High‑Performance Perovskite Photodetector via Trilayer EA/TiO₂ Architecture
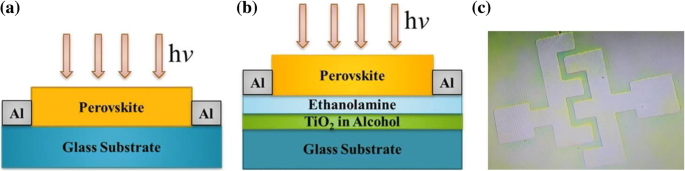
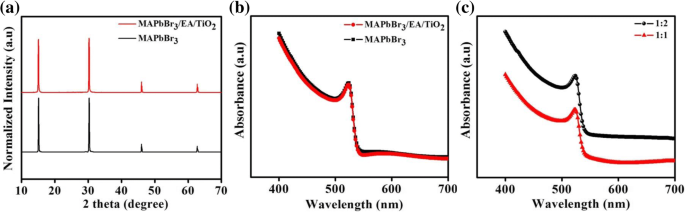
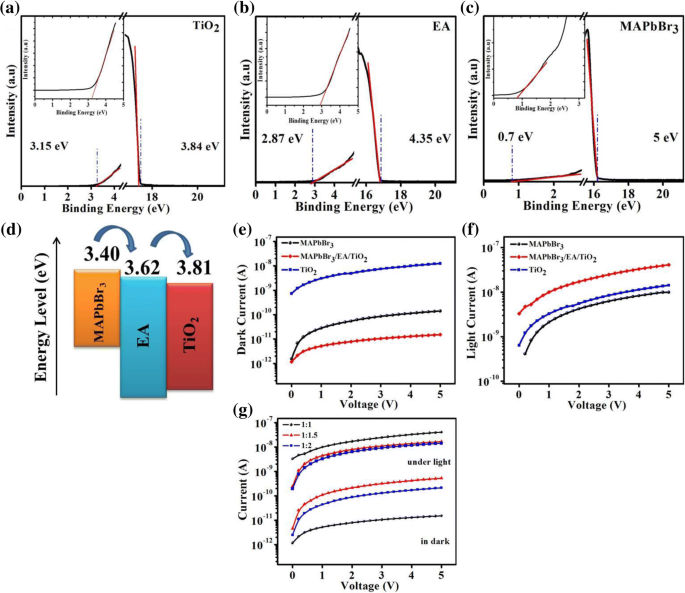
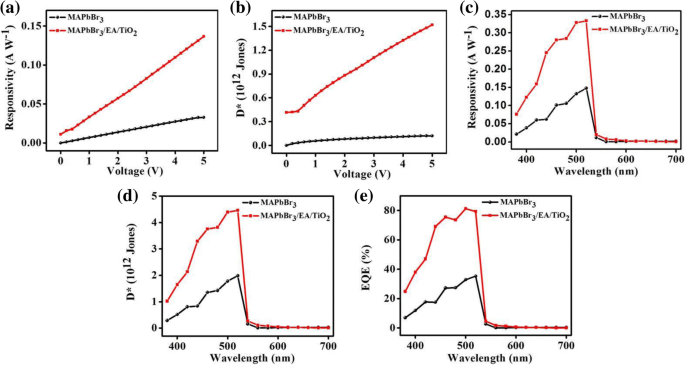
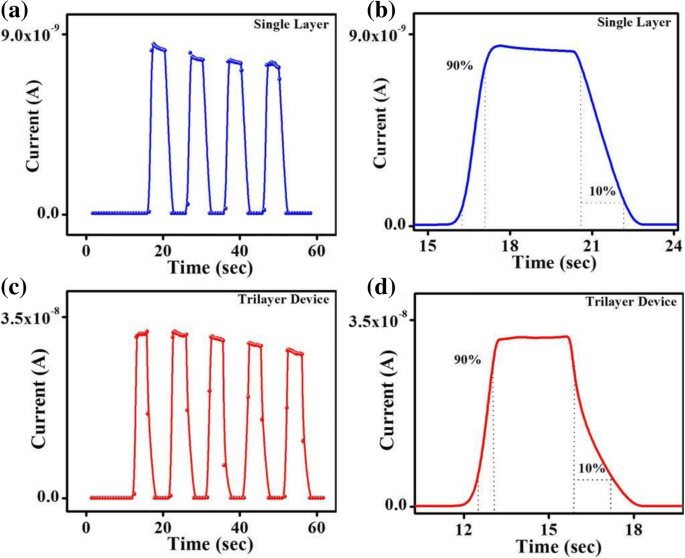
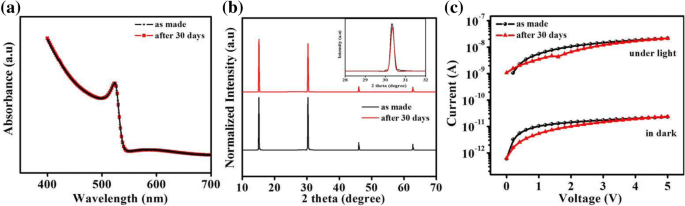
Lead‑halide perovskites are celebrated for their exceptional optical absorption, long carrier lifetimes, and high diffusion lengths, making them ideal for photodetectors. Traditional metal‑oxide, metal‑sulfide, and 2D‑material devices deliver respectable responsivity but often suffer from high dark currents, sluggish response, limited on‑off ratios, and poor long‑term stability. Here we introduce a lateral trilayer architecture—CH₃NH₃PbBr₃/ethanolamine/TiO₂ (mixed in ethanol)—that markedly improves performance. The ethanolamine interlayer facilitates efficient electron extraction and suppresses recombination. This design achieves a dark current of 1.5 × 10⁻¹¹ A, an on‑off ratio of 2700, a detectivity of 1.51 × 10¹² Jones, a responsivity of 0.13 A W⁻¹, and excellent environmental stability, outperforming conventional single‑layer devices. This work provides a clear pathway for elevating metal‑halide perovskite photodetector performance. Photodetectors underpin a wide spectrum of technologies—from optical communication and biomedical sensing to environmental monitoring. In recent years, organometallic lead‑halide perovskites have emerged as a front‑line material owing to their high absorption coefficients, long carrier lifetimes, and extended diffusion lengths, all of which are critical for efficient photodetection. Vertical perovskite photodetectors have dominated the field, yet they often exhibit significant dark currents (≈1.5 nA) and limited responsivity. Lateral architectures, in contrast, offer reduced conduction losses and simpler, lower‑cost fabrication. Single‑layer perovskite photodetectors, however, typically display low on‑off ratios, elevated dark currents, and electrical instability. Prior efforts—such as incorporating organic polymers or modifying heterojunction interfaces—have mitigated some of these issues but still leave room for improvement. The core challenge lies in engineering an interfacial layer that aligns energy levels, reduces recombination, and improves carrier transport. In this study, we present a lateral trilayer device featuring an ethanolamine (EA) dipole layer between the perovskite and TiO₂. EA reduces the energy barrier mismatch, enhances electron extraction, and suppresses recombination, resulting in a device that exhibits markedly superior performance metrics. Single‑layer and trilayer photodetectors were fabricated on glass substrates (Fig. 1a, b). The process began with spin‑coating alcoholic TiO₂ (mixed 1:16 v/v with ethanol) followed by deposition of the EA layer, then 60 nm Al electrodes (channel width = 2000 µm, length = 30 µm). Finally, a 60‑nm MAPbBr₃ film was applied. Figure 1c displays the assembled trilayer device. a The single‑layer device. b The trilayer device. c Optical image of trilayer device X‑ray diffraction confirmed the phase purity of MAPbBr₃ in both single‑layer and trilayer films, with characteristic peaks at 15.16°, 30.32°, 46.04°, and 62.76°. No secondary phases (e.g., PbBr₂ or TiO₂) were detected in the trilayer stack. UV–vis absorption spectra (Fig. 2b) showed identical profiles for the two device types, with a bandgap of 2.3 eV and negligible absorption from the TiO₂ or EA layers. a X‑ray diffraction patterns of the MAPbBr₃ film and the MAPbBr₃/EA/TiO₂ trilayer film. b MAPbBr₃ film and MAPbBr₃/EA/TiO₂ trilayer film absorption spectra. c Absorption spectra at different ratios of PbBr₂ SEM imaging revealed that high‑temperature annealing (>100 °C) introduces pinholes and cracks, which act as recombination centers. Optimizing the perovskite annealing to 75 °C for 10 min produced a dense, crack‑free film with large grains (Fig. 3b), thereby enhancing carrier separation and transport. SEM image of fabricated MAPbBr₃ film treated a at 100 °C and b at 75 °C UPS measurements established the Fermi levels and conduction band maxima of the three layers: alcoholic TiO₂ (E_F = 3.84 eV, CBM = 3.81 eV), EA (E_F = 4.35 eV, CBM = 3.62 eV), and MAPbBr₃ (E_F = 5 eV, CBM = 3.4 eV). The resulting band diagram (Fig. 4d) shows that photo‑generated electrons flow from MAPbBr₃ into EA and subsequently into TiO₂, while holes remain in the perovskite, effectively suppressing recombination. The UPS (ultraviolet photoelectron spectra) of a TiO₂ film. b Ethanolamine film. c MAPbBr₃ film. d Energy band diagram. I–V characteristics of photodetector devices at optimal ratio (1:1): e under dark, f under the light with intensity of 0.50 mW cm⁻². g I–V characteristics of trilayer photodetector at different ratios of perovskite The electrical response (Fig. 4e,f) demonstrates that the trilayer device achieves a dark current of 1.51 × 10⁻¹¹ A and a photocurrent of 4.09 × 10⁻⁸ A—substantially lower dark current and higher photocurrent compared to the single‑layer MAPbBr₃ (dark: 1.41 × 10⁻¹⁰ A; photocurrent: 9.95 × 10⁻⁹ A) and TiO₂‑based (dark: 1.24 × 10⁻⁸ A) controls. The reduced recombination at the MAPbBr₃/EA interface is the key to this improvement. The optimal perovskite to EA ratio was found to be 1:1. Responsivity (R) and detectivity (D*) were calculated using Eqs. (1) and (2). The trilayer device exhibits R = 0.13 A W⁻¹ and D* = 1.51 × 10¹² Jones under 0.5 mW cm⁻² illumination and 5 V bias, whereas the single‑layer device records R = 0.03 A W⁻¹ and D* = 1.19 × 10¹¹ Jones. Spectral measurements (Fig. 5c,d) show peak responsivity of 0.33 A W⁻¹ and detectivity of 4.46 × 10¹² Jones at 520 nm for the trilayer device, with an external quantum efficiency (EQE) of up to 80 % (Fig. 5e). a Responsivity and (b) detectivity of single and trilayer devices at different applied voltage under light intensity of 0.5 mW cm⁻². c Spectral responsivity, d spectral detectivity, and (e) EQE spectra of two photodetectors under light illumination of 10.6 µW cm⁻² and a bias of 5 V Signal‑to‑noise ratio (SNR) and linear dynamic range (LDR) further highlight the device’s superiority. The trilayer detector achieves SNR = 2700 and LDR = 68.6 dB, surpassing the single‑layer’s SNR = 69 and LDR = 36.9 dB. On‑off switching experiments (Fig. 6) show that the trilayer device reaches a stable photocurrent within 0.49 s and decays in 1.17 s, whereas the single‑layer device exhibits slower rise (0.72 s) and decay (1.72 s) times. The rapid response is attributed to efficient carrier separation across the heterojunction. On/off switching characteristics of a, b single‑layer (MAPbBr₃) photodetector. c, d Trilayer photodetector (under light intensity of 0.5 mW cm⁻² and applied bias of 5 V) Environmental stability was assessed over 30 days in 30–40 % relative humidity. XRD and absorption spectra remained unchanged (Fig. 7a,b), and I–V characteristics (Fig. 7c) showed negligible drift, underscoring the robustness of the trilayer architecture. a Visible absorption spectra. b XRD spectra. c I–V curves of the trilayer photodetector as made and after 30 days in ambient environment (under the white light intensity of 0.5 mW cm⁻²) In summary, the perovskite/EA/TiO₂ trilayer device delivers D* = 1.51 × 10¹² Jones, on‑off ratio = 2700, R = 0.13 A W⁻¹, rise/decay times of 0.49/1.17 s, LDR = 68.6 dB, and EQE up to 80 %. The engineered heterojunction and EA interlayer effectively suppress carrier recombination, leading to superior performance relative to single‑crystal and 2D‑material photodetectors. This strategy offers a promising route for next‑generation, high‑performance perovskite photodetectors. PbBr₂ and CH₃NH₃Br were sourced from Xi’an Polymer Light Technology Corporation. DMF, DMSO, ethanolamine, and 2‑methoxyethanol were purchased from Alfa Aesar. TiO₂ (10 % sol‑gel in ethanol, 30 nm particles) came from InnoChem. Glass slides (SAIL BRAND) served as substrates. All reagents were used without further purification. TiO₂ was diluted 1:16 (v/v) with ethanol and stirred for 1 h at room temperature. The resulting alcoholic TiO₂ was employed as the electron‑transport layer to overcome the carrier‑blocking behavior of dense TiO₂ films. A 3 wt % EA solution was prepared by mixing EA with 2‑methoxyethanol, following established protocols. A 1 M CH₃NH₃PbBr₃ solution was formed by reacting CH₃NH₃Br and PbBr₂ (1:1 w/w) in a 1:4 (v/v) DMSO/DMF mixture, then stirring at 70 °C overnight. Glass substrates were sequentially cleaned with detergent, DI water, IPA, acetone (20 min each), dried under N₂, and finished with O₂ plasma (15 min). Alcoholic TiO₂ was spin‑coated (4000 rpm, 30 s), annealed at 150 °C for 30 min. EA in 2‑methoxyethanol was spin‑coated (3000 rpm, 40 s) and annealed at 130 °C for 10 min. 60 nm Al electrodes (channel width = 2000 µm, length = 30 µm) were thermally evaporated through a shadow mask. MAPbBr₃ was deposited via a two‑step spin‑coating: 1000 rpm (10 s) → 5000 rpm (30 s), with a toluene drop at 22 s. Electrical measurements used a Keithley 4200 under ambient, 300 K conditions. White‑light illumination was provided by an XZ‑150WA cold light source; intensity calibrated with a silicon photodiode. Monochromatic light came from a Newport Oriel 200 source. SEM (Hitachi S‑4800), UV–vis absorption (JASCO V‑570), UPS (KRATOS AXIS ULTRA DLD, He I 21.22 eV), and XRD (Rifaku D/MAX‑2004, Cu Kα, λ = 1.54178 Å) characterized the films. We have engineered a lateral perovskite/EA/TiO₂ trilayer photodetector that achieves record‑breaking metrics: D* = 1.51 × 10¹² Jones, on‑off ratio = 2700, R = 0.13 A W⁻¹, rise/decay times of 0.49/1.17 s, LDR = 68.6 dB, and EQE = 80 %. The ethanolamine interlayer reduces the energy barrier, promotes efficient electron extraction, and suppresses recombination, while the alcoholic TiO₂ provides a robust electron‑transport channel. This architecture delivers superior performance compared to single‑crystal, 2D‑material, and conventional metal‑oxide devices, and demonstrates long‑term stability under ambient conditions. The study underscores the critical role of heterojunction engineering in advancing perovskite photodetector technology. Ethanolamine External quantum efficiency Linear dynamic range Phenyl‑C61‑butyric acid methyl ester Polymethylmethacrylate Scanning electron microscope Signal‑to‑noise ratio Ultraviolet photoelectron spectroscopy X‑ray diffractionAbstract
Introduction
Results and Discussion

Methods/Experimental
Material Preparation
TiO₂ in Alcohol
Ethanolamine
Perovskite
Device Fabrication
Characterization
Conclusions
Abbreviations
Nanomaterials
- High‑Performance Na4Mn9O18/Carbon Nanotube Cathodes for Aqueous Sodium‑Ion Batteries
- From Photovoltaic Si Sludge to High‑Performance Li‑Ion Battery Anodes via Oxygen Diffusion
- Rapid, One‑Pot Synthesis of Nitrogen‑Doped Microporous Carbon Spheres for Superior Symmetric Supercapacitor Performance
- Enhanced Organic–Nanostructured Silicon Hybrid Solar Cells via Surface Modification for Superior Efficiency
- Ultra‑High Capacity Supercapacitor Electrodes from One‑Step Hydrothermal Synthesis of Chlorine‑Doped Carbonated Cobalt Hydroxide Nanowires on Nickel Foam
- High‑Performance All‑Perovskite Photodetector Delivering Ultrafast Response
- High‑Performance Flexible Photodetectors Based on Solution‑Processed CsPbI3‑xBrx Inorganic Perovskites
- In Situ-Formed, Low‑Temperature Deposited Nb‑Doped TiO₂ Compact‑Mesoporous Layer Enhances Hysteresis‑Free, High‑Efficiency Perovskite Solar Cells
- Enhancing 2D Perovskite Solar Cell Performance Through Water-Assisted Crystallization
- BD Launches CE‑IVD‑Certified, High‑Throughput Molecular Diagnostic System



