Hollow‑Structured LiNb3O8 Photocatalysts: Synthesis, Characterization, and Superior Methylene Blue Degradation
Abstract
LiNb3O8 powders with a hollow morphology were fabricated via a hydrothermal synthesis followed by a controlled sintering step. The formation of voids arises from lithium volatilization during calcination, yielding aggregates with pronounced cavities. Among the series of samples, the 700 °C‑treated material (LNO700) displayed the most remarkable photocatalytic performance, achieving complete methylene blue (MB) degradation in just 3 h under UV irradiation. The degradation obeyed pseudo‑first‑order kinetics with a rate constant of 0.97 h−1. The superior activity of LNO700 can be attributed to its enlarged specific surface area and abundance of active sites inherent to the hollow structure. Reusability tests, supported by XRD and XPS analyses before and after use, confirmed the structural stability and robust photocatalytic efficiency of the LiNb3O8 powders.
Background
Energy scarcity and environmental contamination pose intertwined challenges that impede sustainable development and public health. Photocatalysis offers a promising route to address both by enabling water splitting for hydrogen production and the mineralization of organic pollutants. Since the seminal work of Fujishima and Honda in 1972, TiO2 has dominated the field, yet the search for semiconductors with higher activity, lower cost, and better solar utilization continues.
Niobates, encompassing alkali, columbite, and rare‑earth orthoniobates, have attracted attention for their versatile applications—from optical devices to solid‑state capacitors—thanks to distinctive distorted [NbO6] octahedral frameworks that serve as active sites. Notably, LiNb3O8 has emerged as a high‑capacity lithium‑ion battery anode and as a photocatalyst capable of hydrogen evolution and degradation of toluidine blue O. Traditional solid‑state routes, however, suffer from lithium loss at elevated temperatures, often leading to phase impurities and sub‑optimal morphologies.
The hydrothermal approach, in contrast, promotes the formation of nanoscale particles with high surface areas, which is essential for photocatalytic processes. Hollow nanostructures, in particular, have been shown to enhance light harvesting via multiple scattering and to increase the density of reactive sites. To date, hollow LiNb3O8 photocatalysts have not been reported, motivating the present study.
In this work, we present a facile hydrothermal synthesis combined with sintering to produce hollow LiNb3O8 powders. We systematically investigate their crystal structures, morphologies, optical properties, and, most importantly, their photocatalytic performance against methylene blue under UV irradiation.
Methods
Photocatalyst Preparation
LiNb3O8 photocatalysts were synthesized from lithium hydroxide monohydrate (LiOH·H2O, 98 % purity) and niobium pentoxide (Nb2O5, 99.9 % purity) without further purification. A 3.5 mmol solution of Nb2O5 was dispersed in 35 mL deionized water, followed by the addition of LiOH·H2O to achieve a Li:Nb molar ratio of 8:1. After 1 h of magnetic stirring, the mixture was transferred to a 50 mL Teflon‑lined autoclave and heated at 260 °C for 24 h. The resulting white precipitate was collected by centrifugation, washed, dried, and then calcined at temperatures ranging from 600 to 1000 °C for 2 h (5 °C min−1 ramp).
Characterization
Phase identification employed X‑ray powder diffraction (XRD, Bruker D8 Discover, Cu Kα). Morphology and elemental distribution were examined by field‑emission scanning electron microscopy (SEM, JSM‑6700F) coupled with energy‑dispersive X‑ray spectroscopy (EDS). Optical absorption was measured via UV‑vis diffuse reflectance (DRS) using a Shimadzu UV‑3600, and the bandgap was derived from Kubelka–Munk plots. Photoluminescence (PL) spectra were recorded on a Jasco FP‑6500. Specific surface areas were determined by N2 adsorption–desorption (BET, Micromeritics ASAP 2460). X‑ray photoelectron spectroscopy (XPS) was performed on a Thermo‑Fisher Escalab 250Xi.
Catalytic Tests
Photocatalytic activity was evaluated by degrading 10 mg L−1 MB solutions under 500 W Hg lamp irradiation (λ = 254 nm) at natural pH. Each 50 mg catalyst was dispersed in 50 mL MB solution, stirred in the dark for 1 h to reach adsorption equilibrium, and then irradiated. MB concentrations were monitored at 665 nm every hour, and total organic carbon (TOC) was measured with an Elementar high‑TOC analyzer to confirm mineralization.
Active species were probed by adding selective scavengers: 5 mM AgNO3 (electron quencher), EDTA‑2Na (hole quencher), tert‑butyl alcohol (t‑BuOH, hydroxyl radical quencher), and benzoquinone (BQ, superoxide quencher).
Results and Discussion
XRD patterns of the powders calcined at 600–1000 °C (Fig. 1) reveal that 600 °C yields LiNbO3 and Nb2O5, whereas 700 °C predominantly forms LiNb3O8 with trace LiNbO3. Above 700 °C the phase purity improves, maintaining monoclinic LiNb3O8 (JCPDS 36‑0307) up to 1000 °C. The evolution of crystallinity and grain growth with temperature is evident.
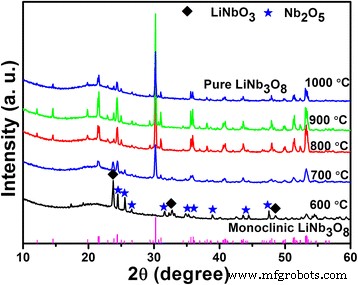
XRD patterns of the LiNb3O8 powders calcined at different temperatures for 2 h
SEM images (Fig. 2) show that at 700 and 800 °C the particles assemble into hollow spheres with well‑defined cavities—a consequence of Li volatilization. As calcination temperature rises, grain size increases from ~100 nm to 1–3 µm, the hollow morphology gradually collapses, and surface roughness diminishes. EDS confirms the presence of C, O, and Nb; lithium is undetectable, consistent with its loss during heating.
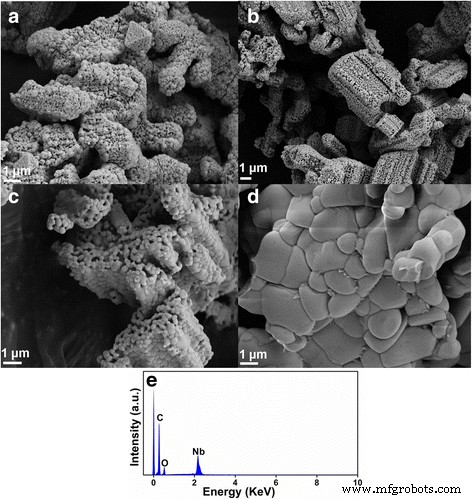
SEM images of LiNb3O8 powders calcined at a 700°, b 800°, c 900°, and d 1000 °C, respectively. e EDS spectrum of LiNb3O8 powders
Diffuse reflectance spectra (Fig. 3) yield direct bandgaps of 3.74, 3.78, 3.76, and 3.71 eV for LNO700, LNO800, LNO900, and LNO1000, respectively—values slightly lower than earlier reports, indicating efficient UV absorption.
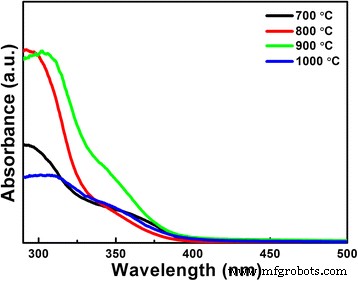
UV‑vis diffuse reflectance absorbance spectra of LiNb3O8 powders
PL measurements (Fig. 4) reveal that emission intensity decreases with higher calcination temperatures, reflecting improved electron–hole separation in better‑crystallized samples. However, the surface area diminishes sharply (BET: 10.7, 4.46, 0.36, and 0.23 m2 g−1 for LNO700–LNO1000), underscoring the trade‑off between crystallinity and surface area.
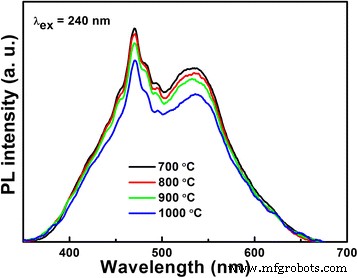
Room temperature PL spectra of the LiNb3O8 photocatalysts
Photocatalytic tests (Fig. 5) demonstrate that LNO700 achieves complete MB removal in 3 h, with a TOC reduction of 83 %—evidence of substantial mineralization. The apparent first‑order rate constants (k) follow the order LNO700 (0.97 h−1) > LNO800 (0.75 h−1) > LNO900 (0.45 h−1) > LNO1000 (0.25 h−1), correlating with the presence of hollow cavities and higher surface areas.
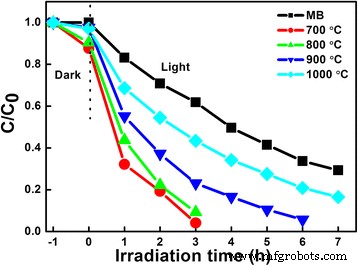
Photo‑degradation of MB with respect to the irradiation time using LiNb3O8 powders exposed to UV light. Absorption ability of LiNb3O8 powders is tested after stirring for 1 h in dark to achieve the equilibrium adsorption
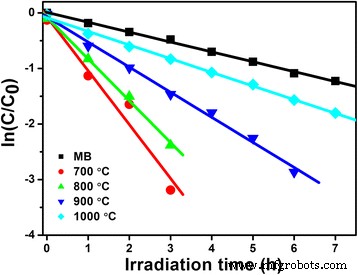
Kinetic fit for the photo‑degradation of MB in the presence of LiNb3O8 powders calcined at different temperatures
Recycling experiments (Fig. 7) confirm that LNO700 retains its photocatalytic activity and adsorption capacity over five consecutive cycles, attesting to the structural resilience of the hollow architecture.
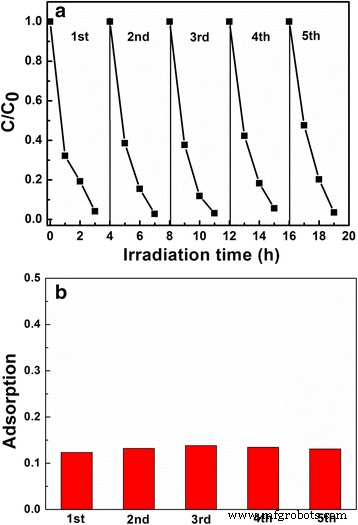
Cycling a photo‑degradation and b adsorption of MB over LNO700 powder
Active‑species trapping (Fig. 8) indicates that photogenerated electrons, superoxide anions, and hydroxyl radicals dominate the degradation pathway, whereas holes play a minor role. The proposed mechanism involves electron transfer to O2 forming O2·−, which subsequently generates ·OH, driving the oxidative breakdown of MB.
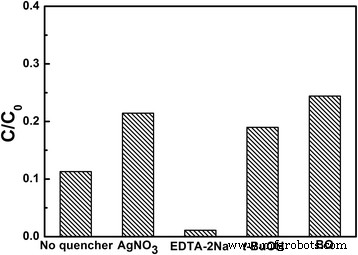
Trapping experiment of active species during the degradation of MB under UV light irradiation with the presence of LiNb3O8 catalysts
Post‑use XRD and XPS of LNO800 (Figs. 9 & 10) reveal negligible structural changes but a slight shift of Nb 3d peaks to lower binding energies, indicating surface‑restricted photoreduction of Nb5+ during photocatalysis.
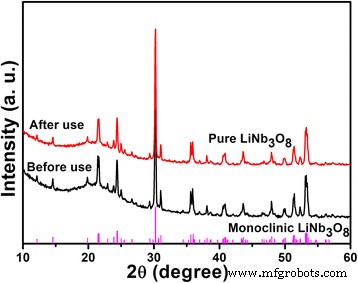
XRD patterns of LNO800 photocatalysts before and after photo‑degradation of MB under UV irradiation
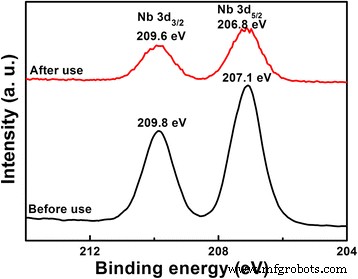
XPS spectra of Nb3d for LNO800 photocatalysts before and after photo‑degradation of MB under UV irradiation
Conclusions
The hydrothermal‑assisted sintering route successfully yields hollow LiNb3O8 powders, whose voids originate from lithium volatilization. Among the series, LNO700 exhibits the highest photocatalytic efficiency, achieving full methylene blue degradation in 3 h with a first‑order rate of 0.97 h−1. Its hollow architecture confers a high surface area and abundant active sites, while the material demonstrates excellent stability and reusability, as confirmed by XRD and XPS after repeated use.
Nanomaterials
- Hydrothermal Sintering Yields Porous, Hollow LiNb3O8 Anodes with Record Discharge Capacity
- Synergistic Graphene-Modified TiO₂ Composites Deliver Superior Photocatalytic Activity
- Impact of Li/Nb Ratio on Hydrothermal Synthesis and Photocatalytic Performance of Li‑Nb‑O Compounds
- Influence of Peptizing Acid on TiO₂ Phase Composition and Photocatalytic Efficiency: A Comparative Study of Sulfuric, Nitric, and Acetic Acids
- Electronic Structure and Transport in InSe Nanoribbons: Edge Effects and Hydrogen Passivation
- Efficient One‑Pot Synthesis of Hierarchical Silicalite‑1/Ag Nanoparticle Catalyst for Rapid 4‑Nitrophenol Reduction
- Phase‑Dependent Charge Dynamics and Photocatalytic Performance of Tin Niobate: Froodite versus Pyrochlore
- Hollow Polyaniline Microparticles Deliver 127.9 mg/g Cr(VI) Removal for Sustainable Wastewater Treatment
- Enhanced Photocatalytic Degradation of Methyl Orange Using CdTe Quantum Dot/BiOI‑Coated TiO₂ Hollow Microspheres under Simulated Sunlight
- One‑Step Hydrothermal Synthesis of Ru/SnₓTi₁₋ₓO₂ Catalysts for Diesel Oxidation: Enhanced Low‑Temperature Activity



