Comparative Analysis of Carbon Nanofiber and Activated Carbon Symmetric Supercapacitors in Aqueous Electrolyte
Abstract
Symmetric supercapacitors were fabricated from carbon nanofibers (CNF) and activated carbon (AC) using a 7 wt % polyvinylidene fluoride (PVDF) binder in an aqueous electrolyte. This study compares the pore architecture and electrochemical performance of CNF‑ and AC‑based symmetric cells assembled without a current collector. The BET surface areas are 83 m² g⁻¹ for CNF and 1042 m² g⁻¹ for AC, reflecting a mesoporous skeleton in CNF and a predominantly microporous network in AC. AC delivers a high specific capacitance of 334 F g⁻¹ at 5 mV s⁻¹, while CNF reaches 52 F g⁻¹ at the same scan rate; both retain excellent performance even at 500 mV s⁻¹. CNF shows superior conductivity, with an ESR of only 0.28 Ω versus 3.72 Ω for AC, and delivers a peak specific power of 1860 W kg⁻¹ compared to 450 W kg⁻¹ for AC. Conversely, AC achieves a higher specific energy of 18.1 Wh kg⁻¹ versus 2 Wh kg⁻¹ for CNF, highlighting its suitability for energy‑dense applications while CNF excels in high‑power scenarios. Both materials maintain >95 % capacitance after 2500 charge‑discharge cycles, demonstrating robust cycling stability.
Background
Supercapacitors have emerged as pivotal energy‑storage devices due to their high power density and long cycling life, making them ideal for electric vehicles, portable electronics, and power‑tool applications [1]. The choice of electrode material determines whether the device prioritizes power or energy: high‑conductivity materials favor power, whereas high‑surface‑area materials favor energy [2]. Electrode performance hinges on material composition, thickness, surface area, pore size distribution, and surface functional groups [3]. Carbon materials dominate the field because of their cost‑effectiveness, morphological versatility, and chemical/thermal stability [4‑7]. Carbon nanofibers (CNFs) offer a unique tubular morphology that combines low resistivity with high porosity, while activated carbon (AC) provides exceptional porosity, abundance, and cycling stability [8‑9].
Electrode fabrication typically incorporates binders such as PVDF, PTFE, or PVDC in 5–10 wt % proportions to maintain structural integrity [10‑13]. However, binders can occlude porosity and raise electrical resistivity, reducing capacitance. Electrolyte selection also influences performance; aqueous electrolytes enable a stable potential window of up to 1.0 V and allow small ions to access both micro- and mesopores, whereas organic electrolytes extend the window to ~2.7 V but are less environmentally benign [14]. The electric double layer (EDL) capacitance is proportional to accessible surface area and inversely proportional to ion‑pore distance, guiding two primary strategies for enhancement: increasing surface area and reducing ion‑pore separation [15].
This study offers a head‑to‑head comparison of AC and CNF symmetric supercapacitors, employing identical PVDF binder content (7 wt %) to isolate material effects.
Methods/Experimental
Preparation of AC and CNF Electrodes
Symmetric cells were fabricated from AC (Carbopal CCP80, Donau Carbon) and CNFs (helicoidally graphitic, 20–80 nm diameter, >30 µm length, 6 % Ni, 10⁻² Ω cm resistivity). Both materials were mixed with 7 wt % PVDF binder in 15 mL acetone using a planetary ball mill (Pulverisette 7) at 500 rpm for 30 min. The slurry was stirred mechanically for 60 min, sonicated for 30 min, and dried at 70 °C for 60 min. Discs (10 mm diameter) were pressed at 10 tons. Final electrode masses were 0.018 g (CNF) and 0.020 g (AC).
Surface Characterization
Nitrogen adsorption at 77 K (Micromeritics TriStar 3000) quantified BET surface area and pore size distribution. Samples were outgassed at 100 °C for 4 h. BET analysis was performed over 0.02–0.2 P/P₀; total pore volume was taken at P/P₀≈0.9932. Micropore parameters were derived via the t‑plot method, while BJH and MP methods provided meso‑ and macro‑pore data for CNF and AC, respectively.
Morphological Characterization
Scanning electron microscopy (SEM) and transmission electron microscopy (TEM, Philips Tecnai G2 F20, 300 kV) assessed morphology. Samples were dispersed in ethanol, sonicated for 15 min, and deposited on carbon‑coated copper grids.
Electrochemical Characterization
Electrochemical performance was evaluated in two‑electrode Swagelok cells using a Gamry 600 potentiostat and 6 M KOH electrolyte. Cyclic voltammetry (CV), galvanostatic charge‑discharge (GCD), and electrochemical impedance spectroscopy (EIS) were employed.
Results and Discussion
Morphological Characterization
SEM and TEM images (Fig. 1) confirm effective binder integration and distinct morphologies: CNFs appear as cylindrical tubes, whereas AC shows interconnected spherical particles. Both exhibit smooth surfaces and homogeneous particle distribution.

SEM and TEM images (inset) for a CNFs and b AC
Pore Texture of CNFs and AC
N₂ adsorption/desorption isotherms (Fig. 2) reveal distinct pore architectures. CNFs exhibit a type II isotherm with a small hysteresis loop, indicative of mesoporosity; pore distribution: 59 % mesopores (2–50 nm), 18 % micropores (0.5–2 nm), 23 % macropores (>50 nm). AC shows a type I isotherm dominated by micropores: 55 % ultramicropores (0.2–0.5 nm), 33 % supermicropores (0.5–2 nm), 12 % mesopores. BET surface areas are 83 m² g⁻¹ (CNF) and 1042 m² g⁻¹ (AC). Pore size distributions (Fig. 3) further confirm CNFs’ mesopores centered at ~3.4 and ~7.1 nm, while AC’s pores cluster around 0.47 nm.
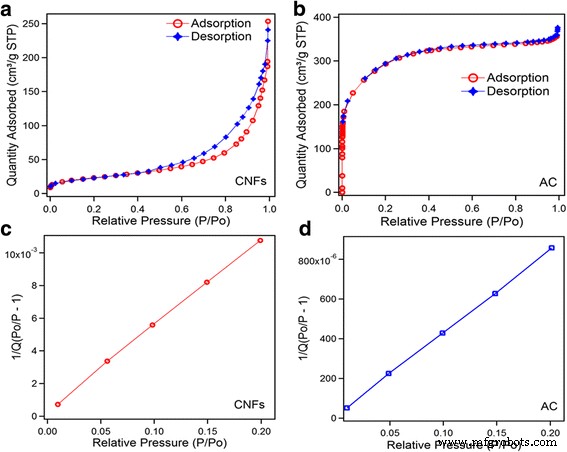
Nitrogen adsorption/desorption isotherms, a CNFs and b AC. BET surface area c CNFs and d AC
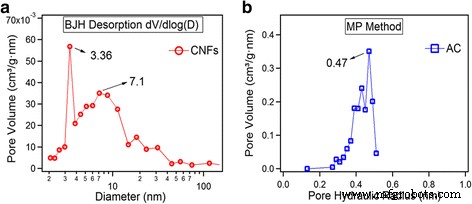
Pore size distribution. a CNFs by BJH method. b AC by MP method
Electrochemical Behavior of CNFs and AC
CV curves (Fig. 4) show near‑rectangular shapes for both materials at low scan rates, confirming double‑layer behavior. AC produces higher currents and retains performance up to 500 mV s⁻¹, whereas CNF’s capacitance drops from 52 to 32 F g⁻¹ over the same range. GCD profiles (Fig. 5) are symmetric, with CNF exhibiting a minimal IR drop (0.28 Ω) versus AC (3.72 Ω). Specific capacitance from GCD: CNF—23.8 F g⁻¹ (0.23 A g⁻¹) to 19 F g⁻¹ (2 A g⁻¹); AC—159 F g⁻¹ (0.5 A g⁻¹) to 139 F g⁻¹ (2.5 A g⁻¹).
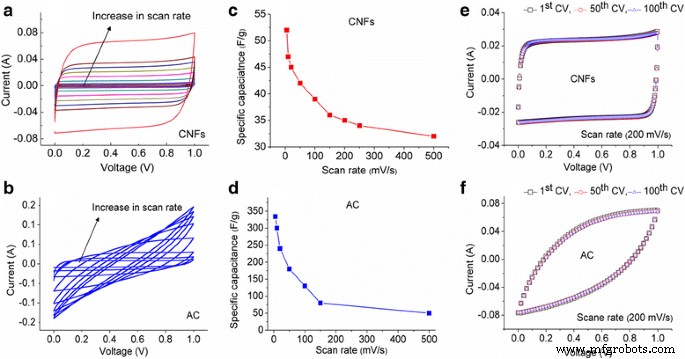
a, b CVs of CNFs and AC, respectively, at 5, 10, 20, 50, 100, 150, and 500 mV s⁻¹ scan rates; c, d specific capacitance comparison at different scan rates; e, f CVs of CNFs and AC, respectively, from 1st to 100th cycle
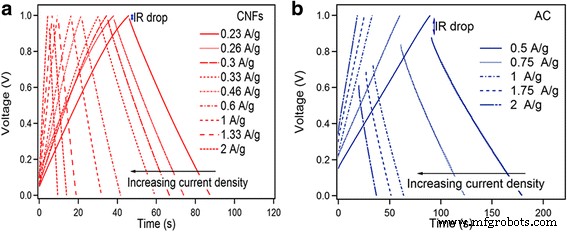
a GCD curves at different current densities of CNFs. b GCD curves at different current densities of AC
Ragone analysis (Fig. 6b) shows CNF achieving a peak specific power of 1860 W kg⁻¹ at 2 Wh kg⁻¹, while AC reaches 1650 W kg⁻¹ at 5.5 Wh kg⁻¹. Energy densities: CNF—2.3 Wh kg⁻¹; AC—18.1 Wh kg⁻¹. Both electrodes retain >95 % capacitance after 2500 cycles (Fig. 6c).
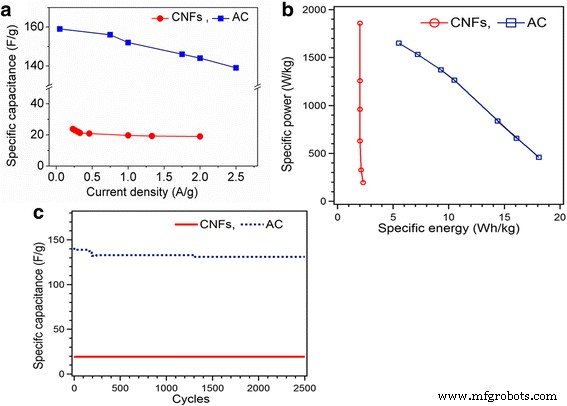
a Specific capacitance comparison from discharge curve of GCD. b Ragone plot of specific power against specific energy for CNFs and AC. c Cycling stability of CNFs and AC
EIS (Fig. 7a) confirms the low ESR of CNF (0.28 Ω) versus AC (3.72 Ω). CNF also displays a steeper low‑frequency slope, indicating superior capacitive behavior. Specific capacitance derived from impedance (Fig. 7b) aligns with CV values: 36 F g⁻¹ (CNF) and 284 F g⁻¹ (AC) at 0.1 Hz.
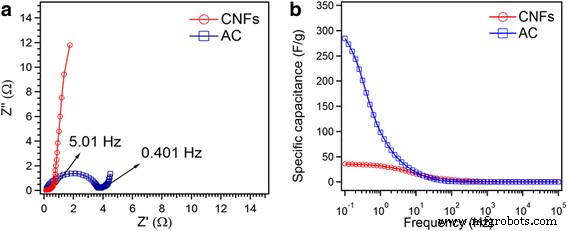
a Nyquist plot of CNFs and AC. b Csp comparison calculated from EIS.
Time‑constant analysis (Fig. 8) shows AC’s τ₀ at 5 kHz and CNF’s τ₀ at 2 Hz, indicating faster relaxation for AC. Phase‑shift plots (Fig. 8c) reveal near‑ideal capacitive behavior at low frequencies for both materials.
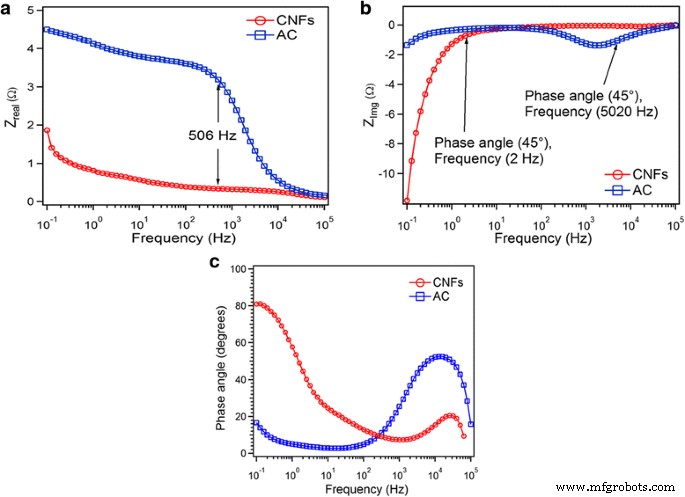
a The real and b imaginary parts are plotted as a function of log of frequency and c phase shift as function of frequency for AC and CNFs
Overall, AC’s high BET surface area and ultramicroporous network yield superior specific capacitance and energy density, while CNF’s lower ESR and mesoporous structure afford higher power density.
Conclusions
Both CNF and AC electrodes were fabricated under identical conditions and evaluated as symmetric supercapacitors in aqueous electrolyte. AC’s 1042 m² g⁻¹ BET area, dominated by ultramicropores, delivers a specific capacitance of 334 F g⁻¹ and a specific energy of 18.1 Wh kg⁻¹. CNF’s 83 m² g⁻¹ surface area and mesoporous skeleton provide a specific capacitance of 52 F g⁻¹, a specific power of 1860 W kg⁻¹, and an ESR of 0.28 Ω. Consequently, AC is ideal for energy‑dense applications, whereas CNF excels in high‑power scenarios. Both materials exhibit excellent cycling stability (>95 % retention after 2500 cycles).
Nanomaterials
- Scientists Successfully Synthesize and Visualize Cyclo[18]Carbon
- Carbon Nanotube Yarns, Artificial Muscles, and Transparent Sheets: Advanced Applications
- Nanofiber & Filament-Based Nanocarriers: Advancing Precision Drug Delivery
- Comprehensive Atomic & Electronic Analysis of Stacked-Cup Carbon Nanofibers
- Graphene‑Polymer Composites for High‑Performance Supercapacitors: A Comprehensive Review
- Comparative Analysis of Electrochemical, Biomedical, and Thermal Properties of Natural vs. Synthetic Nanomaterials
- Enhanced Supercapacitor Performance via Polyaniline‑Coated Nitrogen‑Doped Ordered Mesoporous Carbon Composites
- Advances in Carbon Nanotube Assembly and Integration for Next‑Generation Applications
- Comparative Toxicity of PEG-Coated Cobalt Ferrite Nanoparticles and Nanospheres
- Economical Synthesis and Comparative Catalytic & Antimicrobial Performance of Nb and Ta Selenide/ Telluride Nanocomposites



