Triphenylphosphine‑Derived Porous Polymer as a Robust Heterogeneous Catalyst for CO₂‑Driven Cycloaddition to Form Cyclic Carbonates
Abstract
A novel triphenylphosphine‑based porous polymer (TPDB) with an exceptionally high Brunauer–Emmett–Teller (BET) surface area was constructed via Friedel–Crafts alkylation of triphenylphosphine and α‑dibromo‑p‑xylene. Subsequent post‑functionalization with 3‑bromo‑1‑propanol (BP) and triethanolamine (TEA) introduced hydroxyl groups into the framework, yielding TPDB‑BP‑TEA. Comprehensive characterization (FT‑IR, TG, SEM, EDS mapping, ICP‑MS, N₂ sorption) confirmed the successful grafting and high surface area retention. In a solvent‑free, metal‑free cycloaddition of CO₂ with epoxides, TPDB‑BP‑TEA achieved a 97 % yield and 100 % selectivity, maintaining activity over multiple cycles. Bromide ions, hydroxyl groups, and the porous architecture are identified as key contributors to the superior catalytic performance.
Background
Ionic liquids (ILs) have long attracted attention as versatile reaction media and catalysts owing to their negligible volatility, thermal robustness, exceptional solvation capabilities, and structural tunability [1,2,3]. By incorporating functional groups into IL cations or anions, one can tailor catalytic properties for specific transformations [4,5]. IL‑catalyzed cycloadditions, particularly CO₂ fixation reactions, have emerged as a promising strategy to convert the abundant, non‑toxic CO₂ feedstock into valuable cyclic carbonates [6–11]. Although ILs exhibit excellent activity in CO₂ cycloaddition under metal‑free, solvent‑free conditions, their homogeneous nature hampers catalyst recovery and product purification.
Porous materials with high surface area provide a platform for designing heterogeneous catalysts that combine the benefits of IL functionality with the practicality of solid supports [12–14]. Advances in metal‑organic frameworks (MOFs) [15,16], covalent organic frameworks (COFs) [17,18], and porous organic polymers [19,20] have broadened the toolkit for catalyst development. IL‑containing porous organic polymers are particularly attractive due to their low skeletal density, chemical stability, and the ability to embed diverse functional groups directly into the framework [21–23]. Previous work has immobilized imidazolium‑based ILs on mesoporous polymers, achieving respectable activity in CO₂ cycloaddition [24]; however, multistep IL modification often limits loading and leads to uneven distribution. Radical self‑polymerization approaches have yielded poly(ionic liquid)s with high activity and recyclability [25], yet simultaneously achieving high BET surface area and IL loading remains challenging. Thus, integrating IL motifs into a porous organic polymer with a robust, high‑surface‑area scaffold is still an unmet goal.
In this study, we report the synthesis of a triphenylphosphine‑derived ionic porous polymer featuring high surface area, extensive bromide ions, and abundant hydroxyl groups for the efficient cycloaddition of CO₂ with epoxides. TPDB was first prepared by Friedel–Crafts alkylation of triphenylphosphine and α‑dibromo‑p‑xylene in the presence of FeCl₃. Subsequent post‑functionalization with BP and TEA yielded TPDB‑BP‑TEA, which was thoroughly characterized by FT‑IR, TG, SEM, EDS mapping, ICP‑MS, and N₂ adsorption–desorption. Catalytic testing demonstrates that TPDB‑BP‑TEA serves as an excellent heterogeneous catalyst for CO₂ cycloaddition, offering high activity, selectivity, ease of recovery, and remarkable reusability.
Experimental
Materials and methods
All reagents were of analytical grade and used as received. Thermogravimetric analysis (TG) was performed on a STA409 instrument at a heating rate of 10 K min⁻¹ under N₂. Fourier‑transform infrared (FT‑IR) spectra were recorded on an Agilent Cary 660 FT‑IR spectrometer in the 4000–400 cm⁻¹ range with KBr disks. Scanning electron microscopy (SEM) images were captured on a SUPERSCAN SSX‑550 electron microscope (Shimadzu, Japan) at 20 kV. Elemental mapping was conducted using a Hitachi S‑4800 field‑emission SEM equipped with energy‑dispersive X‑ray spectrometry. N₂ sorption–desorption measurements were performed on a BELSORP‑MINI instrument at 77 K, and BET surface areas were calculated using the BET method; pore size distributions were derived via the BJH method. Phosphorus content was quantified by ICP‑MS (Agilent 7700). CHN elemental analysis was performed on a Vario EL cube analyzer.
Catalyst preparation
Synthesis of TPDB
TPDB was synthesized according to literature [26]. Triphenylphosphine (4 mmol, 1.05 g) and α‑dibromo‑p‑xylene (4 mmol, 1.06 g) were dissolved in 20 mL 1,2‑dichloroethane (DCE). Anhydrous FeCl₃ (16 mmol, 2.59 g) was added to catalyze the alkylation. The mixture was stirred at 45 °C for 5 h, then at 80 °C for an additional 48 h. The resulting brown gel was filtered, Soxhlet‑extracted with DCE and methanol for 24 h, and the cross‑linked polymer was dried at 60 °C under vacuum.
Synthesis of TPDB‑BP
TPDB (1 g) was dispersed in 15 mL acetonitrile, and 3‑bromo‑1‑propanol (0.8 g) was added. The mixture was heated at 80 °C for 24 h. The solid product TPDB‑BP was filtered, washed with acetonitrile three times, and dried at 60 °C under vacuum.
Synthesis of TPDB‑BP‑TEA
TPDB‑BP (1 g) was dispersed in 15 mL acetone, and triethanolamine (0.8 g) was added. The mixture was heated at 60 °C for 24 h. The resulting TPDB‑BP‑TEA was filtered, washed with acetone three times, and dried in vacuum at 60 °C for 12 h. ICP‑MS analysis revealed 3.7 wt % P in TPDB‑BP‑TEA, while CHN elemental analysis gave C 64.91 wt %, H 5.54 wt %, and N 1.65 wt %.
Catalytic test
The cycloaddition was carried out in a 25 mL stainless steel autoclave with magnetic stirring. In a typical run, 20 mmol propylene oxide (PO) and 0.1 g TPDB‑BP‑TEA were loaded. CO₂ was pressurized to 1 MPa and the temperature set to 120 °C. After 6 h, the reactor was cooled to ambient temperature, and the mixture was diluted with ethyl alcohol. The solid catalyst was filtered, and the filtrate was analyzed by GC (FID, DB‑wax SE‑54 30 m × 0.32 mm × 0.25 µm) using biphenyl as an internal standard to determine yield. GC spectra are shown in Additional file 1: Figures S1–S5.
Results and discussion
Synthesis and characterization of catalysts
Following the scheme depicted in Fig. 1, TPDB was prepared via Friedel–Crafts alkylation of PPh₃ with DB in the presence of FeCl₃. TPDB was then functionalized with BP to give TPDB‑BP, and further reaction with TEA produced TPDB‑BP‑TEA. The series of catalysts was thoroughly characterized. TG curves (Fig. 2a–c) show that TPDB remains stable up to ~300 °C; the BP and TEA modifications reduce stability slightly to ~250 °C. ICP‑MS confirms 3.7 wt % P in TPDB‑BP‑TEA, and CHN analysis reports C 64.91 wt %, H 5.54 wt %, N 1.65 wt %.
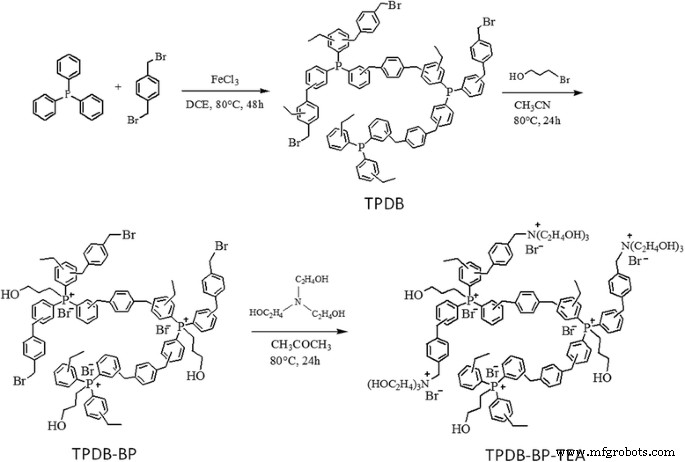
Synthesis of porous organic polymer TPDB‑BP‑TEA. First, triphenylphosphine (PPh₃) and α‑dibromo‑p‑xylene (DB) were reacted to form porous polymer (TPDB) through Friedel–Crafts alkylation with anhydrous FeCl₃ as a promoter. Then, the TPDB can be easily functionalized by 3‑bromo‑1‑propanol (BP) and triethanolamine (TEA), to afford functional porous polymer (TPDB‑BP‑TEA)
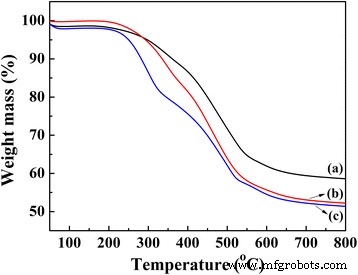
TG curves of (a) TPDB, (b) TPDB‑BP, and (c) TPDB‑BP‑TEA. TPDB was found to be stable up to 300 °C as evidenced by TG (curve a). After the modification with BP and TEA, the thermos stability of the obtained samples TPDB‑BP and TPDB‑BP‑TEA slightly decreased to 250 °C
FT‑IR spectra (Fig. 3) reveal characteristic bands of the PPh₃–C=C linkage (1674 cm⁻¹) and aromatic ring vibrations (1603–1438 cm⁻¹) in TPDB. After BP grafting, a broad –OH stretch appears at 3378 cm⁻¹, which intensifies to 3351 cm⁻¹ upon TEA addition, reflecting the increased hydroxyl content. New bands at 1062 and 1030 cm⁻¹ correspond to C–N and C–O vibrations of TEA, confirming successful functionalization. XPS spectra (Fig. 4) show the presence of P, C, N, Br, and O in TPDB‑BP‑TEA, corroborating the elemental analysis.
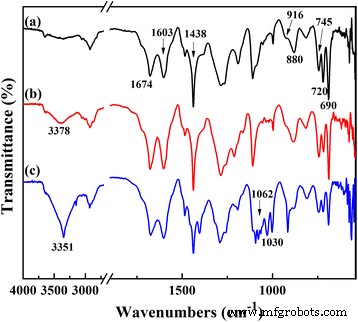
FT‑IR spectra of (a) TPDB, (b) TPDB‑BP, and (c) TPDB‑BP‑TEA. The distinct bands corresponding to the P–C=C (1674 cm⁻¹) in PPh₃ and aromatic ring stretching vibrations (1603–1438 cm⁻¹), as well as to the stretching vibrations of C–H in aromatic ring (916, 880, 745, 720, and 690 cm⁻¹) indicates the presence of both PPh₃ and DB groups in TPDB. TPDB‑BP shows a moderate intensity broad absorption band at 3378 cm⁻¹, which is corresponding to the stretching vibration of the –OH. After further modified by TEA, the intensity of –OH vibration at 3351 cm⁻¹ for TPDB‑BP‑TEA significantly increased. Besides, the new bands appeared at 1062 and 1030 cm⁻¹ are assigned to the stretching vibrations of C–N and C–O in TEA, respectively
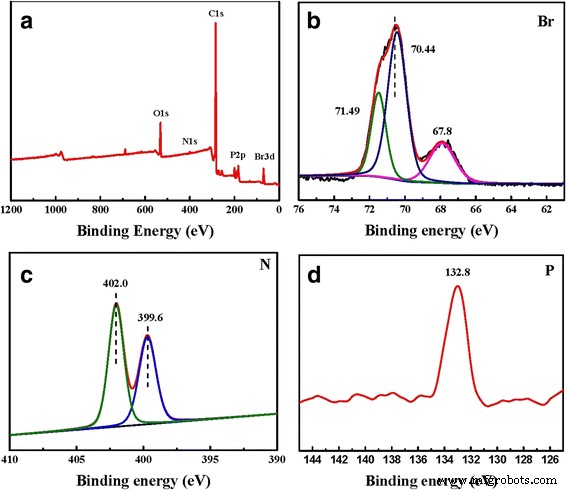
a Wide XPS spectrum, b Br spectrum, c N spectrum, and d P spectrum of TPDB‑BP‑TEA. The wide XPS spectra in Fig. 3 indicate the presence of P, C, N, Br, and O elements on TPDB‑BP‑TEA
SEM and EDS mapping images (Fig. 5) show that TPDB possesses an amorphous morphology. BP functionalization preserves this amorphous character but introduces a nanoscale hollow structure. Subsequent TEA grafting slightly roughens the surface and creates small agglomerated blocks. EDS mapping confirms homogeneous distribution of P and Br in TPDB, with an increased Br signal after BP modification. The appearance of O after BP grafting and N after TEA addition further validates successful functionalization.
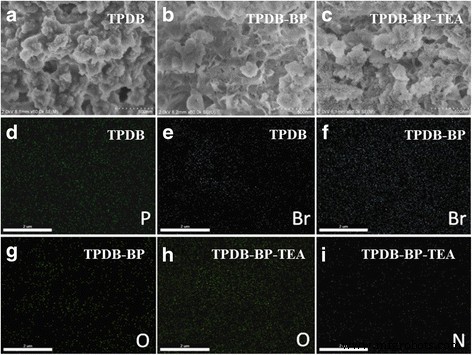
SEM and EDS mapping images of a, d, and e TPDB, b, f, and g TPDB‑BP, and c, h, and i TPDB‑BP‑TEA. TPDB, TPDB‑BO, and TPDB‑BP‑TEA all show amorphous morphology (a). After the modification with BP and TEA, TPDB‑BP‑TEA shows no main changes in the structure, but its surface became rough with some agglomerated blocks (c). EDS mapping image validates the homogeneous distribution of P and Br elements in the polymer framework of TPDB (d, e). The amount of Br increased obviously (f), and a new element O was observed (g) after the modification of TPDB with BP. After the further modification of TPDB‑BP with TEA, a new element N was observed (i), and the amount of O element increased significantly in the image of TPDB‑BP‑TEA (h). These images confirm the successful immobilization of BP and TEA on the TPDB framework
BET surface areas and pore size distributions of TPDB and TPDB‑BP‑TEA were derived from N₂ adsorption–desorption isotherms at 77 K (Fig. 6). TPDB exhibits a high BET surface area of 493.15 m² g⁻¹, a pore volume of 0.54 cm³ g⁻¹, and an average pore diameter of 4.38 nm. After the two‑step functionalization, the surface area and pore volume decrease to 227.12 m² g⁻¹ and 0.41 cm³ g⁻¹, respectively, reflecting a modest loss of porosity while preserving the overall framework.
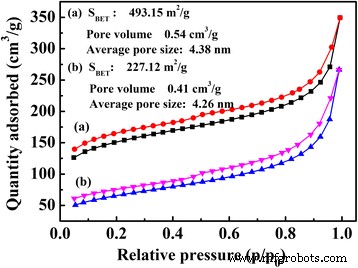
Nitrogen adsorption–desorption isotherms of (a) TPDB and (b) TPDB‑BP‑TEA. BET surface areas and pore size distributions of the polymers TPDB and TPDB‑BP‑TEA were measured by analyzing N₂ adsorption and desorption isotherms at 77 K. The steep rise in the high P/P₀ region indicates that the material consists of micropores and mesopores. TPDB presents a high BET surface area of 493.15 m² g⁻¹, pore volume of 0.54 cm³ g⁻¹, and average pore size of 4.38 nm. After the two‑step modification, the BET surface area and pore volume decreased to 227.12 m² g⁻¹ and 0.41 cm³ g⁻¹, respectively
Catalytic performance of catalysts
Initial screening (Table 1) revealed that no cyclic carbonate formed without a catalyst. TPDB alone gave 44 % yield with 59 % selectivity, producing 1,2‑propandiol as the major by‑product. TPDB‑BP improved yield to 51 % and selectivity to 93 %. TPDB‑BP‑TEA delivered an outstanding 97 % yield with 100 % selectivity. The presence of abundant Br⁻ ions and –OH groups, confirmed by EDS mapping, is consistent with literature reports that Br⁻ acts as the primary catalytic center while –OH groups facilitate epoxide ring opening via hydrogen bonding [27–33]. The high surface area and mesoporous nature further enhance mass transfer, contributing to the superior performance.
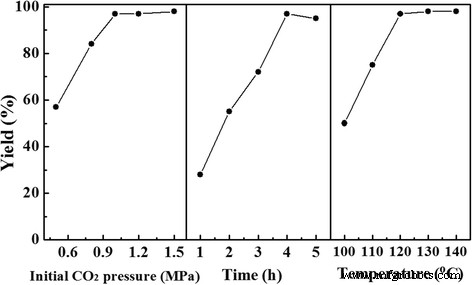
Influence of reaction parameters on the cycloaddition reaction of CO₂ with propylene oxide. The yield remarkably increased from 58 to 97 % when the CO₂ pressure was increased from 0.6 to 1.0 MPa and after that the yield maintained constant. The catalytic reaction finished in 4 h, whereas longer reaction time caused a slightly decrease of yield. The optimal reaction temperature was 120 °C
TPDB‑BP‑TEA also catalyzes a range of epoxides (Table 2), including epichlorohydrin, allyl glycidyl ether, and styrene oxide, with high efficiency. Internal epoxides, such as cyclohexene oxide, give lower conversions (74 % yield, 59 % selectivity) due to steric constraints in the porous matrix.
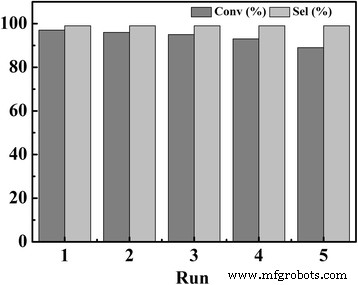
Catalytic reusability of TPDB‑BP‑TEA for cycloaddition of CO₂ with PO. As a solid catalyst, TPDB‑BP‑TEA is recovered readily by filtration or centrifugation and well maintain its activity in the five‑run recycling test under mild conditions
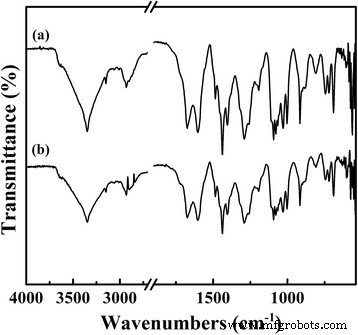
FT‑IR spectra of (a) fresh TPDB‑BP‑TEA and (b) reused TPDB‑BP‑TEA. FT‑IR spectrum of the recovered catalyst suggests the well‑preserved textural properties relative to the fresh one, accounting for its well recyclability
Conclusions
We have developed a triphenylphosphine‑derived porous polymer with a large surface area, dense ionic sites, and integrated –OH functionality. TPDB‑BP‑TEA functions as a highly efficient, metal‑free, solvent‑free heterogeneous catalyst for the CO₂ cycloaddition to epoxides, delivering high yields and selectivities across diverse substrates. The catalyst can be easily separated and reused with negligible loss of activity. The synergy of abundant Br⁻ ions, hydroxyl groups, a mesoporous scaffold, and high surface area underpins the exceptional performance, offering a promising platform for CO₂ fixation and other catalytic transformations.
Nanomaterials
- Green One‑Pot Synthesis of Ag‑Decorated SnO₂ Microspheres: A Highly Reusable Catalyst for 4‑Nitrophenol Reduction
- Optimized Post‑Treatment Strategy for Monodisperse FePt–Fe3O4 Binary Nanoparticles
- Optimizing Upconversion Luminescence in BaYF5:Er³⁺/Yb³⁺ Nanocrystals Through Controlled Solvothermal Synthesis
- Green Synthesis of Gold and Silver Nanoparticles with Platycodon Saponins from Platycodi Radix
- S‑Doped Sb₂O₃ Nanocrystals: Tailored pH‑Controlled Visible‑Light Photocatalysts for Efficient Organic Degradation
- Rapid, One‑Pot Synthesis of Nitrogen‑Doped Microporous Carbon Spheres for Superior Symmetric Supercapacitor Performance
- Ultra‑High Capacity Supercapacitor Electrodes from One‑Step Hydrothermal Synthesis of Chlorine‑Doped Carbonated Cobalt Hydroxide Nanowires on Nickel Foam
- Nitrogen‑Doped Porous Carbon Networks Derived from Graphitic Carbon Nitride for High‑Performance Oxygen Reduction
- Carbon‑Coated Molybdenum Phosphide Nanoparticles: A Low‑Cost Catalyst with 131 mV Overpotential for Alkaline Hydrogen Evolution
- How 5G Will Revolutionize IoT and Industrial IoT



