Platinum‑Based Catalysts on Carbon Supports and Conducting Polymers for Direct Methanol Fuel Cells: A Comprehensive Review
Abstract
Platinum nanoparticles remain the benchmark catalyst for direct methanol fuel cells (DMFCs), yet their high cost, sluggish methanol oxidation kinetics, and CO poisoning limit practical use. Recent advances focus on Pt alloys (e.g., Pt‑Ru, Pt‑Ni, Pt‑Co, Pt‑Sn) and advanced carbon supports (MWCNT, CNF, CNT, CNC, CMS, graphene) to reduce Pt loading, improve durability, and enhance electrocatalytic performance. This review summarizes alloy and support developments that lower Pt usage, increase stability, and boost DMFC metrics, discussing morphology, activity, structural traits, and fuel‑cell results for each system.
Introduction
Fuel cells convert chemical energy into electricity via electrochemical reactions. Direct methanol fuel cells (DMFCs) offer high power density, low‑temperature operation (≈ 373 K), and simple fuel handling, making them attractive for portable and stationary power. Nonetheless, DMFCs suffer from methanol crossover, slow oxidation kinetics, catalyst instability, and CO poisoning, hindering commercialization. This review focuses on strategies that combine Pt or Pt alloys with carbonaceous supports and conductive polymers to address these challenges.
DMFC chemistry is summarized by the anode oxidation of methanol to CO₂ and protons (Eq. 1) and the cathode reduction of O₂ to water (Eq. 2). The overall cell reaction is (Eq. 3). Two operating modes exist: active (recirculated methanol stream controlled by sensors) and passive (autonomous, no external pumping). Passive mode offers simpler, lower‑cost designs suitable for low‑power applications.
Figure 1 shows a single‑cell DMFC stack, including peristaltic methanol pumping, air supply, temperature control, and electrochemical measurement. Figure 2 illustrates schematic diagrams of active (a) and passive (b) DMFC configurations.
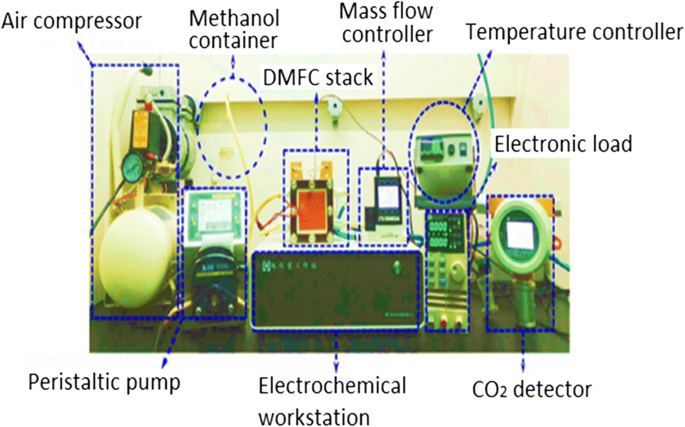
Experimental setup for a single‑cell DMFC.
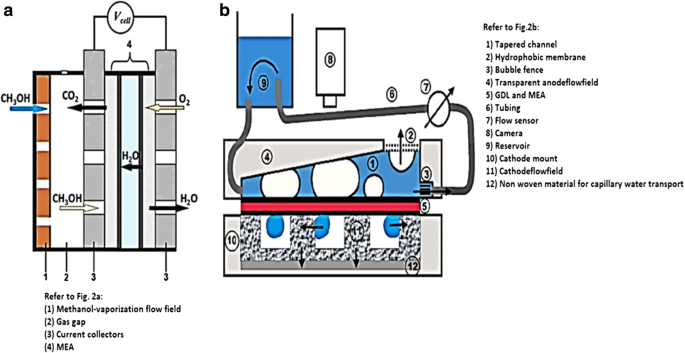
Schematic of active (a) and passive (b) DMFC modes.
Recent research investigates a range of catalysts: Pt alloys (Fe, Ni, Co, Rh, Ru, Sn), metal carbides (WC, MoC), metal nitrides (TiN, VN), transition metal oxides (SnO₂, TiO₂, WO₃), and conductive polymers (PANi, Ppy). Synthesis methods include impregnation, hydrothermal, microemulsion, and chemical reduction, each influencing particle size, dispersion, and activity.
Performance of Various Types of Pt-Based Catalysts
Pt alone is highly active for methanol oxidation but is readily poisoned by CO intermediates, and its scarcity drives costs. Alloying Pt with non‑precious metals (e.g., Ru, Ni, Co, Sn) introduces bifunctional and ligand effects that enhance kinetics and reduce CO adsorption. Table 1 (not reproduced here) reports average particle sizes and performance metrics for many Pt alloys synthesized via different routes.
Performance of Pt‑Based Alloys
Key findings: a 1:1 Pt:Ru ratio with 2–4 nm particles yields optimal activity; 3:2 Pt:Ru ratios also improve MOR. Ni incorporation (Pt‑Ni/C) boosts faradaic current by an order of magnitude, while Co and Sn provide additional OH species that oxidize CO. Ternary alloys (e.g., PtRuSn, PtRuNi) further enhance activity, stability, and CO tolerance, although their synthesis remains complex.
Mechanistic insight: Pt adsorbs methanol and forms CO_ads, while Ru or other secondary metals generate OH_ads that oxidize CO_ads to CO₂ (bifunctional mechanism, Eqs. 7–10). Similar pathways apply to PtNi and PtCo systems (Eqs. 11–15).
Performance of Pt‑Based Catalysts with Transition Metal Carbide
Wulfenium carbide (WC) and molybdenum carbide (MoC) support Pt, offering high electrical conductivity, acid resistance, and CO tolerance. Pt/WC catalysts exhibit higher CO₂ evolution and lower CO desorption potentials. Low‑load Pt (≤ 40 wt %) on WC or MoC yields superior MOR activity while reducing precious‑metal usage.
Performance of Pt‑Based Catalysts with Transition Metal Nitride
Metal nitrides (TiN, VN, TiCoN) provide metallic conductivity and structural stability. Pt/TiN and Pt/TiCoN composites show enhanced electrochemical surface area (ECSA) retention (> 65 % after 10 000 cycles) and MOR activity, outperforming commercial Pt/C. Nitride support promotes electron transfer and CO oxidation via strong Pt–nitride interactions.
Performance of Pt‑Based Catalysts with Transition Metal Oxide
Oxide supports such as SnO₂, TiO₂, WO₃, and MnO₂ introduce oxygen vacancies that aid CO oxidation. Pt/SnO₂ and Pt/TiO₂ composites exhibit higher MOR currents, improved CO tolerance, and enhanced durability compared to Pt/C. Mesoporous WO₃ and WO₃‑based templates further increase surface area and mass transport.
Carbon Support
Carbon materials—graphene, CNTs, CNFs, mesoporous carbon, and carbon black—are essential for dispersing Pt nanoparticles, increasing surface area, and ensuring conductivity. Each support influences particle size, alloying degree, and CO tolerance.
Graphene Support
Reduced graphene oxide (rGO) and functionalized graphene (N‑doped, polymer‑coated) provide high surface area (up to 2630 m² g⁻¹), excellent conductivity, and oxygen functional groups that anchor Pt and Pt alloys. Pt/rGO, PtCo/rGO, and Pt/NCL‑rGO composites demonstrate up to 2.8‑fold higher MOR current densities and superior CO tolerance relative to commercial Pt/C.
Multi‑wall and Single‑wall Carbon Nanotube Supports
MWCNTs and SWCNTs offer high aspect ratios, large surface areas, and good electronic pathways. Pt–Ru/MWCNT and Pt–Ru/SWCNT catalysts achieve current densities > 39 mW cm⁻² and enhanced CO oxidation due to efficient charge transfer and oxygen‑rich functional groups.
Carbon Nanofiber Support
CNFs, when combined with Pt or Pt alloys, yield high dispersion and improved MOR activity. Pt/CNF and PtRu/CNF composites show current densities 3–5 × higher than commercial Pt/C, attributed to the hydrophilic CNF surface and high surface area.
Mesoporous Carbon Support
Ordered mesoporous carbons (OMCs) with pore sizes 3–10 nm provide uniform dispersion of Pt nanoparticles and fast mass transport. PtFe/OMC and Pt/WC/OMC composites exhibit high MOR activity, excellent stability, and CO tolerance.
Carbon Black
Commercial carbon blacks (Vulcan XC‑72, Ketjen Black) remain common supports due to their porosity and conductivity. Ketjen Black EC 300J offers the highest ECSA and best DMFC performance among tested black carbons.
Carbon Nanocoils
Highly graphitic CNCs with high porosity support Pt or PtRu nanoparticles, yielding high ECSA (up to 85 m² g⁻¹ Pt) and superior MOR performance compared to Pt/Vulcan.
Conductive Polymer Supports
Polyaniline (PANi) and polypyrrole (PPy) provide high conductivity and hydrophilicity, enabling better Pt or Pt alloy dispersion. PtRu/PANi and PtRu/PPy/C composites show enhanced MOR activity, CO tolerance, and reduced methanol crossover relative to Pt/C.
Problems and Limitations of Using Pt for DMFC Systems
Key issues: CO formation during methanol oxidation, slow kinetics, high Pt cost, and methanol crossover. Alloying with Ru, Ni, Co, Sn, and incorporating carbides, nitrides, oxides, or carbon supports mitigates CO poisoning and improves kinetics. Understanding the reaction pathways—direct (no CO intermediate) versus indirect (CO intermediate)—guides catalyst design.
Conclusion and Prospects
Significant progress has been made in tailoring Pt‑based catalysts and their supports to reduce Pt loading, enhance CO tolerance, and accelerate methanol oxidation. Advanced nanostructures (hollow, mesoporous, 3‑D) and alloy compositions (PtRu, PtCo, PtNi, PtSn, ternary/quadruple alloys) combined with high‑surface‑area carbon or conductive polymer supports hold promise for commercial DMFCs. Future work should focus on optimizing support morphology, achieving uniform alloy distribution, and integrating cost‑effective materials to enable scalable, durable, and high‑performance DMFC technologies.
Abbreviations
- CB:
Carbon black
- CH3O:
methoxy group
- CNC:
Carbon nanocoil
- CNF:
Carbon nanofiber
- CNT:
Carbon nanotube
- Co:
Cobalt
- CO:
carbon monoxide
- CO2:
carbon dioxide
- DMFC:
direct methanol fuel cell
- FC:
fuel cell
- Fe:
iron
- MOR:
methanol oxidation reaction
- MPC:
mesoporous carbon
- MWCNT:
multi‑wall carbon nanotube
- Ni:
nickel
- OMC:
ordered mesoporous carbon
- ORR:
oxygen reduction reaction
- PANi:
polyaniline
- PEMFC:
proton exchange membrane fuel cell
- Ppy:
polypyrrole
- Pt:
platinum
- Pt/MWCNT:
platinum‑supported MWCNT
- Pt‑Ru/MWCNT:
platinum‑ruthenium‑supported MWCNT
- Rh:
rhodium
- Ru:
ruthenium
- Sn:
tin
- SOFC:
solid oxide fuel cell
- SWCNT:
single‑wall carbon nanotube
- TMN:
transition metal nitride
Nanomaterials
- Gold Nanoparticles: Advancing Diagnostic and Therapeutic Applications in Medicine – A Comprehensive Review
- Fluorescent Nanomaterials: Advances, Synthesis, and Biomedical Applications
- Graphene‑Polymer Composites for High‑Performance Supercapacitors: A Comprehensive Review
- Electrospun TiO₂–Carbon Nanofiber Support Enhances PtRu Catalysts for Direct Methanol Fuel Cells
- Enhanced Anodic Catalyst Support Using TiO₂‑Carbon Nanofibers for Direct Methanol Fuel Cells
- PdAu Nanoparticle Catalysts on Vapor‑Grown Carbon Nanofibers: Performance in Passive Direct Glycerol Fuel Cells
- Advances in Carbon Nanotube Assembly and Integration for Next‑Generation Applications
- Ultrasmall Superparamagnetic Iron Oxide Nanoparticles for Stem Cell Tracking in Parkinson’s Therapy
- SGL Carbon & Hyundai Motor Group Strengthen Fuel Cell Partnership for NEXO Production
- Comparing Polypropylene and Nylon (Polyamide): Advantages, Disadvantages, and Best Uses



