Rapid, One‑Pot Synthesis of Nitrogen‑Doped Microporous Carbon Spheres for Superior Symmetric Supercapacitor Performance
Abstract
We present a concise, one‑step hydrothermal route to synthesize nitrogen‑doped microporous carbon spheres (NMCSs) via carbonization and KOH activation of phenol‑formaldehyde (PF) resin spheres. By tuning the ethanol/water ratio and carbonization temperature, we obtain NMCSs with a specific surface area of 1517 m2 g−1, a pore volume of 0.8 cm3 g−1, and a nitrogen content of 2.6 at.% (XPS). When employed as electrodes, these NMCSs deliver an outstanding specific capacitance of 416 F g−1 at 0.2 A g−1 and retain 96.9 % after 10,000 cycles. A symmetric supercapacitor (SSD) assembled with a PVA/KOH gel electrolyte achieves 60.6 F g−1 at 1 A g−1, a peak energy density of 21.5 Wh kg−1 at 800 W kg−1, and 13.3 Wh kg−1 even at 16 kW kg−1. These results demonstrate a rapid, scalable route to high‑performance electrode materials for next‑generation energy storage.
Background
Global energy security is threatened by the rapid depletion of fossil fuels and rising climate‑change impacts. Sustainable, high‑power, high‑energy, and long‑lived storage technologies are therefore critical. Supercapacitors, with their fast charge/discharge cycles, high power density, and excellent cycle life, are prime candidates for such applications. They are traditionally classified as electrical double‑layer capacitors (EDLCs) or pseudocapacitors. While EDLCs benefit from high power and long life, their energy density is limited by the available surface area. Pseudocapacitors can deliver higher energy but often suffer from lower power and shorter cycle life. Bridging this gap requires materials that combine high surface area with redox‑active sites.
Carbon spheres (CSs) are attractive due to their uniform geometry, structural stability, and tunable porosity. Heteroatom doping, especially nitrogen, introduces pseudocapacitance and improves conductivity and wettability. Phenolic resins are inexpensive, thermally stable precursors that can be converted into carbon via simple thermal treatments. The Stöber method and its extensions have been widely used to produce uniform CSs, yet many protocols are time‑consuming and yield limited performance.
Here we report a rapid, one‑pot hydrothermal synthesis of nitrogen‑doped microporous carbon spheres (NMCSs) that bypasses lengthy aging steps. The resulting NMCSs exhibit high surface area, optimal nitrogen content, and superior electrochemical performance, establishing a scalable route to high‑performance symmetric supercapacitors.
Methods
Materials
Phenol, formaldehyde (37 wt %), ammonia solution (25 wt %), anhydrous ethanol, polyvinyl alcohol (PVA), and KOH were purchased from Sinopharm Chemical Reagent Co. Ltd. Triblock copolymer Pluronic F108 (MW = 14,600, PEO132‑PPO50‑PEO132) and PTFE (60 wt %) were obtained from Aladdin. All reagents were used as received.
Synthesis of NMCSs
In a typical synthesis, 0.5 g of F108 was dissolved in 80 mL of ethanol/water (4.3:1) at room temperature for 10 min. Then 3 mL of ammonia, 1.2 g phenol, and 4.5 mL formaldehyde were added and stirred for 30 min. The mixture was transferred to a 100 mL Teflon‑lined autoclave and heated at 170 °C for 6 h, producing PF resin spheres. After washing with water and ethanol and drying at 80 °C for 12 h, the powders were calcined at 500–800 °C (3 h) and activated with KOH (mass ratio 1:2) at 700 °C for 1 h under N2 flow, yielding NMCSs‑x (x = carbonization temperature).
Characterization
SEM (Nova NanoSEM230) and TEM (Tecnai G2 F20) assessed morphology. XRD (Siemens D500) with Cu Kα radiation (λ = 0.15056 nm) examined crystallinity. XPS (ESCALAB 250Xi) quantified elemental composition. N2 sorption (ASAP 2020) measured surface area (BET) and pore size (BJH). Electrochemical tests used a CHI660E workstation: CV, GCD, and EIS in a three‑electrode cell (6 M KOH, Pt foil, Hg/HgO). Symmetric cells were assembled with PVA/KOH gel electrolyte.
Electrochemical Measurement
Working electrodes comprised 80 % NMCSs, 10 % PTFE, and 10 % acetylene black in ethanol, coated on nickel foam (≈3 mg cm−2). The SSDs were formed by immersing two NMCSs‑600 electrodes in the PVA/KOH gel for 5 min and sealing without encapsulation. Capacitance, energy, and power were calculated from GCD curves using standard equations (1–4).
Results and Discussion
Fabrication of NMCSs
The synthesis strategy is illustrated in Scheme 1. Pluronic F108 acts as a soft template, forming micelles in the ethanol/water mixture. Phenol and formaldehyde polymerize in the presence of ammonia, which simultaneously catalyzes the reaction and supplies nitrogen. The hydrothermal step at 170 °C for 6 h yields uniform PF resin spheres. Subsequent carbonization and KOH activation transform them into nitrogen‑doped microporous spheres (NMCSs). The short reaction time and single‑step procedure mark a significant improvement over conventional Stöber or soft‑template methods.
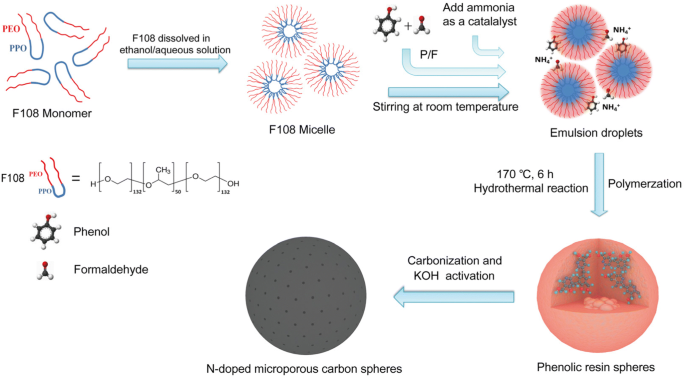
Schematic illustration of the fabrication process of NMCSs
Morphology and Structure
SEM images (Fig. 1a–d) reveal that the ethanol/water ratio critically influences particle uniformity. A 1:1 ratio yields smooth, well‑dispersed spheres (1.2–2 µm diameter). Higher ethanol content promotes aggregation due to increased cross‑linking density. TEM (Fig. 1e) confirms the spherical morphology, while HR‑TEM (Fig. 1f) shows a well‑defined microporous network essential for high capacitance.

SEM images of the NMCSs samples synthesized at different ethanol/water volume ratios of (a) 7:1, (b) 4.3:1, (c) 3:1 and (d) 1:1, (e) TEM and (f) HR‑TEM images of the NMCSs-600
PXRD patterns (Fig. 2a) show broad peaks at ~25° and 44°, characteristic of amorphous carbon. XPS (Fig. 2b) confirms the presence of C, N, and O. The nitrogen content peaks at 2.6 at.% for NMCSs‑600, decreasing to 0.9 at.% at 800 °C due to thermal loss of nitrogen species. High‑resolution N 1 s spectra (Fig. 2c–f) identify pyridinic (N‑6), pyrrolic (N‑5), graphitic (N‑Q), and oxidized (N‑X) nitrogen. The optimal distribution of N‑5 (31.7 %) and N‑6 (10.7 %) in NMCSs‑600 enhances pseudocapacitance, while increasing N‑Q improves conductivity.
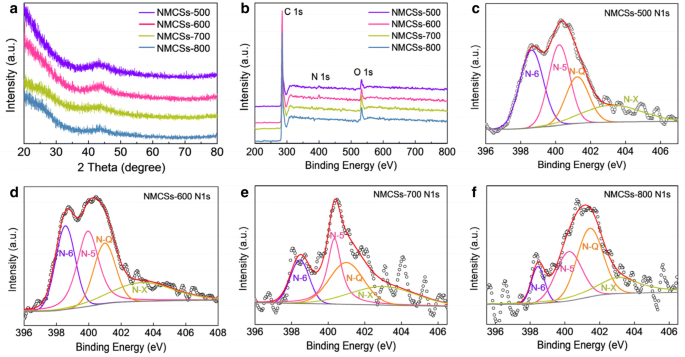
(a) XRD patterns and (b) XPS survey spectra of the as‑prepared NMCSs materials, and the high‑resolution N 1 s spectra at different carbonization temperatures of (c) 500 °C, (d) 600 °C, (e) 700 °C and (f) 800 °C
Composition Analysis
Ammonia not only catalyzes polymerization but also incorporates nitrogen into the carbon framework. XPS elemental analysis (Table 1) confirms successful doping. The decrease of pyridinic N with temperature is expected, while graphitic N increases, improving electronic conductivity.
Nitrogen Adsorption Studies
All NMCSs exhibit Type I isotherms (Fig. 3a) with steep uptake at low P/P0, indicative of abundant microporosity. The pore size distribution (Fig. 3b) shows a dominant range of 0.7–2 nm, resulting from F108 decomposition and KOH activation. NMCSs‑600 achieves the highest BET surface area (1517 m2 g−1) and pore volume (0.8 cm3 g−1), which is ideal for electrolyte access and ion transport. Higher temperatures (>700 °C) collapse pores, reducing surface area.
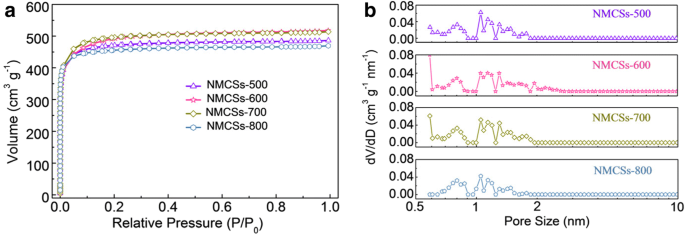
(a) Nitrogen adsorption/desorption isotherms and (b) pore size distribution curves of NMCSs materials
Electrochemical Performance of the NMCSs Electrodes
CV curves (Fig. 4a) of all samples show quasi‑rectangular shapes at 10 mV s−1, with reversible humps between −0.8 and −0.2 V attributable to N/O redox. NMCSs‑600 exhibits the most pronounced hump, correlating with its high nitrogen content. The CV shape remains intact up to 100 mV s−1 (Fig. 4b), demonstrating excellent rate capability thanks to the microporous network.
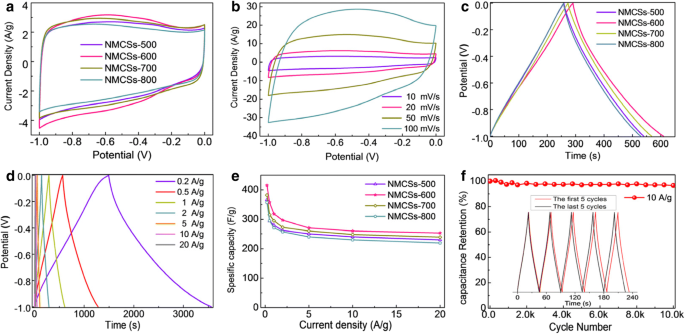
(a) CV curves of NMCSs electrodes at scan rate of 10 mV s−1, (b) CV curves of the NMCSs‑600 electrode at different scan rates from 10 to 100 mV s−1, (c) GCD curves of NMCSs electrodes at current density of 1 A g−1, (d) GCD curves of the NMCSs‑600 electrode at different current densities, (e) Specific capacitance of NMCSs electrodes as a function of current densities, and (f) Cycling performance of the NMCSs‑600 electrode at current density of 10 A g−1 for 10,000 cycles and the inset shows the GCD curves of the first five and last five cycles, with a three‑electrodes system in 6 M KOH aqueous solution
GCD at 1 A g−1 (Fig. 4c) yields 318 F g−1 for NMCSs‑600, surpassing other samples. The high surface area and optimal nitrogen distribution synergistically enhance both electric double‑layer and pseudocapacitive contributions. Rate studies (Fig. 4d–e) reveal 253 F g−1 at 20 A g−1 (61 % retention), underscoring excellent kinetics. Long‑term cycling (Fig. 4f) shows 96.9 % capacitance retention after 10,000 cycles at 10 A g−1.
EIS (Fig. 5a) indicates low equivalent series resistance (<0.5 Ω) and charge‑transfer resistance (<1 Ω), while the nearly vertical low‑frequency line confirms ideal capacitive behavior and minimal diffusion limitation.
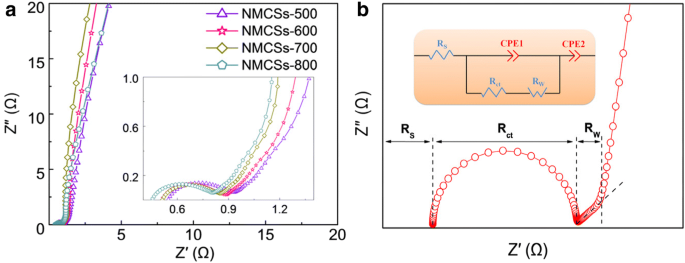
(a) Nyquist plots of NMCSs materials and the inset shows the magnify plots at high frequency range and (b) a typical Nyquist plot of EDLCs and the equivalent circuit model
Electrochemical Performance of the NMCSs‑600‑Based SSDs
Symmetric cells with a PVA/KOH gel were evaluated in a two‑electrode configuration. CV (Fig. 6a) shows a stable rectangular shape up to 1.6 V; the 1.6 V window was adopted for all subsequent tests. At 1 A g−1, the SSD delivers 60.6 F g−1 and retains 37.5 F g−1 at 20 A g−1 (Fig. 6d). EIS (Fig. 6e) confirms negligible interfacial resistance (0.83 Ω) and efficient ion transport. Ragone analysis (Fig. 6f) highlights a maximum energy density of 21.5 Wh kg−1 at 800 W kg−1, decreasing to 13.3 Wh kg−1 at 16 kW kg−1, outperforming many reported carbon‑sphere devices. Two SSDs in series successfully powered a red LED for 60 s, demonstrating practical viability.
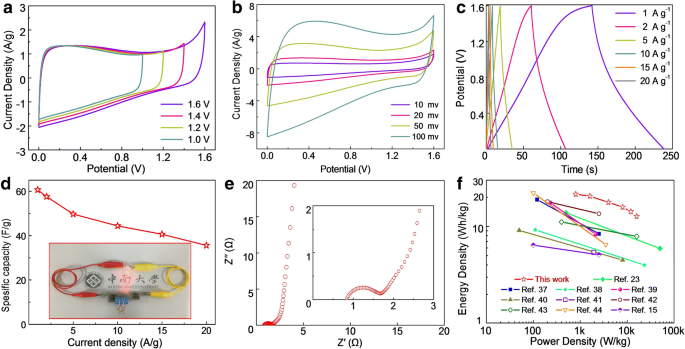
The electrochemical characteristics of the assembled SSDs based on the NMCSs‑600 materials using PVA/KOH as the gel electrolyte in two‑electrode system. a CV curves of the SSD in different voltage windows from 1 to 1.6 V at the scan rate of 20 mV s−1. b CV curves of the SSD at various scan rates within a voltage window of 1.6 V. c GCD curves at different current densities. d The gravimetric capacitance of the SSD as a function of current density, the inset image shows a commercial red LED powered by two SSDs in series. e Nyquist plot of the SSD, the inset gives the magnify plot for high frequency range. f Ragone plots of the SSD and the other carbon spheres based symmetric supercapacitors
Conclusions
We have developed a rapid, one‑pot hydrothermal route to nitrogen‑doped microporous carbon spheres (NMCSs) using phenol‑formaldehyde resin and Pluronic F108. The short 6‑h synthesis, combined with KOH activation, produces NMCSs‑600 with 1517 m2 g−1 surface area, 0.8 cm3 g−1 pore volume, and 2.6 at.% nitrogen. As supercapacitor electrodes, NMCSs‑600 deliver 416 F g−1 at 0.2 A g−1 and 96.9 % capacitance retention after 10,000 cycles. In symmetric cells, the material achieves 60.6 F g−1 at 1 A g−1 and a peak energy density of 21.5 Wh kg−1 at 800 W kg−1. The synthesis simplicity, short reaction time, and high electrochemical performance establish NMCSs as promising candidates for high‑energy, high‑power energy storage devices.
Abbreviations
- NMCSs:
Nitrogen‑doped microporous carbon spheres
- EDLCs:
Electrical double‑layer capacitors
- CSs:
Carbon spheres
- SSDs:
Symmetric supercapacitor devices
- CV:
Cyclic voltammetry
- GCD:
Galvanostatic charge/discharge
- EIS:
Electrochemical impedance spectroscopy
Nanomaterials
- High‑Performance Na4Mn9O18/Carbon Nanotube Cathodes for Aqueous Sodium‑Ion Batteries
- Enhanced Supercapacitor Performance via Polyaniline‑Coated Nitrogen‑Doped Ordered Mesoporous Carbon Composites
- Efficient Humate-Driven Co-Precipitation/Calcination Synthesis of MoS₂/C Nanocomposites for Superior Lithium‑Ion Battery Performance
- 5‑Minute Microwave‑Assisted Synthesis of Polydopamine‑Passivated Carbon Dots with 5 % Quantum Yield and 35 % Photothermal Efficiency for Cancer Therapy
- Ultra‑High Capacity Supercapacitor Electrodes from One‑Step Hydrothermal Synthesis of Chlorine‑Doped Carbonated Cobalt Hydroxide Nanowires on Nickel Foam
- High‑Performance Perovskite Photodetector via Trilayer EA/TiO₂ Architecture
- 3D Graphitic Carbon Nitride Nanowire Scaffold Boosts Flexibility and Capacitance in Solid‑State Supercapacitors
- One‑Step Synthesis of Nitrogen‑Doped Hydrophilic Mesoporous Carbon Spheres from Chitosan for Enhanced Hydroxycamptothecin Delivery
- Innovative, Cost‑Effective Fabrication of Flexible rGO/MWCNT Films for High‑Performance Symmetric Supercapacitors
- DISPAL® S220 AM – High‑Stiffness Aluminum Alloy for Precision Additive Manufacturing



