Green One‑Pot Synthesis of Ag‑Decorated SnO₂ Microspheres: A Highly Reusable Catalyst for 4‑Nitrophenol Reduction
Abstract
Hierarchical silver‑decorated tin(IV) oxide (SnO₂) microspheres were obtained through a single‑step hydrothermal route that requires no templates or surfactants. X‑ray diffraction (XRD), scanning and transmission electron microscopy (SEM/TEM), X‑ray photoelectron spectroscopy (XPS), Brunauer–Emmett–Teller (BET) surface analysis, and Fourier‑transform infrared spectroscopy (FTIR) confirmed the formation of crystalline SnO₂ nanosheets uniformly decorated with ~5 nm Ag nanoparticles. The resulting nanocomposite was tested as a catalyst for the reduction of 4‑nitrophenol (4‑NP) to 4‑aminophenol (4‑AP) using potassium borohydride (KBH₄) as a reducing agent. A 36‑h hydrothermal product exhibited a normalized rate constant (κnor) of 6.20 min⁻¹ g⁻¹ L, surpassing many previously reported catalysts. The material retained over 80 % of its initial activity after ten successive cycles, demonstrating robust reusability. These results suggest that the Ag‑decorated SnO₂ microspheres are promising candidates for sustainable water‑pollutant remediation and resource recovery.
Background
SnO₂ is a wide‑bandgap n‑type semiconductor (Eg = 3.6 eV) known for its high electron mobility and low cost. Its versatile properties have driven research into gas sensing, lithium‑ion batteries, optoelectronics, and dye‑sensitized solar cells. While various nanostructures of SnO₂ have been explored, three‑dimensional (3D) hierarchical assemblies built from nanosheets are particularly attractive because of their high surface area and accessible active sites. However, the catalytic performance of SnO₂, especially in pollutant degradation, remains limited. Decorating SnO₂ with noble metals such as Ag, Au, Pt, or Pd is a proven strategy to boost catalytic activity, yet most synthesis routes involve multi‑step procedures or hazardous reagents.
4‑Nitrophenol (4‑NP) is a persistent industrial contaminant. Its conversion to 4‑aminophenol (4‑AP) not only removes a toxic compound but also produces a valuable chemical feedstock used in pharmaceuticals and dyes. Chemical reduction of 4‑NP with KBH₄ is a widely adopted model reaction for evaluating catalytic efficiency. In this study, we present a straightforward, environmentally friendly synthesis of Ag‑decorated SnO₂ microspheres and demonstrate their superior catalytic performance in 4‑NP reduction.
Methods
Materials
Silver nitrate (AgNO₃, 99.8 %), urea (CO(NH₂)₂, 99 %), ammonium hydroxide (25–28 %), and potassium borohydride (KBH₄, 97 %) were purchased from Sinopharm Chemical Reagent Co. Ltd. Sodium stannate dihydrate (Na₂SnO₃·3H₂O, 98 %) and 4‑nitrophenol (C₆H₅NO₃, 98 %) were supplied by Aladdin Reagent Co. Ltd. All reagents were used without further purification.
Synthesis of Ag‑Decorated SnO₂ Microspheres
In a typical procedure, 2.67 g of Na₂SnO₃·3H₂O and 0.2 g of urea were dissolved in 25 mL of ultrapure water and stirred vigorously for 30 min. Separately, 1.69 g of AgNO₃ was dispersed in 25 mL of ultrapure water; 2.4 mL of ammonium hydroxide was then added to generate a silver‑ammonia complex. After 5 min of stirring, this complex was slowly introduced into the SnO₂ precursor solution and stirred for 1 h. The resulting mixture was transferred to a 50 mL Teflon‑lined autoclave and heated at 150 °C for 5, 10, 24, or 36 h. After cooling to room temperature, the solids were collected by centrifugation, washed with deionized water and ethanol, and dried at 60 °C under vacuum. Variations in the Ag:SnO₂ molar ratio (1.5:1, 1:1, 0.5:1, 0.01:1) were achieved by adjusting the amounts of AgNO₃ and ammonium hydroxide. For comparison, pure SnO₂ and Ag nanoparticles were also prepared following the same protocol without the corresponding precursor.
Sample Characterizations
Phase purity was examined by XRD (Cu Kα radiation, λ = 1.5418 Å). Morphology and microstructure were studied using a SU‑70 field‑emission SEM and a Tecnai G2 F20 S‑TWIN TEM/HRTEM. XPS (Mg Kα radiation) provided surface composition and oxidation states. Nitrogen adsorption–desorption isotherms (V‑sorb X2008) yielded BET surface areas and pore size distributions (BJH method).
Catalytic Activity Assessment
The reduction of 4‑NP by KBH₄ was monitored in a quartz cuvette (1‑cm path length, ~4 mL). A 4‑NP solution (20 mg L⁻¹) and excess KBH₄ (1.5 mg) were mixed, and the addition of 1.5 mg of catalyst immediately turned the solution from light yellow to dark yellow, indicating 4‑NP complexation. UV–Vis spectra (250–500 nm) were recorded every minute to follow the decay of the 400 nm absorption band (4‑NP) and the emergence of the 300 nm band (4‑AP). The pseudo‑first‑order kinetics were analyzed using ln(At/A0) = −κapp t, where κapp is the apparent rate constant. Multiple reaction cycles were performed to evaluate catalyst durability.
Results and Discussion
Structural and Morphological Characterization
Figure 1 shows XRD patterns for products prepared at 150 °C for different times. All samples matched the tetragonal rutile SnO₂ (JCPDS 41‑1445) and face‑centered cubic Ag (JCPDS 04‑0783). Peaks at 38.12° and 44.2° (Ag (111) and (200)) grew stronger with longer hydrothermal times, indicating increased Ag crystallinity and loading. No secondary phases were detected, confirming the purity of the composites.
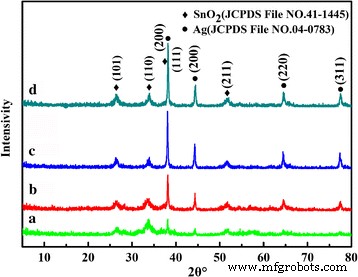
XRD patterns of Ag‑decorated SnO₂ microspheres prepared at 150 °C for different times: (a) 5 h, (b) 10 h, (c) 24 h, (d) 36 h.
SEM images (Figure 2) reveal a clear morphological evolution. The 5‑h sample consists of irregular microspheres assembled from primary nanoparticles. As the hydrothermal time extends to 24 h, the particles reorganize into nanosheets, forming a well‑defined hierarchical microsphere. After 36 h, the structure stabilizes, producing uniform spheres with diameters of 2–4 µm. TEM/HRTEM of the 36‑h product (Figure 3) confirms a ~5 µm microsphere composed of ~5 nm Ag nanoparticles uniformly distributed on SnO₂ nanosheets. The lattice fringes (0.26 nm for Ag (111) and 0.33 nm for SnO₂ (110)) corroborate the crystalline nature of both components. Elemental mapping further demonstrates the homogeneous dispersion of Ag, Sn, and O throughout the microsphere.
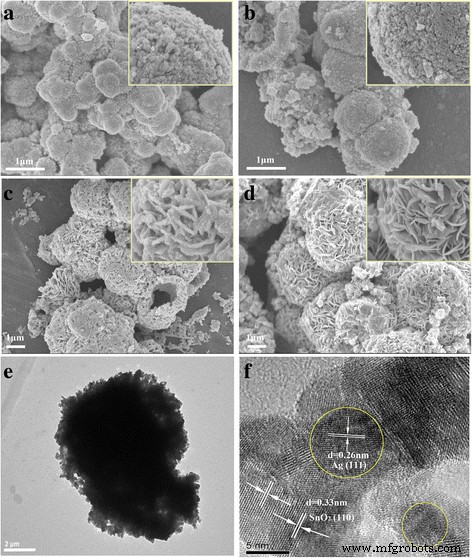
FESEM and TEM images of Ag‑decorated SnO₂ microspheres at different hydrothermal times: (a) 5 h, (b) 10 h, (c) 24 h, (d) 36 h; (e) low‑magnification TEM, (f) HRTEM of the 36‑h sample.
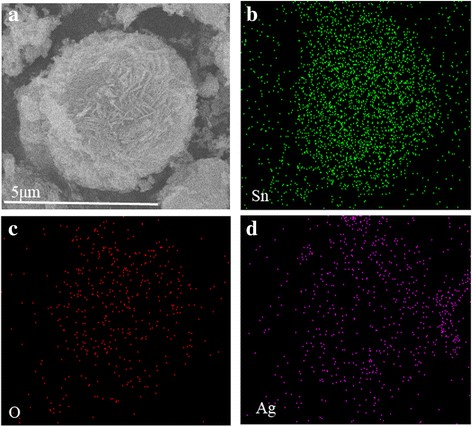
EDS element mapping of the 36‑h microsphere: (a) SEM image, (b) Sn map, (c) O map, (d) Ag map.
Nitrogen adsorption–desorption isotherms (Figure 4) exhibit type IV behavior with H₃ hysteresis, indicating mesoporosity. BET surface areas range from 21.8 to 25.7 m² g⁻¹, with an average pore diameter of ~2 nm across all samples. The consistent surface properties suggest that morphological changes do not significantly affect the porosity.
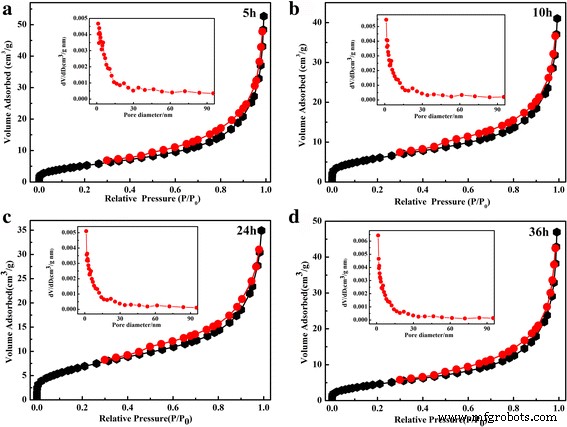
Nitrogen adsorption–desorption isotherms of the 36‑h microsphere at 150 °C for different hydrothermal times: (a) 5 h, (b) 10 h, (c) 24 h, (d) 36 h.
High‑resolution XPS spectra (Figure 5) reveal metallic Ag⁰ (Ag 3d5/2 = 369.1 eV, Ag 3d3/2 = 375.2 eV) and Sn⁴⁺ (Sn 3d5/2 = 488.0 eV). The slight binding‑energy shifts suggest strong electronic coupling between Ag and SnO₂, which facilitates electron transfer during catalysis. The O 1s spectrum shows lattice oxygen (530.5 eV) and surface‑adsorbed species (532.6 eV), indicating a highly reactive surface.
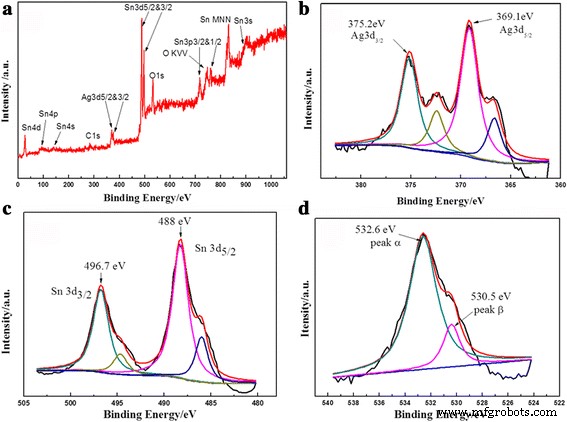
Representative XPS spectra of the 36‑h microsphere: (a) survey, (b) Ag 3d, (c) Sn 3d, (d) O 1s.
Catalytic Reduction of 4‑NP
UV–Vis monitoring of the 4‑NP reduction (Figure 6) demonstrates that the 400 nm absorption band disappears within 1 min, while a new 300 nm band emerges, confirming rapid conversion to 4‑AP. The apparent rate constant for the 36‑h sample is κapp = 3.10 min⁻¹. When evaluated across different hydrothermal times, κapp increases with longer synthesis times, reaching a maximum at 36 h. The catalyst also shows excellent recyclability: after ten consecutive runs, κapp drops only slightly, and the normalized rate constant κnor remains 6.20, 0.64, and 0.54 min⁻¹ g⁻¹ L for the first, fifth, and tenth cycles, respectively. These values outperform many reported SnO₂‑based and noble‑metal‑modified catalysts.
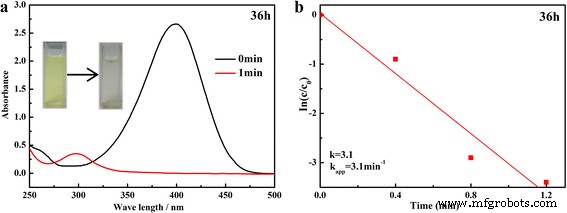
Time‑dependent UV–Vis spectra and ln(At/A0) versus time plot for the 36‑h sample.
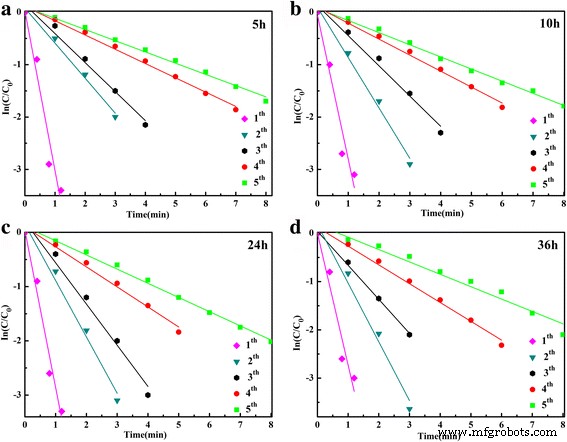
ln(At/A0) versus time for microspheres prepared at 5 h, 10 h, 24 h, and 36 h.
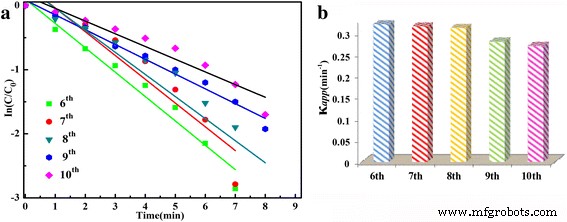
ln(At/A0) versus time for the sixth to tenth cycles of the 36‑h sample.
Comparative studies with bare SnO₂ and Ag nanoparticles (Figure S8–S9) show lower κapp values (1.24 and 1.16 min⁻¹, respectively), confirming that the synergistic interaction between Ag and SnO₂ nanosheets is responsible for the enhanced activity.
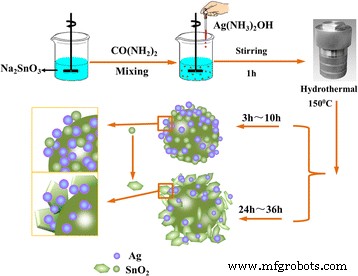
Schematic of the one‑pot synthesis of Ag‑decorated SnO₂ microspheres.
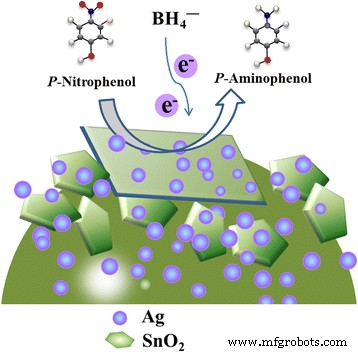
Illustration of the catalytic reduction of 4‑NP to 4‑AP over Ag‑decorated SnO₂ microspheres.
Conclusions
We have demonstrated a green, single‑step hydrothermal synthesis that produces Ag‑decorated SnO₂ microspheres featuring uniform ~5 nm Ag nanoparticles on SnO₂ nanosheet frameworks. The resulting catalyst achieves a record κnor of 6.20 min⁻¹ g⁻¹ L for 4‑NP reduction and retains high activity over ten cycles, underscoring its potential for large‑scale pollutant remediation and resource recovery. The methodology offers a versatile platform for fabricating other noble‑metal‑modified semiconductor composites.
Nanomaterials
- Fluorescent Nanomaterials: Advances, Synthesis, and Biomedical Applications
- Triphenylphosphine‑Derived Porous Polymer as a Robust Heterogeneous Catalyst for CO₂‑Driven Cycloaddition to Form Cyclic Carbonates
- One‑Pot Synthesis of Cu₂ZnSnSe₄ Nanoplates and Their Efficient Visible‑Light Photocatalytic Degradation of Rhodamine B
- Green Production of Copper Nanoparticles: Catalytic, Antibacterial, Cytotoxic, and Antioxidant Potentials
- Green Synthesis of Gold and Silver Nanoparticles with Platycodon Saponins from Platycodi Radix
- S‑Doped Sb₂O₃ Nanocrystals: Tailored pH‑Controlled Visible‑Light Photocatalysts for Efficient Organic Degradation
- Efficient One‑Pot Synthesis of Hierarchical Silicalite‑1/Ag Nanoparticle Catalyst for Rapid 4‑Nitrophenol Reduction
- Rapid One‑Pot Synthesis of Monodisperse CoFe₂O₄@Ag Core–Shell Nanoparticles with Exceptional Plasmonic and Magnetic Properties
- 5‑Minute Microwave‑Assisted Synthesis of Polydopamine‑Passivated Carbon Dots with 5 % Quantum Yield and 35 % Photothermal Efficiency for Cancer Therapy
- Efficient One-Step Green Synthesis of Multifunctional Gold Nanoparticles for Targeted Tumor Imaging and Therapy



