UV Photodetectors
Abstract
In this study, we introduce a simple, ambient‑temperature synthesis of In₂S₃ quantum dots (QDs) that delivers superior crystal quality and narrow size distribution. Using sodium sulfide, indium chloride, and sodium dodecyl sulfate as a surfactant, we obtained monodispersed QDs with an average diameter of 2 nm. Photodetectors fabricated from these QDs demonstrate a detectivity of ~10¹³ Jones under 365 nm UV illumination at room temperature, highlighting their promise for high‑performance, low‑cost optoelectronic devices.
Background
Two‑dimensional and zero‑dimensional nanomaterials have revolutionised optoelectronics due to their exceptional electronic and optical properties [1, 2]. Quantum dots (QDs) in particular offer tunable bandgaps and high photoluminescence quantum yields, making them attractive for photovoltaics, photodetectors, bio‑imaging, and photocatalysis [3–10]. Among III–VI semiconductors, indium sulfide (In₂S₃) QDs exhibit a direct bandgap and favourable thermal and mechanical stability, yet conventional synthesis routes such as hydrothermal, template, and microwave methods are often limited by high temperature, long reaction times, or complex post‑processing [12–15].
To overcome these challenges, we report a mild, surfactant‑assisted synthesis that produces stable, high‑quality In₂S₃ QDs at atmospheric pressure and room temperature. The resulting QDs are characterised extensively, and their potential in UV photodetection is demonstrated through device fabrication and performance metrics.
Methods
Materials
Sodium sulfide (Na₂S·9H₂O), indium chloride (InCl₃·4H₂O), sodium dodecyl sulfate, and a 300 Da regenerated cellulose dialysis membrane were purchased from commercial suppliers and used without further purification.
In₂S₃ QDs Fabrication
The synthesis follows a two‑step mixing protocol (Fig. 1a). 0.1 M Na₂S and 0.1 M InCl₃ solutions were prepared in deionised water. Each solution was combined with 0.008 M SDS and stirred at 1500 rpm for 20 min to generate monodispersed complexes. The Na₂S–SDS mixture was then added to the InCl₃–SDS solution, initiating a rapid reaction that yielded a yellow suspension within 10 min. The product was washed by centrifugation (3000 rpm, 5 min) and purified via dialysis. The final QDs were collected inside the dialysis bag.
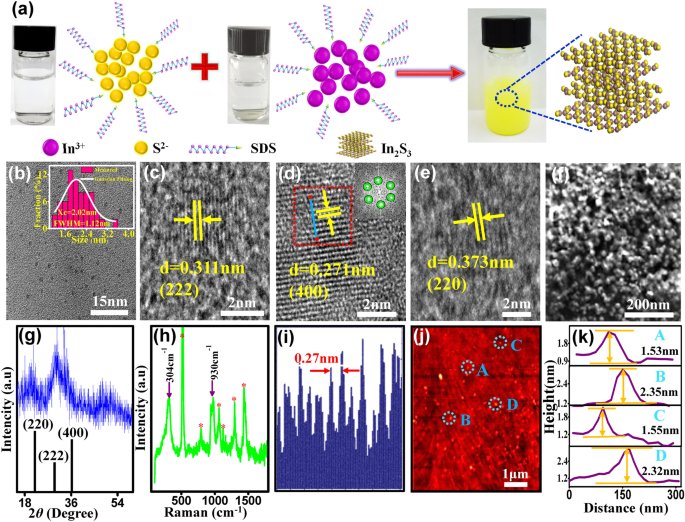
a Schematic of the In₂S₃ QD synthesis. b TEM image and size distribution (Gaussian fit). c–e HRTEM images with FFT inset of a selected region. f SEM image. g XRD pattern. h Raman spectrum. i Line profile of lattice fringes in (d). j AFM image. k Height analysis of selected QDs.
Characterization
High‑resolution TEM (JEM‑2100, 200 kV) examined particle morphology. SEM (FEI Quanta 200) and AFM (SPA‑400) provided surface and topography data. XRD (Rigaku D/Max‑RA) and Raman (Renishaw in via) assessed crystallinity. UV‑vis, UV‑vis‑NIR (UV‑3600), and fluorescence (Hitachi F‑7000) spectroscopies quantified optical behaviour. XPS (PHI Versa Probe II) verified surface chemistry. Electrical performance was evaluated using a Keithley 2400 source meter for J‑V curves and a Keysight B1500A analyzer for C‑V measurements.
Results and Discussion
Structure and Morphology Studies
Figure 1b–e show that the In₂S₃ QDs are spheroidal, uniformly distributed, and exhibit a narrow size distribution following a Gaussian trend (1–3 nm, FWHM = 1.12 nm; mean = 2.02 nm). HRTEM reveals lattice spacings of 0.271 nm, 0.311 nm, and 0.373 nm, corresponding to the 400, 222, and 220 planes of the cubic phase [18]. The FFT pattern confirms a well‑defined crystalline structure. SEM images indicate that QDs aggregate into compact clusters, reducing surface energy. XRD peaks match the 400, 222, and 220 planes, with particle sizes calculated via the Scherrer equation agreeing with TEM measurements. Raman peaks at 304 cm⁻¹ and 930 cm⁻¹ are consistent with literature [19]. AFM height measurements (1.53–2.35 nm) corroborate the TEM size, yielding an average height of 1.94 nm.
Using the Brus equation, the QDs exhibit a quantum‑confined bandgap of 3.50 eV, exceeding the bulk value of 2.3 eV. UV‑vis absorption spectra display two peaks at 225 nm and 283 nm (Fig. 2a). The bandgap is also extracted from (αhv)² vs. hv plots, giving 3.54 eV, in excellent agreement with the theoretical estimate.
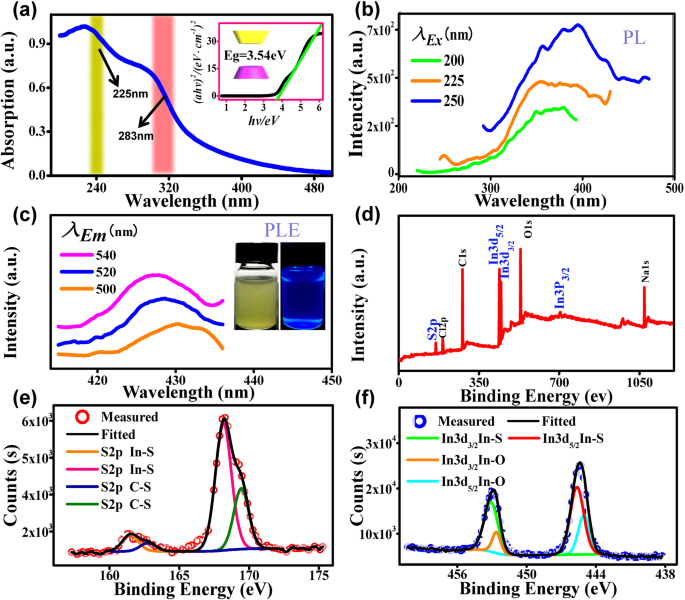
a UV‑vis absorption; inset: bandgap estimate. b PL emission (300–450 nm). c PLE spectra; inset: fluorescence under visible and 365 nm UV. d XPS full scan. e XPS S 2p. f XPS In 3d₅/₂ and 3d₃/₂.
PL peaks around 390 nm under 250 nm excitation confirm strong UV fluorescence, while PLE peaks at 500–540 nm illustrate a broadened absorption range due to quantum confinement. XPS confirms the presence of In–S and S–C bonds, with minor Cl, Na, O, and C from reagents and surfactant.
Photodetectors fabricated with these QDs (Fig. 3a) exhibit a responsivity of 4.13 A W⁻¹ at 1 V and a detectivity of 1×10¹³ Jones at 365 nm UV, surpassing many 2D nanomaterial devices [24, 25]. The I‑V characteristics show rectifying behaviour that scales with reverse bias and incident power density.
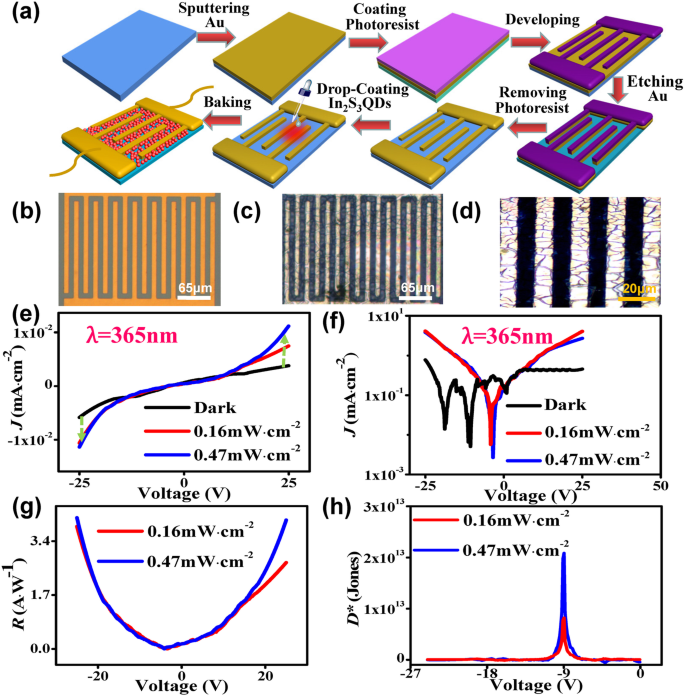
a Device fabrication schematic. b Bare Au electrodes. c‑d Optical micrographs of QD‑filled electrodes. e‑h Device performance: J‑V, log‑J‑V, R‑V, and D* curves.
Temperature‑dependent resistance measurements (Fig. 4b) reveal a slight decrease with increasing temperature, while ln ρ vs. 1/T analysis yields a thermal activation energy of 0.011 eV and a finger‑leading factor of 4.16×10⁸ Ω cm. Capacitance‑frequency studies (Fig. 4d) indicate interface states that dampen AC signals at high frequencies. C‑V profiling provides a built‑in potential of 0.6 V and a carrier concentration of 4.3×10¹⁹ cm⁻³, leading to a depletion width of 12.34 nm (Fig. 4f). These parameters are comparable to or exceed those reported for graphene QD devices [30, 31], explaining the superior photodetector performance.
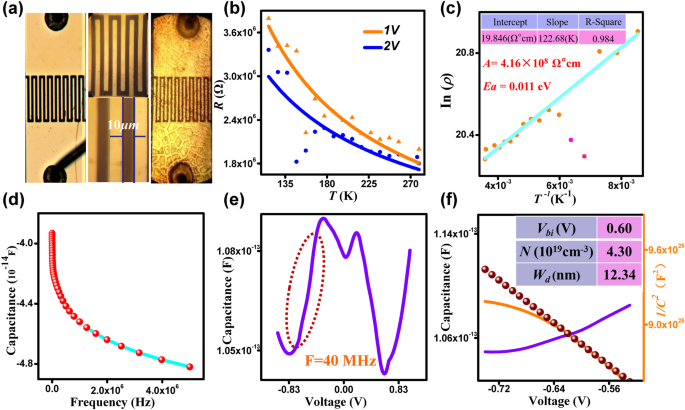
a Photodetector with QD active layer. b R‑T at 1 V and 2 V. c ln ρ‑1/T. d C‑F curves. e C‑V at 40 MHz. f 1/C² vs. V analysis.
Conclusions
We have established a rapid, low‑temperature route to produce crystalline In₂S₃ QDs with a 2 nm average size and narrow size distribution. These QDs serve as an efficient, standalone photoactive layer in UV photodetectors, achieving a record detectivity of 2×10¹³ Jones at room temperature under 365 nm illumination without amplification. The method’s simplicity, low cost, and scalability position it as a compelling platform for high‑performance, large‑area In₂S₃ QD photodetectors.
Abbreviations
- AFM:
Atomic force microscope
- CMC:
Critical micelle concentration
- FFT:
Fast Fourier transform
- FWHM:
Full width at half maximum
- HRTEM:
High‑resolution transmission electron microscope
- PL:
Photoluminescence
- PLE:
Photoluminescence excitation
- QDs:
Quantum dots
- SDS:
Sodium dodecyl sulfate
- SEM:
Scanning electron microscope
- TEM:
Transmission electron microscope
- XPS:
X‑ray photoelectron spectroscopy
- XRD:
X‑ray diffractometer
Nanomaterials
- Cobalt‑Doped FeMn₂O₄ Spinel Nanoparticles: Size‑Controlled Synthesis and Magnetic Behavior
- Phosphine‑Free Synthesis of Reabsorption‑Suppressed ZnSe/CdS/ZnS Core–Shell Quantum Dots and Their Application in Sensitive CRP Detection
- Water‑Soluble Antimony Sulfide Quantum Dots: Efficient Synthesis and Superior Photo‑Electric Performance
- Efficient One‑Step Photo‑Ultrasonic Synthesis of rGO/Ag₃PO₄ Quantum‑Dot Composites for Enhanced Visible‑Light Photocatalysis
- Blue‑Emitting Mn‑Alloyed CdTe Quantum Dots: Synthesis, Structural Insight, and Electrochemical Characterization
- High‑Performance Dual‑Emissive Mn‑Doped InP/ZnS Quantum Dots with 78 % Photoluminescence Quantum Yield: A Growth‑Doping Approach
- Tailoring Optical Properties of InAs Quantum Dots with InAlAs Interlayers in GaAsSb Strain‑Reducing Layers
- Synthesis, Structure, and Superior Capacitance of Sulfur‑Nitrogen Co‑Doped Graphene Quantum Dots
- Eco‑Friendly One‑Pot Hydrothermal Synthesis of Water‑Soluble WS₂ Quantum Dots for Sensitive Luminescent Detection of Hydrogen Peroxide and Glucose
- Improving WLED Color Rendering with Composite Orange‑Red Quantum Dots



