Graphene‑Polymer Composites for High‑Performance Supercapacitors: A Comprehensive Review
Abstract
Supercapacitors combine ultrahigh capacitance, rapid charge‑discharge, and exceptional cycling life, making them ideal for next‑generation energy storage. Materials that offer high specific surface area, robust mechanical and chemical stability, and low cost are essential. Graphene—a single layer of carbon atoms—has emerged as a standout due to its extraordinary conductivity, flexibility, and theoretical surface area of 2630 m² g⁻¹. When blended with polymers, graphene can deliver synergistic properties that enhance energy density, structural integrity, and flexibility. This review surveys the latest polymer–graphene composites for supercapacitor electrodes, examines binder strategies, explores conductive‑polymer hybrids, and highlights flexible device architectures. We conclude with key challenges and future directions for commercial deployment.
Introduction
Since its isolation in 2004, graphene has captivated researchers across physics, chemistry, and engineering. Its remarkable electrical conductivity, mechanical strength, and large specific surface area make it an attractive candidate for energy‑storage electrodes. While activated carbon remains the industrial workhorse—primarily because of its low cost and mature manufacturing—graphene’s theoretical surface area far exceeds that of conventional carbon materials. However, the high‑purity, CVD‑grown graphene that offers the best electronic properties is prohibitively expensive and difficult to scale. Consequently, most supercapacitor research now focuses on graphene derivatives (e.g., graphene oxide, reduced graphene oxide, and nanoplatelets) produced via scalable chemical or mechanical exfoliation. These materials, though defect‑rich, present higher electrochemical capacitance and are more cost‑effective for practical applications.
Review
Graphene and Polymer Binders
Effective electrode fabrication requires that graphene nanosheets adhere strongly to the current collector while preserving electrical conductivity. Fluoropolymer binders—most notably polyvinylidene fluoride (PVDF) and polytetrafluoroethylene (PTFE)—are the industry standards due to their chemical inertness and mechanical robustness.
Graphene and PVDF Supercapacitors
PVDF, a thermoplastic fluoropolymer, offers high thermal stability and aging resistance. In graphene‑based electrodes, a typical formulation contains 10–20 wt% PVDF mixed with graphene nanoplatelets, followed by dispersion in N‑methyl‑2‑pyrrolidone (NMP) or dimethylformamide (DMF) to achieve a slurry suitable for coating by doctor blade, bar, or drop‑casting. The binder fraction must be carefully balanced: insufficient PVDF leads to poor adhesion and electrode delamination, whereas excess PVDF reduces electronic conductivity and, consequently, energy density. Incorporating sub‑50 nm conductive carbon additives (e.g., carbon black) mitigates these effects by providing conductive pathways and suppressing graphene aggregation. Reported results include a 175 F g⁻¹ specific capacitance with only 9.1 % loss after 6000 cycles in a reduced‑graphene/PVDF/CB composite.
Vacuum filtration onto porous nickel foam offers an alternative fabrication route. By adjusting vacuum pressure and dwell time, the graphene loading can be precisely tuned. A 25 wt% PVDF binder on graphene nanoplatelets coated onto 95 % porous nickel foam achieved 152 F g⁻¹ and retained 95 % of its capacitance after 2000 cycles.
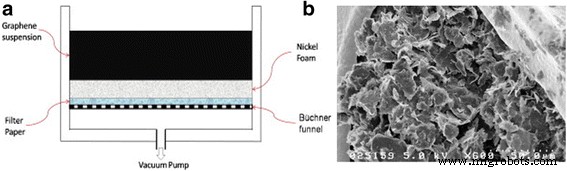
a Schematics of vacuum filtration method to build graphene‑containing supercapacitor electrode. b SEM image of graphene/PVDF on the nickel foam formed by vacuum filtration. Reprinted with permission from [10]. Copyright 2012 Elsevier
Graphene and PTFE Supercapacitors
PTFE, another fluoropolymer, offers superior water dispersibility (e.g., 60 wt% PTFE in H₂O) and eliminates the need for hazardous organic solvents. Its hydrophobic nature, however, can impede ion transport if used in excess. Studies show that 5–10 wt% PTFE maximizes specific capacitance for activated‑carbon electrodes, while lower binder levels compromise mechanical adhesion and long‑term stability. Adding a small amount of polyvinylpyrrolidone (PVP, ~3 wt%) dramatically improves wettability, reducing the contact angle from 151° to 22°, thereby facilitating electrolyte penetration.
Electrolyte selection further influences performance. A graphene/PTFE electrode demonstrated 116 F g⁻¹ in 3 M KOH, surpassing 100 F g⁻¹ in TEABF₄/AN and 95 F g⁻¹ in TEABF₄/PC, with corresponding variations in equivalent series resistance (ESR).
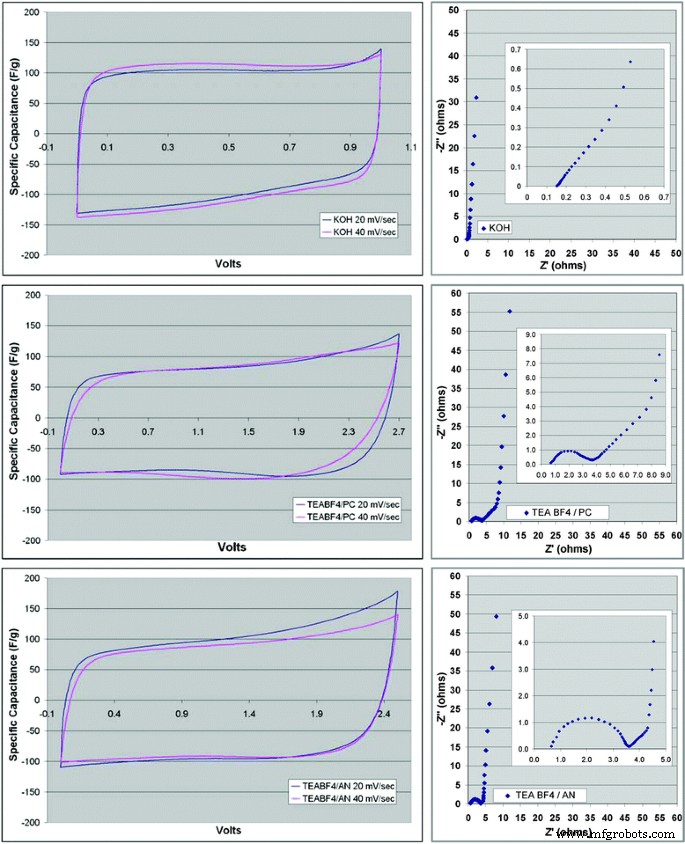
CV (left) and Nyquist (right) plots of CMG material with KOH, TEABF₄/PC, and TEABF₄/AN electrolytes. Reprinted with permission from [24]. Copyright 2008 American Chemical Society
Graphene and Conducting Polymers Composites
While polymer binders provide mechanical integrity, they are typically insulating. Conducting polymers (CPs) such as polypyrrole (PPy), polyaniline (PANI), and PEDOT introduce pseudocapacitance through fast redox reactions. However, CPs alone suffer from volume changes during cycling, leading to cracks and capacity fade. Integrating CPs with graphene—either as a binder or as a structural scaffold—alleviates these issues by offering mechanical support, high conductivity pathways, and enhanced surface area.
Graphene and Polyaniline (PANI) Composites
PANI’s high conductivity and redox activity make it a benchmark for pseudocapacitive electrodes. In situ polymerization of aniline on graphene oxide (GO) yields GO/PANI composites whose morphology—and thus electrochemical performance—depends on GO loading. Pure PANI nanofibers achieve 420 F g⁻¹ but lose ~40 % capacitance after five cycles. Increasing GO content lowers the specific capacitance (down to 158 F g⁻¹ at 80 % GO) but significantly improves cycling stability by buffering volumetric changes.
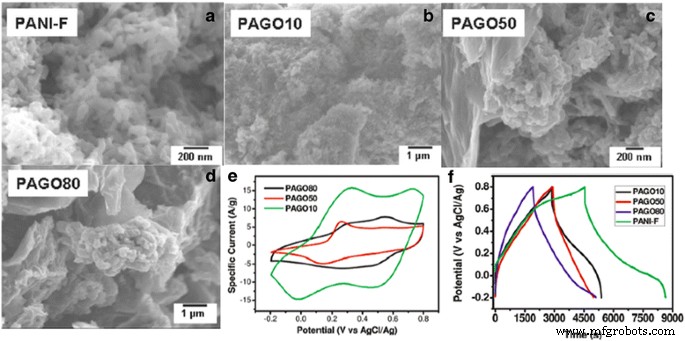
a–d SEM images of PANI and GO/PANI composites at 10 %, 50 %, and 80 % GO. e CVs in 2 M H₂SO₄ at 100 mV s⁻¹. f Charge–discharge cycling at 0.1 A g⁻¹. Reprinted with permission from [41]. Copyright 2010 American Chemical Society
Reduced graphene oxide (RGO) offers superior conductivity. A three‑step in‑situ polymerization–reduction process produced RGO/PANI composites with a peak specific capacitance of 1129 F g⁻¹ and >80 % retention after 1000 cycles.
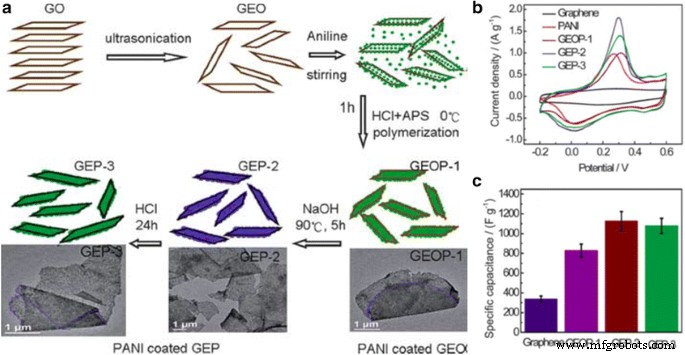
a Schematic of graphene/PANI hybrid synthesis. b–c CV curves and specific capacitance trends for GO, GEOP‑1, GEP‑2, and GEP‑3. Reprinted with permission from [44]. Copyright 2010 Royal Society of Chemistry
Graphene and Polypyrrole (PPy) Composites
PPy films, while conductive, often form dense aggregates that hinder ion transport. In situ polymerization on surface‑modified graphene nanosheets (e.g., Na‑PSS‑treated GO) yields PPy/graphene composites with roughly double the specific capacitance of pure PPy and markedly superior cycling performance.
Graphene and Poly(3,4‑ethylenedioxythiophene) (PEDOT) Composites
PEDOT, especially when doped with poly(styrene sulfonic acid) (PSS), delivers high conductivity (up to 500 S cm⁻¹) and excellent stability (>80 % retention over 70,000 cycles). Incorporating graphene or RGO into PEDOT matrices doubles conductivity and enhances mechanical strength by sixfold, while simultaneously expanding the porous network and boosting specific capacitance to 300 F g⁻¹ in aqueous electrolytes.
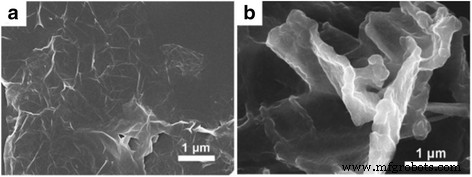
SEM images of (a) pure RGO, (b) RGO/PEDOT composite film. Reprinted with permission from [57]. Copyright 2013 Royal Society of Chemistry
Comparison and Summary of CP‑Graphene Composites
When benchmarked against similar fabrication routes, RGO/PANI, RGO/PPy, and RGO/PEDOT electrodes deliver specific capacitances of 361, 249, and 108 F g⁻¹, respectively, at 0.3 A g⁻¹. RGO/PEDOT exhibits the best long‑term retention (88 % after 1000 cycles), while RGO/PANI and RGO/PPy maintain ~82 % and 81 % respectively. These trends underscore the importance of balancing conductivity, pseudocapacitive contribution, and structural stability.
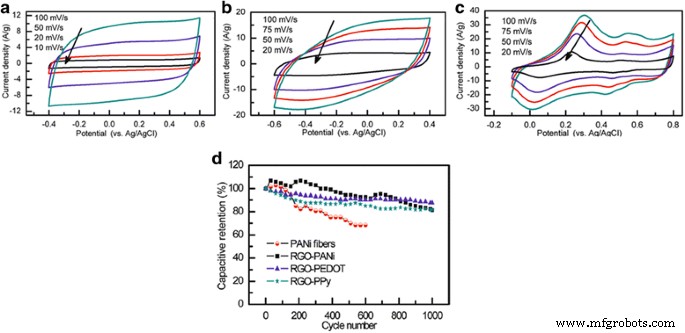
CVs of (a) RGO‑PEDOT, (b) RGO‑PPy, (c) RGO‑PANI; (d) cycling stability of each composite. Reprinted with permission from [42]. Copyright 2012 American Chemical Society
Graphene and Polymer Composites for Flexible Supercapacitors
Flexible energy storage is critical for wearable electronics, foldable displays, and implantable medical devices. Graphene‑based films—whether ultrathin (<100 nm) membranes, paper, foam, or coated textiles—offer the mechanical flexibility required for such applications. However, challenges such as graphene restacking, low intrinsic capacitance (~100 F g⁻¹ for 100‑nm films), and scalable manufacturing remain. A promising strategy is to fabricate graphene/CP composite films that can be integrated with solid‑state electrolytes and thin metal current collectors.
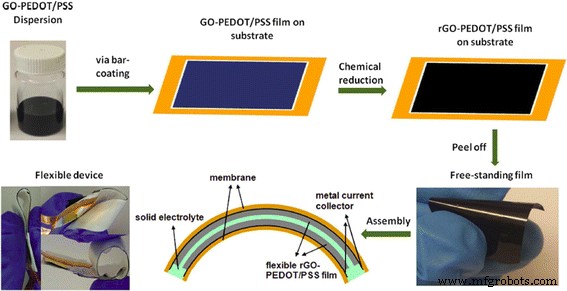
Schematic of rGO‑PEDOT/PSS film fabrication and flexible supercapacitor assembly. Reprinted with permission from [84]. Copyright 2015 Nature Publishing Group
During bending tests, rGO‑PEDOT/PSS electrodes retain 95 % of their capacitance after 10,000 charge–discharge cycles at 180° curvature, and a 15 × 2 cm strip can power an LED for 20 s when fully charged.
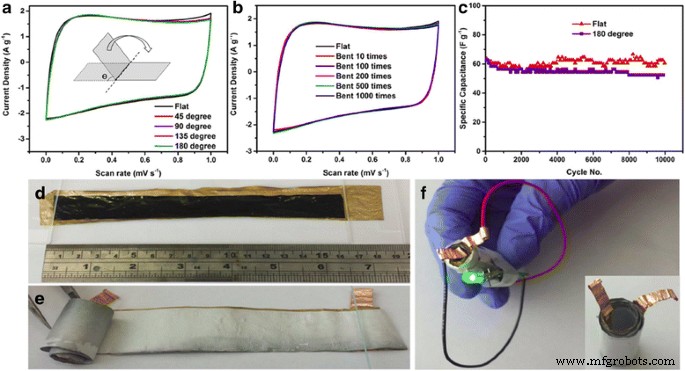
a–b CVs during bending; c long‑term test under flat and 180° bent states; d–f flexible device illustration and LED demonstration. Reprinted with permission from [84]. Copyright 2015 Nature Publishing Group
Conclusions
Graphene’s exceptional electrical, mechanical, and surface‑area attributes have propelled it to the forefront of supercapacitor research. Polymer binders mitigate graphene’s insulating nature, while conductive‑polymer composites leverage graphene’s structural support to enhance both specific capacitance and cycle life. The flexibility of graphene/CP films also paves the way for roll‑to‑roll manufacturing of wearable, conformable energy‑storage devices. Nonetheless, translating laboratory performance to commercial products demands scalable synthesis that preserves nanoscale morphology, rational pore design to eliminate dead volume, and realistic assessment of full‑device parameters—including current‑collector, electrolyte, and separator weights. Continued interdisciplinary efforts will be required to overcome these hurdles and unlock the full commercial potential of graphene‑polymer supercapacitors.
Nanomaterials
- Gold Nanoparticles: Advancing Diagnostic and Therapeutic Applications in Medicine – A Comprehensive Review
- Fluorescent Nanomaterials: Advances, Synthesis, and Biomedical Applications
- Hybrid Graphene/WO₃ and Graphene/CeOx Electrodes for High‑Performance Supercapacitors
- Enhanced Supercapacitor Performance via Polyaniline‑Coated Nitrogen‑Doped Ordered Mesoporous Carbon Composites
- Electrochemical Energy Storage: Enhancing Capacitance with Reduced Graphene Oxide/Carbon Nanotube Composites
- Platinum‑Based Catalysts on Carbon Supports and Conducting Polymers for Direct Methanol Fuel Cells: A Comprehensive Review
- FeF3·0.33H2O Cathode Enhanced by CNTs and Graphene: A High‑Performance Solution for Lithium‑Ion Batteries
- ZnSe/N‑Doped Carbon Composites Derived from ZIF‑8 Deliver High Capacity and Long‑Term Cycling for Lithium‑Ion Batteries
- Controlling Terahertz and Near‑Infrared Transmission in FeCl₃‑Intercalated Graphene and WS₂ Thin Films for Advanced THz‑TDS Applications
- Advances in Carbon Nanotube Assembly and Integration for Next‑Generation Applications



