Impact of Sintering Temperature on the Electrochemical Performance of Ce0.8Sm0.05Ca0.15O2-δ/La0.6Sr0.4Co0.2Fe0.8O3-δ Heterostructure Electrolytes
Abstract
Semiconductor‑ionic materials (SIMs) have emerged as a compelling class of electrolytes for low‑temperature solid‑oxide fuel cells (LT‑SOFCs) due to their exceptional ionic conductivity. In this study we investigate a composite pellet comprising the mixed‑valence perovskite La0.6Sr0.4Co0.2Fe0.8O3-δ (LSCF) and the doped ceria Ce0.8Sm0.05Ca0.15O2-δ (SCDC). The composite was sintered at 600, 800, 900 and 1000 °C, and the resulting fuel cells were evaluated at 550 °C. A pellet sintered at 600 °C delivered a peak power density (Pmax) of 543 mW cm–2, with a respectable 312 mW cm–2 even at 500 °C. In stark contrast, the 1000 °C pellet achieved only 106 mW cm–2. The pronounced performance decline is attributed to the loss of interfacial pathways between SCDC and LSCF at high sintering temperatures, underscoring the pivotal role of interfacial conduction in SIMs.
Introduction
Hydrogen fuel has attracted intense research interest as a clean, high‑energy carrier [1, 2]. Solid‑oxide fuel cells (SOFCs) can convert hydrogen directly into electricity without combustion, offering high efficiency, fuel flexibility and environmental benefits [3, 4]. Nevertheless, conventional SOFCs suffer from high operating temperatures (> 800 °C) and the requirement of high‑temperature sintering (> 1000 °C) to obtain dense electrolytes, both of which inflate costs and impede commercialization [5–7]. Electrolyte‑free fuel cells (EFFCs) based on SIMs provide an attractive alternative: by combining a semiconductor with an ionic conductor, they generate high ionic conductivity even at < 600 °C, eliminating the need for high‑temperature sintering [8]. Previous work has shown that SrTiO3‑YSZ, SDC‑SrTiO3 and Ce0.8Gd0.2O2‑δ/CoFe2O4 heterostructures exhibit ionic conductivities that exceed those of their individual constituents by orders of magnitude [9–11]. The interfacial region between the two phases—characterized by space‑charge layers and lattice misfit—plays a crucial role in facilitating ionic transport [12–15]. However, the influence of sintering temperature on the microstructure, interfacial integrity and resulting electrochemical performance of SIM composites remains poorly understood. In this work we systematically sinter LSCF‑SCDC composites at 600–1000 °C, examine their microstructure, and evaluate the electrochemical performance of EFFC devices to elucidate the relationship between sintering conditions, interfacial preservation and ionic conductivity.
Method
Materials synthesis
The SCDC phase was synthesized via a one‑step co‑precipitation route. Cerium, samarium and calcium nitrates were dissolved in deionized water to yield a 1 M solution. A 1 M sodium carbonate solution (metal:carbonate = 1:1.5) served as precipitant; the mixture was added dropwise (10 mL min–1) under vigorous stirring, producing a white precipitate. After filtration, the precipitate was washed, dried at 120 °C for 10–12 h, and calcined at 800 °C for 4 h. The resultant powder was ground to a fine, yellowish product. Commercial LSCF (Ningbo SOFCMAN Energy Technology Co., Ltd.) was used without further modification. Composite powders (40 % LSCF, 60 % SCDC) were pressed at 220 MPa into 13 mm discs (~1.2 mm thick) and sintered in static air at 600, 800, 900 and 1000 °C (10 h, 10 °C min–1 ramp).
Microstructural characterization
Phase identification employed a Bruker D8 X‑ray diffractometer (Cu Kα, λ = 1.54060 Å). Morphology and elemental distribution were examined using a JEOL JSM‑7100F field‑emission SEM equipped with an Oxford EDS detector.
Fuel cell fabrication and performance test
NCAL powder (Tianjin Baomo) was dispersed in terpineol to form a slurry, then coated on nickel foam to create Ni‑NCAL anodes and cathodes. After drying at 120 °C, the LSCF‑SCDC electrolyte was sandwiched between the electrodes to form a full cell. Cells were pre‑heated to 550 °C for 1 h before testing. Hydrogen (80–120 mL min–1) and air (150–200 mL min–1, 1 atm) were supplied, and I‑V and I‑P curves were recorded with an ITECH8511 potentiostat.
Conductivity measurements
Electrodes were painted with Ag paste and sintered at 550 °C for 1 h. Electrochemical impedance spectroscopy (EIS) was performed with a Gamry 3000 workstation over 0.1–10⁶ Hz (10 mV amplitude). Data were fitted using Zsimwin software.
Results and discussion
Crystalline structure analysis
Figure 1a shows the XRD patterns of pure SCDC, LSCF and their reference PDF cards. The SCDC pattern matches the cubic fluorite structure (JCPDS 75‑0158) and exhibits a slight shift to lower 2θ compared to CeO₂ (JCPDS 34‑0394), confirming successful Sm and Ca incorporation and lattice expansion. LSCF displays the expected perovskite peaks (22.939°, 32.665°, 40.291°, 46.867°, 52.799°, 58.296°, 68.446°, 73.243°, 77.923°, 82.522°, 87.073°). XRD of the sintered composites (Fig. 1b) reveals that peak intensities decrease with increasing sintering temperature, likely due to partial Sr‑O segregation. Minor peak shifts toward lower angles indicate lattice expansion from interfacial solubility, whereas the 900 °C sample shows a slight high‑angle shift attributed to Sr/Co precipitation, consistent with literature [19]. Importantly, no new phases appear even at 1000 °C, confirming chemical stability of the composite up to the highest sintering temperature.
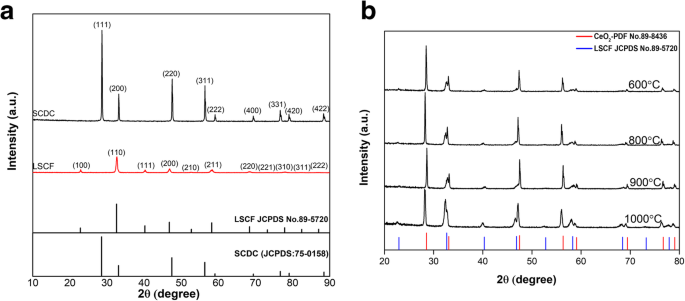
a XRD patterns of SCDC and LSCF with reference PDFs. b XRD patterns of LSCF‑SCDC composites sintered at 600–1000 °C.
Morphological characterization
Cross‑sectional SEM images (Fig. 2) reveal that the 600 °C pellet consists of well‑necked particles spanning nanometers to micrometers. As the sintering temperature rises to 800 and 900 °C, moderate agglomeration appears. At 1000 °C, extensive grain growth and particle coalescence reduce the specific surface area, yielding a dense, bulk‑like microstructure. The pellet thickness diminishes from 1.294 mm (600 °C) to 1.067 mm (1000 °C), reflecting typical sintering shrinkage. Despite the pronounced densification, EDS mapping (Fig. 3a) confirms uniform distribution of all constituent elements (Ca, Sm, Ce, La, Sr, Co, Fe) even at 1000 °C, indicating that the composite remains chemically homogeneous. The interface between the electrolyte and NCAL electrode (Fig. 3b) shows intimate contact with no discernible gaps, suggesting effective pre‑heating bonding.

Cross‑sectional SEM of LSCF‑SCDC pellets sintered at 600 °C (a,b), 800 °C (c,d), 900 °C (e,f) and 1000 °C (g,h).
Fuel cell performances
Cells assembled from the 600 °C pellet achieved a peak power density of 543 mW cm–2 and an open‑circuit voltage (OCV) above 1 V at 550 °C (Fig. 4a). The high OCV indicates minimal gas crossover and effective densification. In contrast, the 800 and 900 °C pellets show modest performance drops while retaining ~1 V OCV, whereas the 1000 °C pellet suffers a severe decline (Pmax = 106 mW cm–2, OCV ≈ 0.7 V). These trends are attributable to the loss of interfacial area between SCDC and LSCF at high sintering temperatures, which disrupts fast oxygen‑ion pathways. The 550 °C cell delivers 245 mW cm–2 at 550 °C, underscoring that a thin, porous electrolyte can still perform well if interfacial integrity is maintained.
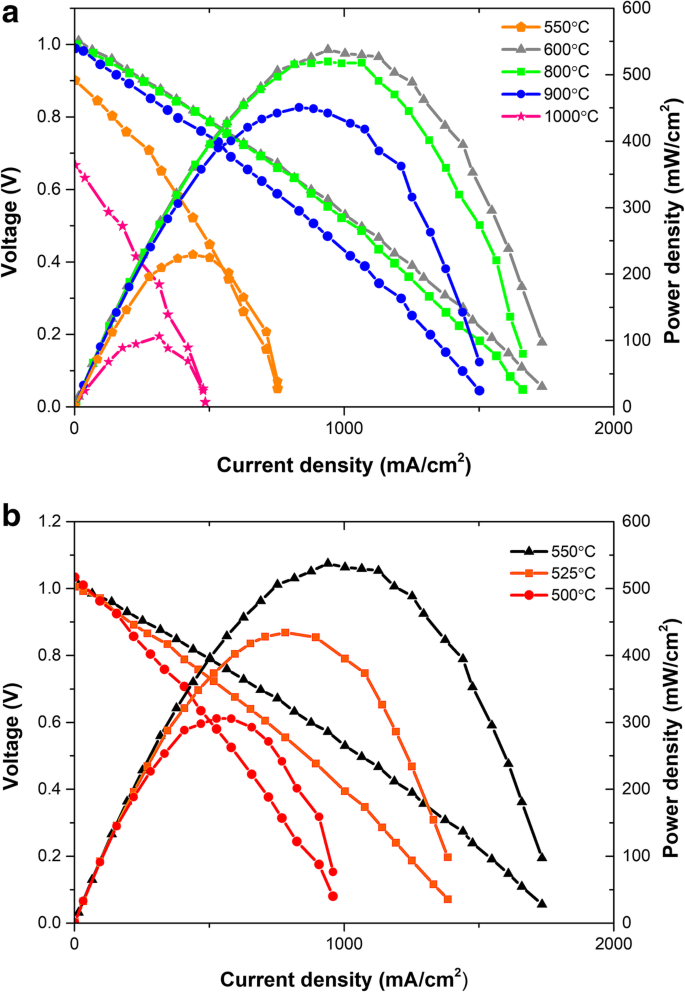
Voltage‑current density and power density characteristics for cells made from pellets sintered at 600, 800, 900 and 1000 °C (a). Power density at 500–550 °C for the 600 °C pellet (b).
EIS characterization
EIS spectra recorded at 550 °C (Fig. 5) consist of a high‑frequency arc (ohmic resistance R1) followed by a depressed low‑frequency arc (R2Q2) and a tail (R3Q3). Fitting with an equivalent circuit R1(R2Q2)(R3Q3) yields R1 values increasing from 0.111 Ω cm² (600 °C) to 0.217 Ω cm² (1000 °C), reflecting enhanced grain‑boundary resistance due to particle coalescence. R2 and R3 are associated with electrode‑electrolyte interfacial processes and gas diffusion, respectively. The polarization resistance rises sharply to 1.22 Ω cm² for the 1000 °C pellet, corroborating the loss of interfacial pathways.
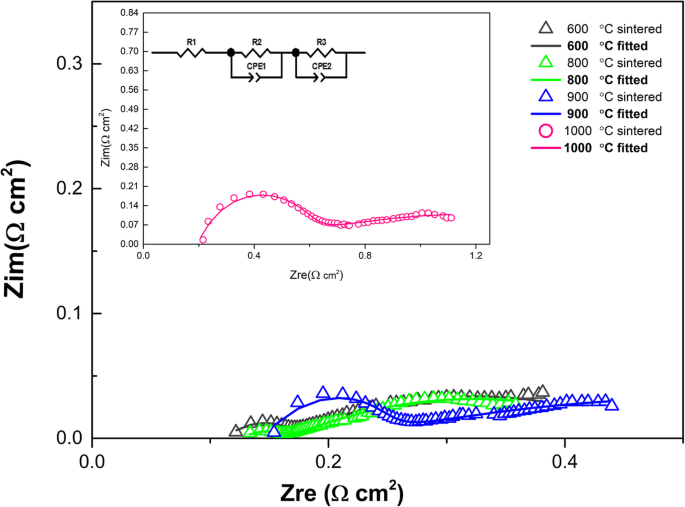
EIS spectra of LSCF‑SCDC cells sintered at 600–1000 °C in H₂/air at 550 °C (primary plots) and fitted curves (secondary plots).
Electrical conductivity
Bulk (Rb) and grain‑boundary (Rgb) resistances were extracted from EIS data (450–650 °C). Conductivity (σ = L/(R S)) follows a linear Arrhenius relationship for both 600 °C and 1000 °C pellets (Fig. 6a). The 600 °C pellet achieves 0.604 S cm–1 at 650 °C, whereas the 1000 °C pellet reaches only 0.346 S cm–1, illustrating the detrimental effect of excessive sintering on ionic pathways. Activation energies are similar (6.07 kJ mol–1 vs. 6.21 kJ mol–1), indicating that the conduction mechanism remains unchanged; the difference lies in interfacial density. Grain‑boundary conductivity (σgb) (Fig. 6b) mirrors the bulk trend, confirming that interfacial conduction is the key contributor to the overall ionic transport.
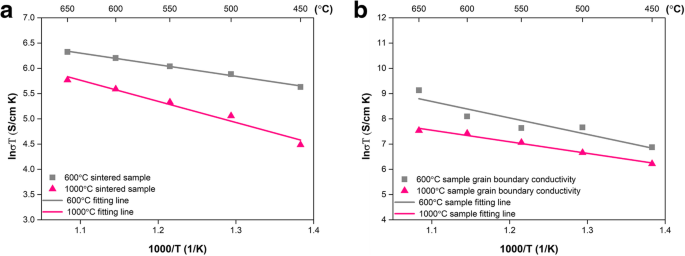
Arrhenius plots for (a) total conductivity and (b) grain‑boundary conductivity of pellets sintered at 600 °C vs. 1000 °C.
Conclusion
We have shown that sintering LSCF‑SCDC composites at 600 °C yields an electrolyte with optimal microstructure and interfacial density, delivering 543 mW cm–2 at 550 °C and 312 mW cm–2 at 500 °C. Raising the sintering temperature to 1000 °C degrades performance to 106 mW cm–2 and 0.7 V OCV due to excessive grain growth, loss of interfacial area and increased grain‑boundary resistance. The study demonstrates that the interfacial conduction mechanism—rooted in space‑charge layers and lattice misfit—is the primary driver of ionic conductivity in SIM composites. Careful control of sintering temperature is therefore essential to preserve the high‑performance interfacial pathways in EFFC electrolytes.
Abbreviations
- CPE:
Constant phase element
- EDS:
Energy‑dispersive spectrometer
- EFFC:
Electrolyte‑free fuel cell
- EIS:
Electrochemical impedance spectroscopy
- LSCF:
La0.6Sr0.4Co0.2Fe0.8O3‑δ
- LT‑SOFC:
Low‑temperature solid‑oxide fuel cell
- NCAL:
Ni0.8Co0.15Al0.05LiO2‑δ
- OCV:
Open‑circuit voltage
- Pmax:
Peak power density
- Rb:
Bulk resistance
- Rgb:
Grain‑boundary resistance
- SCDC:
Ce0.8Sm0.05Ca0.15O2‑δ
- SDC:
Samarium‑doped ceria
- SEM:
Scanning electron microscopy
- SIM:
Semiconductor‑ionic material
- XRD:
X‑ray diffraction
- YSZ:
Yttrium‑stabilized zirconia
Nanomaterials
- Impact of a Trace SiO₂ Addition on the Early‑Stage Sintering of 3Y‑TZP Nanopowders
- Effect of Contact Non‑Equilibrium Plasma on Mn_xFe_3−xO_4 Spinel Nanoparticles: Structural and Magnetic Insights
- Binder Selection for Copper Oxide Anodes: Why SBR+CMC and LA133 Outperform PVDF in Lithium‑Ion Batteries
- Comparative Analysis of Electrochemical, Biomedical, and Thermal Properties of Natural vs. Synthetic Nanomaterials
- PVP‑Enhanced SnO₂ Nanoflowers Deliver Ultra‑Fast, Highly Selective H₂S Sensing
- Thermal Annealing Enhances Emission in InP/ZnS Quantum Dot Films: Photoluminescence Insights
- How Temperature Influences the Young’s Modulus of Electrospun Polyurethane Nanofibers
- High‑Conductivity Nano‑Silver Ink Achieves Low‑Temperature Sintering for Paper‑Based Electronics
- Crystal Structure Drives Supercapacitive Performance of Hydrogenated TiO₂ Nanotube Arrays
- How Inclusions Influence Steel Performance and Strength



