Efficient Synthesis of CNT–Cu₂O Nanocomposites for Rapid p‑Nitrophenol Reduction: A Low‑Cost, Reusable Catalyst
Developing stable, high‑performance nanocomposites is essential for advanced catalytic applications. Here we report a simple, scalable route to fabricate carbon nanotube (CNT)–Cu₂O nanocomposites without surfactants or stabilizers. By tuning the reaction temperature and duration, we achieved uniform Cu₂O nanoparticles (30–50 nm) anchored on CNTs, yielding a catalyst that reduces p‑nitrophenol (p‑NP) to p‑aminophenol (p‑AP) in just 35 s. The catalyst retains 92 % of its activity after eight consecutive runs, underscoring its robustness and suitability for wastewater remediation and composite‑material fabrication. Since their discovery, carbon nanotubes (CNTs) have emerged as versatile platforms for catalysis, energy storage, sensing, and environmental remediation due to their exceptional chemical stability, electrical conductivity, high surface area, and mechanical strength. Numerous studies have integrated transition‑metal nanoparticles—such as Cu, Pd, Au, Ag, and Pt—onto CNTs to create hybrid catalysts with enhanced activity and selectivity. Cu-based nanomaterials, in particular, offer a low‑cost, abundant alternative to noble metals, and their catalytic behavior can be tuned by controlling particle size, morphology, and surface composition. While previous reports have demonstrated Cu/CNT and CuO/CNT composites for various reactions, the synthesis of Cu₂O/CNT hybrids remains less explored, largely because conventional methods involve complex, multi‑step procedures or hazardous reagents. We address this gap by presenting a facile, green synthesis that leverages ascorbic acid as a mild reducing agent and avoids stabilizing agents, thereby preserving the intrinsic catalytic properties of Cu₂O nanoparticles. Materials: Multi‑walled aminated CNTs (95 %, 3–5 nm inner diameter, 8–15 nm outer diameter, 50 µm length), CuCl (97 %) and CuCl₂ (98 %) as Cu precursors, NaOH, L‑ascorbic acid, NaBH₄, and p‑NP were purchased from Aladdin, Tianjin Kermel, and Shanghai Hushi. All reagents were analytical grade and used as received. Synthesis of CNT–Cu₂O composites: 100 mg CNT and 100 mg CuCl were dispersed in 250 mL deionized water under magnetic stirring (300 rpm) at 30 °C for 20 min. After 1 h of stirring, 0.88 g L‑ascorbic acid and 5.0 mL 1 M NaOH were added. The mixture was washed repeatedly with ethanol and water, then dried at 50 °C under vacuum for 48 h. For comparison, reactions were also carried out at 30 °C for 6 h and at 60 °C for 1 h. All samples were finally dried at 50 °C for 48 h. Catalytic test: 0.0174 g 4‑NP and 0.1892 g NaBH₄ were dissolved in 25 mL deionized water to prepare a 0.313 mM 4‑NP solution. 10 mg of CNT/Cu₂O catalyst was dispersed in 16 mL of this solution at room temperature. UV–vis spectra (220–550 nm) were recorded every 4 min to monitor the disappearance of the 401 nm peak (4‑NP⁻). After each run, the catalyst was recovered by centrifugation, washed with ethanol and water, and reused. Characterization: Morphology was examined by SEM (Hitachi S‑4800II, 15 kV) and TEM (Hitachi HT7700, 200 kV). Elemental mapping used EDXS (Oxford Link‑ISIS). Raman spectra were collected with a Horiba Jobin Yvon Xplora PLUS confocal Raman microscope. XRD patterns were recorded on a Rigaku SmartLab (Cu Kα radiation). Figure 1 shows SEM/TEM images of CNT–Cu₂O composites prepared under different conditions. The sample synthesized at 30 °C for 1 h (CNT‑Cu‑30‑1) displays Cu₂O nanoparticles uniformly distributed on CNT surfaces with diameters of 30–50 nm. In contrast, samples prepared at 30 °C for 6 h (CNT‑Cu‑30‑6) or 60 °C for 1 h (CNT‑Cu‑60‑1) exhibit larger, agglomerated Cu₂O particles up to several micrometres. Elemental mapping (Figure 2) confirms homogeneous distribution of C, O, and Cu, indicating that CNTs effectively act as a scaffold for Cu₂O nucleation. SEM and TEM images of the synthesized CNT‑Cu₂O nanocomposites. a, d CNT‑Cu‑30‑1. b, e CNT‑Cu‑60‑1. c, f CNT‑Cu‑30‑6 SEM image of CNT‑Cu‑30‑1 nanocomposite and elemental mappings of C, O, and Cu Powder XRD patterns (Figure 3) reveal characteristic peaks of CNTs (26°) and Cu₂O (PDF 05‑0667), with no detectable impurities. Re‑analysis after one month shows identical patterns, confirming the long‑term stability of the Cu₂O nanoparticles. Raman spectra (Figure 4) display the expected G (≈1605 cm⁻¹), D (≈1344 cm⁻¹), and 2D (≈2693 cm⁻¹) bands of CNTs, while Cu₂O lattice vibrations appear at 223 cm⁻¹ and 485 cm⁻¹. The D/G intensity ratio decreases from 1.64 (pure CNT) to 1.34 (CNT‑Cu‑30‑1), indicating improved crystallinity due to Cu₂O loading. XRD curves of the synthesized CNT‑Cu₂O nanocomposites Raman spectra of the CNT and synthesized CNT‑Cu₂O nanocomposites Catalytic performance was evaluated by UV–vis monitoring of the 4‑NP → 4‑AP conversion. As shown in Figure 5, CNT‑Cu‑30‑1 achieves complete conversion within 35 s, while CNT‑Cu‑60‑1 and CNT‑Cu‑30‑6 require 11–12 min. The rapid kinetics are attributed to the high surface area and optimal particle size of Cu₂O nanoparticles. Photographs (Figure 5d) confirm the color change from bright yellow (4‑NP⁻) to clear (4‑AP). Catalytic reduction of 4‑NP via present synthesized CNT‑Cu₂O nanocomposites. a CNT‑Cu‑30‑1. b CNT‑Cu‑60‑1. c CNT‑Cu‑30‑6. d The photos of 4‑NP solution before and after the catalytic process Reusability tests (Figure 6) demonstrate that the catalyst retains 92 % activity after eight cycles, with only minor loss due to possible surface fouling or incomplete recovery. Future designs may incorporate magnetic nanoparticles to simplify catalyst retrieval. Reusability test of CNT‑Cu‑30‑1 nanocomposite as a catalyst for the reduction of 4‑nitrophenol with NaBH₄ Schematic illustration of the fabrication and catalytic reduction of the synthesized CNT‑Cu₂O nanocomposites In summary, the low‑temperature, one‑pot synthesis of CNT‑Cu₂O nanocomposites provides a cost‑effective, scalable route to produce highly active catalysts for the rapid reduction of nitroaromatic pollutants. The uniform Cu₂O nanoparticles (30–50 nm) anchored on CNTs enable fast electron transfer and high surface reactivity, achieving 99 % conversion in 35 s and maintaining 92 % efficiency after eight cycles. These results highlight the potential of CNT‑Cu₂O hybrids for wastewater treatment and the broader field of composite catalyst development. We have established a facile, surfactant‑free synthesis of CNT–Cu₂O nanocomposites that yields uniform 30–50 nm Cu₂O particles on CNTs. The optimized catalyst (30 °C, 1 h) converts 4‑NP to 4‑AP in 35 s and retains 92 % activity after eight uses, demonstrating exceptional catalytic stability. The combination of CNT’s structural support and Cu₂O’s catalytic prowess offers a promising platform for environmental remediation and composite‑material applications.Abstract
Background
Methods
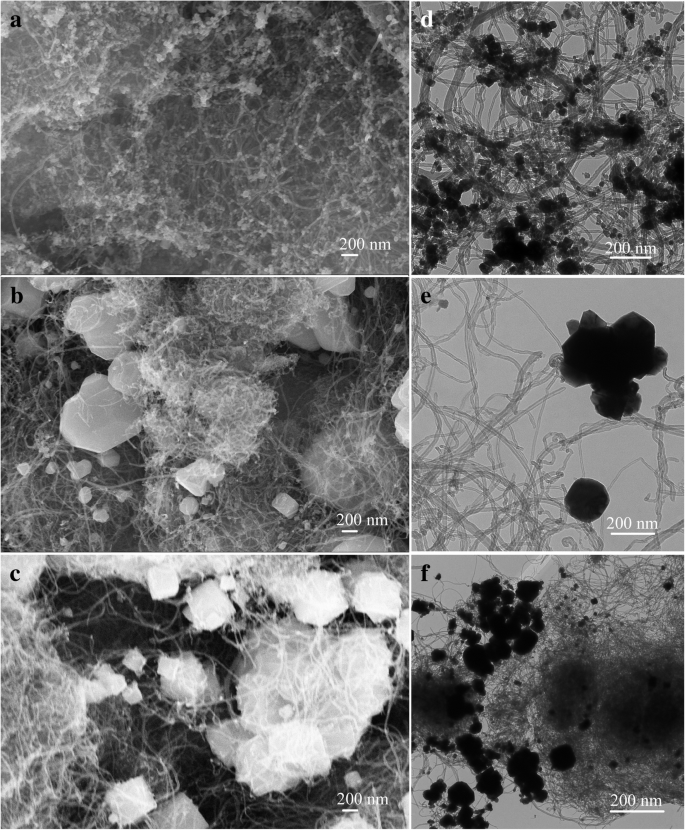
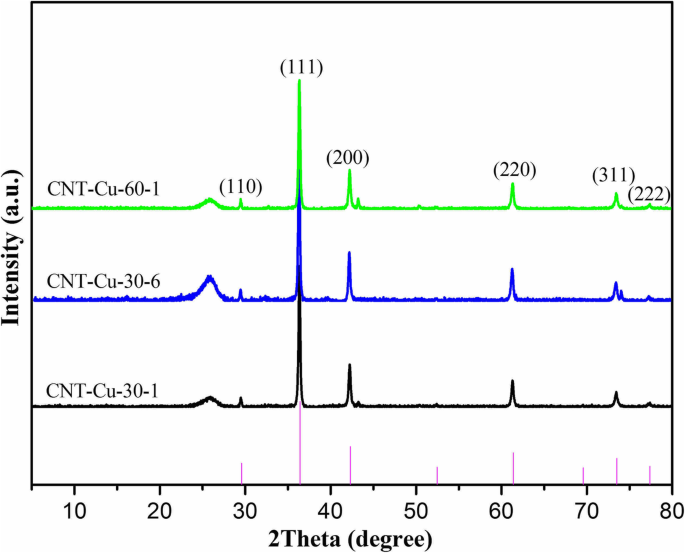
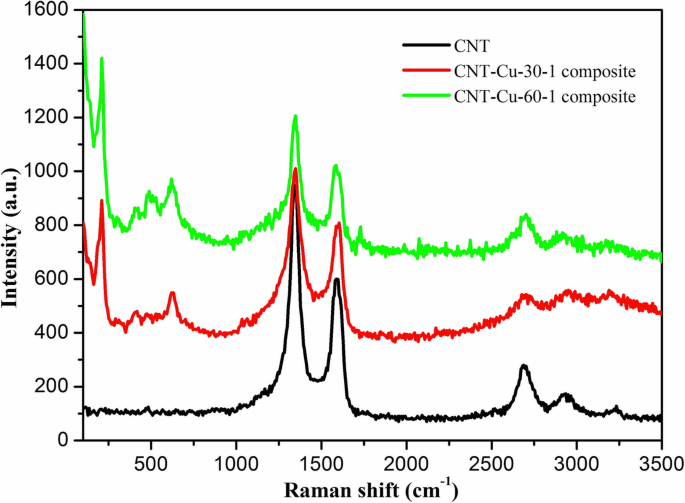
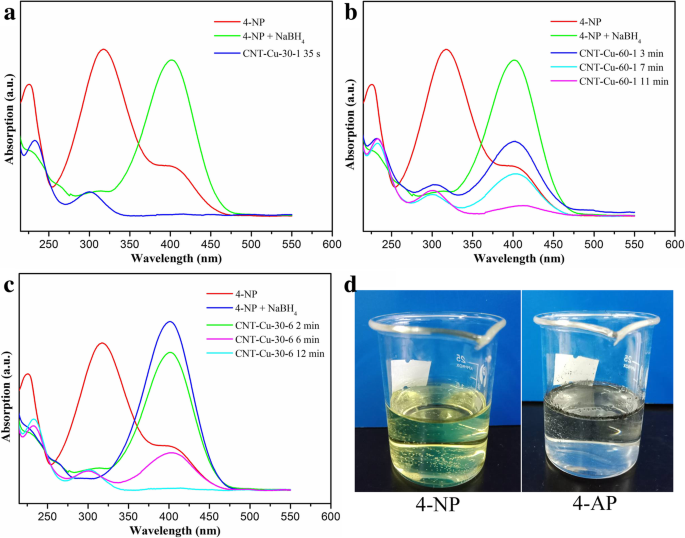
Results and Discussion

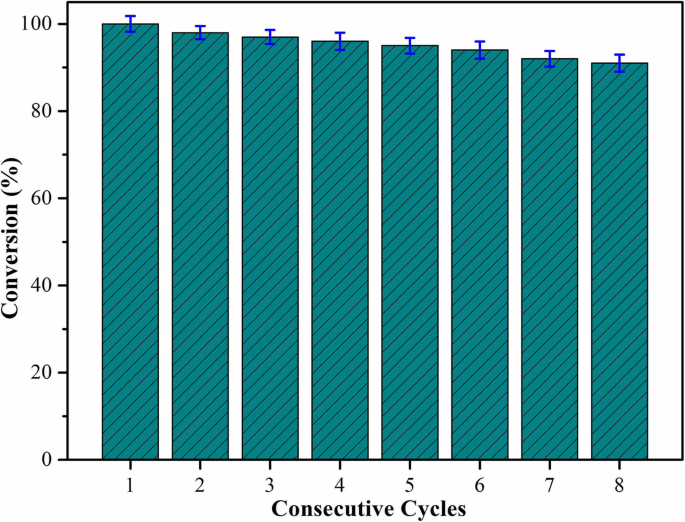
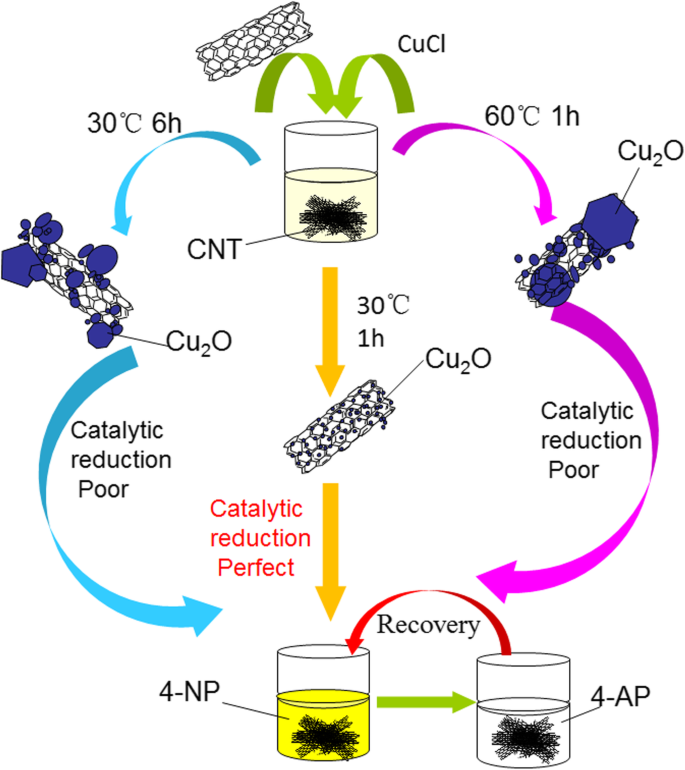
Conclusions
Nanomaterials
- Introducing Ultem 1010 & ASA: Advanced FDM Materials for Stratasys Fortus Printers
- Electrospun TiO₂–Carbon Nanofiber Support Enhances PtRu Catalysts for Direct Methanol Fuel Cells
- Polycatechol‑Modified Fe<sub>3</sub>O<sub>4</sub> Magnetic Nanoparticles: A Highly Selective, Magnetically Recoverable Adsorbent for Cationic Dye Removal
- Efficient Fabrication of Cu₂O/TiO₂ Nanotube Heterojunctions for Enhanced Visible‑Light Photocatalysis
- 5‑Minute Microwave‑Assisted Synthesis of Polydopamine‑Passivated Carbon Dots with 5 % Quantum Yield and 35 % Photothermal Efficiency for Cancer Therapy
- Nitrogen‑Doped Porous Carbon Networks Derived from Graphitic Carbon Nitride for High‑Performance Oxygen Reduction
- Li₂RuO₃-Activated Lithia Nanocomposites for Advanced Oxygen Redox Cathodes
- Carbon Dots: Pioneering Next-Generation Nanothermometers – A Comprehensive Review
- Trelleborg Partners with Innovate Composites to Expand Tooling Material Distribution in New Zealand
- Solvay Introduces Advanced Materials & Processes for Industrial Production



