Comparative Analysis of Electrochemical, Biomedical, and Thermal Properties of Natural vs. Synthetic Nanomaterials
Abstract
In this study, we synthesized and evaluated natural nanomaterials—cellulose nanocrystal (CNC) and nanofiber cellulose (NFC)—and synthetic carbon-based nanoparticles—carbon nanofiber (CNF) and carbon nanotube (CNT). By systematically varying structure, morphology, and surface area, we compared their electrochemical, biomedical, and thermal performance. Natural nanomaterials exhibited negligible cytotoxicity at concentrations of 12.5 µg mL⁻¹ (NFC) and 3.125 µg mL⁻¹ (CNC). In contrast, synthetic nanomaterials, with surface areas of 15.3–21.1 m² g⁻¹ and thermal stability between 480 °C and 600 °C, significantly enhanced electrode output through higher surface area and reduced current‑flow resistance.
Background
Natural nanomaterials, particularly nanocellulose in forms such as CNC and NFC, are derived from hydrogen‑bonded cellulose chains that form microfibrils. When these microfibrils are reduced to the nanometer scale, the resulting nanocellulose boasts high mechanical strength, low thermal expansion, large surface area, renewability, optical transparency, biodegradability, and minimal toxicity.
Kenaf, a tropical plant with 44–63.5 % cellulose, serves as an abundant source for nanocellulose production. CNC is obtained via acid hydrolysis, whereas NFC results from mechanical defibrillation. Their favorable properties have enabled applications ranging from polymer composites and transparent barrier films to photonic crystals, shape‑memory polymers, drug carriers, and advanced composites.
Carbon nanomaterials, including CNT and CNF, are constructed from sp²‑hybridized carbon atoms in one‑dimensional architectures. Their properties depend on atomic arrangement, diameter, length, and morphology. CNFs feature cylindrical structures with stacked graphene layers, while CNTs form tubular lattices. These materials, produced through arc‑discharge, laser ablation, chemical vapor deposition (CVD), or self‑assembly, exhibit exceptional mechanical, thermal, electrical, and electrochemical performance, and are increasingly explored in biomedical applications.
To date, no comprehensive comparative study has examined the structure‑property relationships of natural and synthetic nanomaterials. This work addresses that gap by characterizing CNC, NFC, CNF, and CNT in terms of crystallinity, surface area, thermal stability, cytotoxicity, and electrochemical behavior using SEM, TEM, XRD, BET, TGA, CV, and MTT assays.
Results and Discussion
Morphology of Nanomaterials
SEM and TEM images (Fig. 1) reveal distinct morphologies: CNCs are needle‑like (≈150 nm long, 12 nm diameter); NFCs form highly entangled, web‑like fibers (50–200 nm diameter); CNFs display rod‑shaped particles (150–200 nm diameter) with coarse surfaces; CNTs are multi‑walled, curly, and entangled with wall thicknesses of 10–30 nm.

SEM/TEM images of a CNC, b NFC, c CNF, and d CNT
Energy Dispersive X‑ray Spectroscopy
EDX analysis confirms that all nanoparticles consist mainly of carbon and oxygen, with trace sulfur in CNC and residual nickel (~0.5 wt %) in CNF and CNT due to the CVD catalyst. No significant contaminants were detected in NFC.
BET Surface Area
BET measurements show synthetic nanomaterials possess higher surface areas than natural counterparts: CNT (≈21 m² g⁻¹) > CNF (≈15 m² g⁻¹) > NFC (≈4 m² g⁻¹) > CNC (≈3 m² g⁻¹). The greater surface area in CNT is attributed to its networked, high‑aspect‑ratio structure.
X‑ray Diffraction
XRD patterns (Fig. 2) exhibit sharp crystalline peaks for CNC (2θ ≈ 22.5°) indicating higher crystallinity than NFC. For carbon nanostructures, prominent peaks at 2θ ≈ 25.5° (C(002)) and 43° (C(100)) confirm graphitic ordering, especially in CNTs.
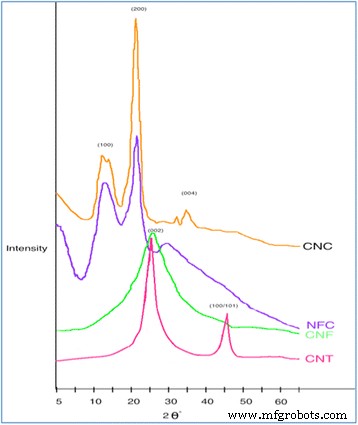
XRD pattern of nanomaterials
Thermal Resistance
TGA curves (Fig. 3) reveal that CNC undergoes two degradation steps at ≈180 °C and 300 °C, whereas NFC degrades at a single peak near 300 °C. CNF decomposes between 480 °C and 615 °C, and CNT exhibits even higher thermal stability, decomposing between 600 °C and 690 °C. The superior stability of CNT is linked to its robust graphitic structure and higher surface area.
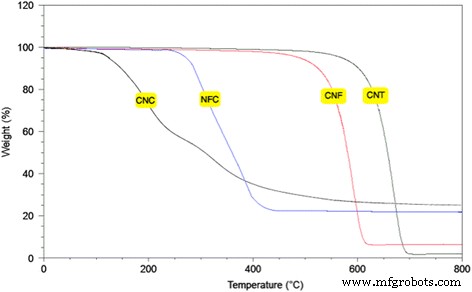
TGA curves of CNC, NFC, CNF, and CNT
Electrochemical Results
Cyclic voltammograms (Fig. 4) demonstrate that synthetic nanomaterials (CNF, CNT) yield larger electrochemical double‑layer capacitance and pseudocapacitive behavior compared to natural counterparts, attributable to their higher porosity and surface area. A plateau between −0.5 V and 0.5 V indicates SEI formation on CNC and NFC electrodes.
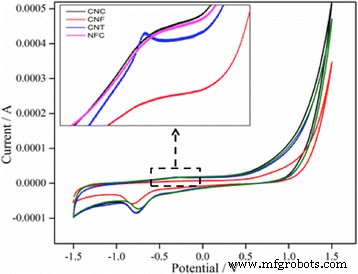
Cyclic voltammograms of CNC, CNF, CNT, and NFC in PBS (pH 7.0) at 0.1 V s⁻¹
Cytotoxicity Analysis
MTT assays on 4T1 breast cancer cells reveal that natural nanomaterials exhibit minimal toxicity: NFC remains 100 % viable at 12.5 µg mL⁻¹, and CNC reduces viability by only 7 % at 100 µg mL⁻¹. Synthetic nanomaterials, however, display higher cytotoxicity—CNF kills 34 % and CNT 28 % of cells at 100 µg mL⁻¹. At lower concentrations (12.5 µg mL⁻¹), CNF and CNT still reduce viability to 4.3 % and 1.7 % respectively, whereas NFC and CNC remain largely non‑toxic.
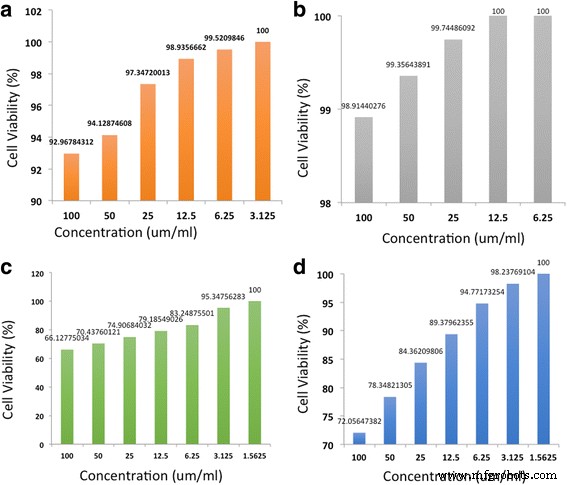
Cytotoxicity of a CNC, b NFC, c CNF, and d CNT
Conclusions
We successfully synthesized natural nanomaterials (CNC, NFC) via acid hydrolysis and mechanical defibrillation, and synthetic nanomaterials (CNF, CNT) via CVD. Structural analyses confirmed crystalline CNC and highly graphitized CNT, while SEM/TEM revealed smaller diameters for CNC and NFC compared to CNF and CNT. BET analysis showed that synthetic nanomaterials possess substantially higher surface areas, correlating with superior electrochemical performance and thermal stability. Although synthetic nanoparticles outperform natural ones in electrochemical and thermal metrics, natural nanomaterials demonstrate lower cytotoxicity, making them preferable for biomedical applications.
Methods
All experimental protocols were designed for reproducibility and transparency. Detailed synthesis, characterization, and testing procedures are described below.
Preparation of Natural Nanofibers
Cellulose extraction from kenaf bast fibers followed Kargarzadeh et al. (2012). CNC was produced by 65 % H₂SO₄ hydrolysis at 50 °C for 40 min, followed by centrifugation, dialysis, and ultrasonic dispersion. NFC was obtained by cooking ret‑ed fibers in 25 % NaOH with anthraquinone at 160 °C for 2 h, then washing and drying.
Preparation of Synthetic Nanofibers
Ni(NO₃)₂·6H₂O served as the catalyst precursor. After dehydration at 160 °C and calcination to 400 °C, Ni particles formed. CNF and CNT were synthesized at 650 °C and 800 °C respectively via acetylene decomposition (50 sccm) with H₂/N₂ (100/100 sccm) for 30 min. Residual Ni was removed by FeCl₃/HCl etch, followed by washing and drying.
Characterization
- SEM, TEM, EDX for morphology and composition.
- XRD for crystalline structure.
- BELSORP‑mini II for BET surface area.
- PGSTAT204 for cyclic voltammetry (CV) at 0.1 V s⁻¹ in PBS.
- MTT assay for cytotoxicity on 4T1 cells (IC₅₀ determination).
- TGA (Mettle Stare SW 9.10) for thermal stability (200–900 °C, 10 °C min⁻¹).
Abbreviations
- CNC
- Cellulose nanocrystal
- NFC
- Nanofiber cellulose
- CNF
- Carbon nanofiber
- CNT
- Carbon nanotube
- BET
- Brunauer, Emmett, and Teller
- EDX
- Energy dispersive X‑ray
- XRD
- X‑ray diffraction
- TGA
- Thermogravimetric analysis
- CV
- Cyclic voltammograms
- SEM
- Scanning electron microscopy
- TEM
- Transmission electron microscopy
- MTT
- 3‑[4,5‑dimethylthiazol‑2‑yl]-2,5‑diphenyltetrazolium bromide
- SEI
- Solid electrolyte interface
Nanomaterials
- Why Resin and Fiber Composites Deliver Superior Strength, Lightness, and Performance
- Fluorescent Nanomaterials: Advances, Synthesis, and Biomedical Applications
- Comparative Analysis of Carbon Nanofiber and Activated Carbon Symmetric Supercapacitors in Aqueous Electrolyte
- Structural, Electronic, and Magnetic Characterization of Ag<sub>n</sub>V (n = 1–12) Clusters via DFT and CALYPSO Search
- Enhancing CL‑20 Safety: Lowering Impact Sensitivity via Carbon Nanomaterial‑Assisted Thermal Conductivity
- Nanomaterials and Nanotechnologies in Wastewater Treatment: Insights from a Bibliometric Analysis (1997‑2016)
- Antimicrobial Performance of PBAT Nanocomposites and Composites Incorporating Copper, Cu/Cu₂O Nanoparticles, and Copper Sulfate
- Efficient Oxidation and Nano‑Dispersion of Bombyx mori and Antheraea pernyi Silk Fibers for High‑Performance Membranes
- Carbon Nanomaterials for Treating Heavy‑Metal‑Contaminated Water and Advancing Environmental Remediation
- Comparative Toxicity of PEG-Coated Cobalt Ferrite Nanoparticles and Nanospheres



