Electrospun TiO₂–Carbon Nanofiber Support Enhances PtRu Catalysts for Direct Methanol Fuel Cells
Abstract
Platinum remains the benchmark catalyst for direct methanol fuel cells (DMFCs), yet it suffers from rapid poisoning by carbonaceous intermediates, which degrades cell performance. In this work, we report the fabrication of a TiO₂–carbon nanofiber (CNF) support engineered by electrospinning, designed to mitigate poisoning and enhance electrocatalytic activity. By systematically varying the tip‑to‑collector distance (DTC) and the precursor flow rate, we identified the optimal conditions that produce ultrafine fibers with superior electrochemical properties. Comprehensive characterization—FTIR, XRD, FESEM, TEM—and electrochemical testing (cyclic voltammetry) demonstrate that the D18 sample, fabricated at a flow rate of 0.1 mL h⁻¹ and a DTC of 18 cm, attains the smallest mean diameter (136.73 ± 39.56 nm). This nanofiber delivers the highest catalyst activity (274.72 mA mg⁻¹ PtRu) and electrochemical surface area (226.75 m² g⁻¹ PtRu) among the variants studied.
Background
Direct methanol fuel cells (DMFCs) represent a promising, environmentally friendly power source, generating electricity directly from liquid methanol without combustion. Their simplicity, high specific energy, low operating temperature, and rapid refueling make them attractive for portable and stationary applications. Nevertheless, DMFC performance is constrained by catalyst poisoning and sluggish reaction kinetics, largely stemming from the catalyst and support materials used.
Platinum (Pt) has traditionally served as the active component in DMFC anodes; however, its susceptibility to carbonaceous intermediates limits long‑term activity. The introduction of a bimetallic PtRu catalyst improves reaction rates, yet the kinetic limitations persist. Researchers have therefore explored metal oxides and nanostructured materials as synergistic supports to enhance electron transfer, thermal stability, and catalyst dispersion. Titanium dioxide (TiO₂) is particularly appealing due to its non‑toxicity, corrosion resistance, and ability to modify electronic properties of composite catalysts.
Nanofiber architectures, produced by electrospinning, offer high surface‑to‑volume ratios, excellent electrical conductivity, and uniform catalyst dispersion—features that collectively boost electrocatalytic performance. This study focuses on TiO₂–carbon nanofibers fabricated via electrospinning, aiming to reduce catalyst poisoning and elevate DMFC output.
Electrospinning is favored for producing sub‑micron fibers owing to its simplicity, versatility, and scalability. Key process parameters—solution composition, ambient conditions, and operating variables—dictate fiber morphology. Here, we concentrate on two process parameters: the flow rate of the polymer precursor and the distance between the needle tip and collector (DTC), as these directly influence fiber diameter and surface characteristics.
Methods/Experimental
Materials
Poly(vinyl acetate) (PVAc, Mw = 500 000), dimethylformamide (DMF, 99.8 %), titanium isopropoxide (TiPP, 97 %), acetic acid (99.7 %), and a ruthenium precursor (45–55 % content) were purchased from Sigma‑Aldrich. A platinum precursor (40 % content) and ethanol (99.8 %) were supplied by Merck and R&M Chemical Reagents, respectively. All reagents were used as received. Electrospinning was performed with an Nfiber N1000 system (Progene Link), and ultrasonic homogenization employed an INS‑650Y crusher (INS Equipments).
Preparation of TiO₂‑CNF Nanofibers
The precursor solution was prepared by dissolving PVAc (11.5 wt %) in DMF, stirring at 60 °C for 1 h, then at room temperature overnight. TiPP was added at a 1:1 ratio with the polymer solution, followed by a trace of acetic acid and ethanol to promote hydrolysis. The mixture was sonicated for 60 s to ensure homogeneity, then loaded into a syringe for electrospinning. A voltage of 16 kV was applied, with flow rates of 0.1, 0.5, and 0.9 mL h⁻¹ (denoted F0.1, F0.5, F0.9) and tip‑to‑collector distances of 14, 16, and 18 cm (denoted D14, D16, D18). After electrospinning, fibers rested 5 h, stabilized at 130 °C for 8 h, and carbonized at 600 °C for 2 h under nitrogen. The resulting nanofibers were ground to a fine powder and weighed to achieve a uniform loading of 6.67 mg g⁻¹.
Deposition of Catalyst
TiO₂‑CNF powder was dispersed in isopropyl alcohol and deionized water, sonicated for 20 min, and mixed with a 1:1 Pt:Ru precursor (20 wt %). After pH adjustment to 8 using NaOH, the mixture was heated to 80 °C and reduced with 25 mL of 0.2 M NaBH₄ dropwise. Following 1 h stirring, the suspension was filtered, washed, dried at 120 °C for 3 h, and milled to a fine catalyst powder.
Characterization of the Catalyst
FTIR (PerkinElmer), XRD (D8 Advance/Bruker), FESEM (SUPRA 55VP), and TEM (Tecnai G2 F20 X‑Twin) were employed to assess chemical composition, crystal structure, morphology, and elemental distribution.
Evaluation of the Electrochemical Measurement
Electrochemical performance was evaluated in a standard three‑electrode cell using a Pt counter, Ag/AgCl reference, and a 3 mm glassy carbon working electrode. The catalyst ink (15 mg catalyst, 400 µL DI water, 400 µL IPA, 125 µL 5 wt % Nafion) was dropped (2.5 µL) onto the electrode, dried, and thermally treated. The electrolyte was 0.5 M H₂SO₄/2 M methanol, purged with N₂ for 20 min. Cyclic voltammetry (−0.1 V to 1.1 V, 50 mV s⁻¹) was performed with an Autolab workstation.
Results and Discussion
Structural Characterization
Effect of Flow Rate
FTIR spectra of fibers produced at 0.1, 0.5, and 0.9 mL h⁻¹ reveal characteristic Ti–O and C–O bands, confirming consistent chemistry across flow rates. XRD patterns show coexistence of anatase and rutile TiO₂ and carbon peaks (30° and 55° 2θ). FESEM images illustrate that the lowest flow rate (F0.1) yields the smoothest fibers with the smallest mean diameter (161.18 ± 26.08 nm). Higher flow rates produce rougher fibers with larger diameters and occasional beads.

FESEM image of TiO₂‑CNF (F0.1), (F0.5), and (F0.9) at ×30,000 magnification
Effect of the Distance Between the Tip and Collector
FTIR analysis of samples with varying DTC (14, 16, 18 cm) shows no significant change in bonding patterns. XRD confirms TiO₂ anatase/rutile coexistence across all distances. FESEM images reveal that increasing DTC leads to progressively thinner fibers, with D18 achieving the smallest mean diameter (136.73 ± 39.56 nm). This trend reflects enhanced jet whipping and thinning over longer travel distances.

FESEM image of TiO₂‑CNF (D14), (D16), and (D18) at ×30,000 magnification
Electrochemical Characterization of the Methanol Oxidation Reaction
PtRu was uniformly dispersed on both F0.1 and D18 fibers, as confirmed by TEM. XRD indicates anatase TiO₂ crystallite sizes of 20–22 nm across the F series, while the D series shows decreasing TiO₂ and Pt crystallite sizes with smaller fiber diameters. These morphological changes translate into higher electrochemical surface areas (ECSA). CV measurements in 0.5 M H₂SO₄/2 M methanol reveal that the D18 support yields the highest peak current density (274.72 mA mg⁻¹ PtRu) and ECSA (226.75 m² g⁻¹ PtRu), surpassing both the F series and other D samples.
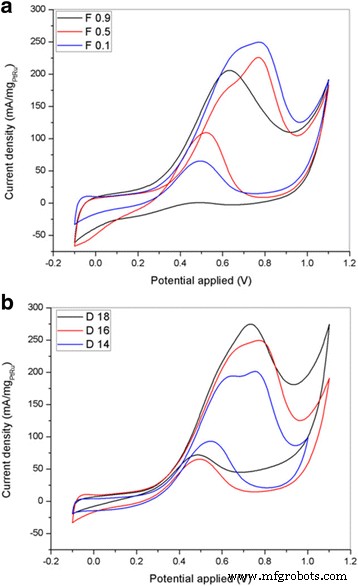
Cyclic voltammograms for PtRu/TiO₂‑CNF with different flow rate and DTC in 2 M methanol/0.5 M H₂SO₄ at 50 mV s⁻¹
The forward/reverse current ratio (I_f/I_b) for the D18 sample exceeds 3.8, indicating strong tolerance to carbonaceous poisoning and excellent durability.
Conclusion
Electrospun TiO₂–carbon nanofibers fabricated at a 0.1 mL h⁻¹ flow rate and 18 cm DTC (D18) exhibit the thinnest morphology (136.73 ± 39.56 nm). When coupled with PtRu, the D18 support delivers a 1.4‑fold increase in current density and a ten‑fold rise in ECSA relative to other samples, underscoring the critical role of fiber diameter in catalyst performance. These findings affirm that ultrafine TiO₂‑CNF supports are promising anode materials for high‑performance DMFCs.
Nanomaterials
- Optimizing Carbon Nanotube Materials and Composites through Advanced Porosimetric Characterization
- Optimizing PdAu/VGCNF Anode Catalyst for Enhanced Glycerol Fuel Cell Performance
- Enhanced Anodic Catalyst Support Using TiO₂‑Carbon Nanofibers for Direct Methanol Fuel Cells
- Facile Thermal Decomposition Loading of CeO₂ Nanoparticles onto Anodic TiO₂ Nanotube Arrays
- Platinum‑Based Catalysts on Carbon Supports and Conducting Polymers for Direct Methanol Fuel Cells: A Comprehensive Review
- PdAu Nanoparticle Catalysts on Vapor‑Grown Carbon Nanofibers: Performance in Passive Direct Glycerol Fuel Cells
- Efficient Synthesis of CNT–Cu₂O Nanocomposites for Rapid p‑Nitrophenol Reduction: A Low‑Cost, Reusable Catalyst
- Hemp‑Stem‑Derived Nanoporous Carbon Anodes for High‑Capacity Lithium‑Ion Batteries
- Efficient ICP Etching for Black GaAs Nanoarrays: Simple Fabrication & Low Reflectance
- Enhanced TiO₂ Catalysis and Antimicrobial Efficacy via Nitrogen and Carbon Nitride Co‑Doping



