Hydrothermal Sintering Yields Porous, Hollow LiNb3O8 Anodes with Record Discharge Capacity
Abstract
We report the first synthesis of porous and hollow LiNb3O8 anode material using a hydrothermal‑assisted sintering route. The phase evolution and structure‑forming mechanism were elucidated. The unique porous‑hollow morphology originates from local liquid‑phase formation caused by lithium volatilization. As an anode, the material delivers an initial discharge capacity of 285.1 mAh g−1 at 0.1 C, the highest reported for LiNb3O8. After 50 cycles, a reversible capacity of 77.6 mAh g−1 is retained, roughly 2.5‑fold that of conventionally prepared samples. The superior performance is attributed to the high density of active sites and short Li+ diffusion pathways afforded by the porous, hollow architecture.
Background
Hollow and porous nanostructures have attracted intense interest across catalysis, energy storage, environmental remediation, drug delivery, and sensing [1–4]. In lithium‑ion batteries (LIBs), electrodes with open frameworks provide abundant active sites and continuous channels for rapid Li+ intercalation, leading to enhanced rate capability and cyclability [5–6]. Nevertheless, fabricating such nanostructures remains challenging.
Beyond the conventional graphite anode, transition‑metal oxides (TMOs), MoS2, and graphene hybrids have been explored for high‑capacity anodes [13–14]. Niobium‑based oxides, with a theoretical capacity of 389 mAh g−1 and dual Nb redox couples (Nb5+/Nb4+, Nb4+/Nb3+), offer both high capacity and intrinsic SEI suppression [15–19]. LiNb3O8 is a well‑known impurity phase in LiNbO3 synthesis due to Li loss, yet its potential as an anode has been largely overlooked [20]. Prior work using solid‑state synthesis reported initial capacities up to 351 mAh g−1 and limited cycling stability [18–19].
In this study, we present a hydrothermal‑assisted sintering method that yields porous, hollow LiNb3O8 with superior electrochemical performance, and we detail the phase evolution and structural origins.
Methods
Preparation of Samples
LiNb3O8 powders were synthesized via hydrothermal treatment of LiOH·H2O and Nb2O5 (Li:Nb = 8:1). 3.5 mmol Nb2O5 was dispersed in 35 mL LiOH·H2O with stirring for 1 h, then sealed in a 50 mL Teflon autoclave, heated at 260 °C for 24 h, and cooled naturally. The precipitate was washed, dried at 60 °C for 12 h, and calcined at 500–800 °C for 2 h (5 °C min−1 ramp).
Characterization
TG/DSC (Netzsch STA 409 PC/PG) measured 25–1200 °C under N2 (10 °C min−1). XRD (Bruker D8 Discover, Cu Kα) identified crystal phases. SEM (JSM‑6700F) examined morphologies. XPS (Thermo‑Fisher Escalab 250Xi) analyzed elemental states, calibrated to C 1s = 284.6 eV.
Electrochemical Measurements
Electrodes were cast from a slurry of LiNb3O8, carbon black, and PVDF (8:1:1) onto Al foil, dried at 120 °C under vacuum, and punched into 16 mm disks. Coin cells (CR2025) used Li foil, Celgard 2320 separator, and 1.0 M LiPF6 in EC/DMF (1:1). Galvanostatic cycling (Land system) ran 0–3 V (vs Li/Li+) at 0.1–1 C (1 C = 389 mAh g−1). CV (CHI604E) scanned 1–3 V at 0.1 mV s−1.
Results and Discussion
Figure 1 shows TG/DSC of the hydrothermally treated powder. Weight loss is modest (~5 %) up to 1100 °C, reflecting Li volatilization. Endothermic peaks at 330 °C (LiNbO3 formation) and 580 °C (LiNbO3 + Nb2O5 → LiNb3O8) confirm the reaction pathway, while a pronounced exotherm above 1100 °C indicates LiNb3O8 decomposition.
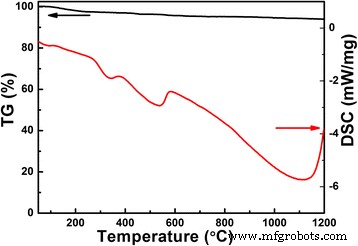
TG/DSC curves of the Li‑Nb‑O powder from room temperature to 1200 °C at 10 °C min−1 in N2
Calcined XRD patterns (Fig. 2) reveal LiNbO3 and Nb2O5 peaks at 500 °C. At 700 °C, monoclinic LiNb3O8 dominates, and a single‑phase material is obtained at 800 °C (JCPDS 36‑0307, space group P21/a). The hydrothermal route thus facilitates pure LiNb3O8 formation more readily than conventional solid‑state synthesis.
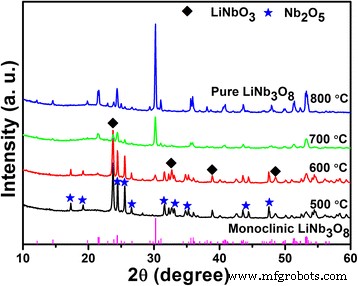
XRD patterns of the Li‑Nb‑O powder calcined at different temperatures for 2 h
SEM images (Fig. 3) show a honeycomb‑like porous and hollow framework composed of ~200 nm LiNb3O8 nanoparticles. The irregular, warped morphology contrasts sharply with the aggregated particles from solid‑state routes. The high porosity and internal voids arise from Li loss; excess Li migrates to particle surfaces, forming a transient liquid phase that nucleates new LiNb3O8 and interconnects particles into a network.

a–c SEM images of LiNb3O8 powder with different magnifications
XPS confirms the oxidation states: Nb 3d peaks at 207.1 and 209.8 eV (Nb5+), and O 1s deconvolution shows lattice (530.3 eV) and non‑lattice (532.0 eV) oxygen [22–23].
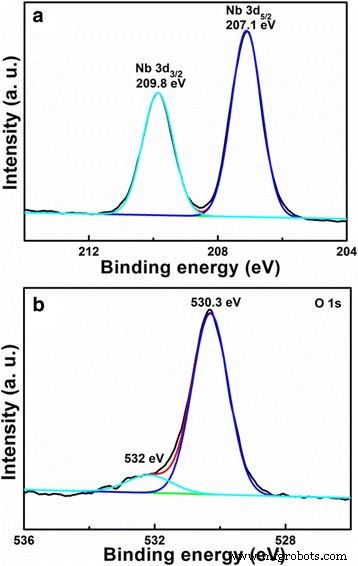
XPS spectra of (a) Nb 3d and (b) O 1s for the porous, hollow LiNb3O8
Electrochemical behavior (Fig. 5) shows two reduction peaks at 1.13 and 1.30 V during the first cycle (Nb4+→Nb3+ and Nb5+→Nb4+), which vanish in subsequent cycles, indicating irreversible phase changes. Oxidation peaks at 1.71 and 1.96 V remain stable, reflecting reversible Li extraction.
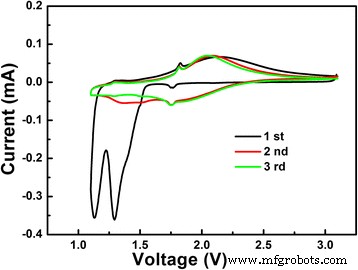
The initial three CV curves of the LiNb3O8 powder at a scan rate of 0.05 mV s−1 between 3–1 V
Galvanostatic cycling (Fig. 6) yields an initial discharge capacity of 285.1 mAh g−1 at 0.1 C. The first charge capacity (106.4 mAh g−1) indicates that ~1.6 Li⁺ are reversibly extracted, while the remaining capacity loss (~2.8 Li⁺) remains to be elucidated. After 50 cycles, the reversible capacity retains 77.6 mAh g−1, a 2.5‑fold improvement over solid‑state LiNb3O8 (≈30 mAh g−1) [18].
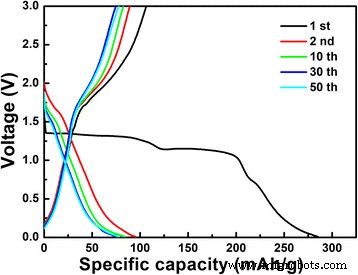
Galvanostatic charge‑discharge profiles of the LiNb3O8 powder at 0.1 C between 3 and 1 V
Rate performance (Fig. 7) shows 250 mAh g−1 at 0.5 C and 228 mAh g−1 at 1 C, with capacities after 50 cycles of 39.7 and 29.4 mAh g−1, respectively. The porous, hollow structure provides abundant active sites and short Li⁺ pathways, accounting for the high capacity and rate capability. Further surface modification could enhance long‑term stability.
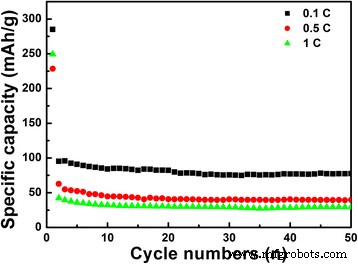
Cycling performance of the LiNb3O8 powder at different current rates of 0.1 C, 0.5 C and 1 C
Conclusions
Hydrothermal‑assisted sintering successfully produces porous, hollow LiNb3O8 anodes with record electrochemical performance. The unique structure originates from localized liquid‑phase formation during Li volatilization. The material delivers an initial discharge capacity of 285.1 mAh g−1 at 0.1 C and retains 77.6 mAh g−1 after 50 cycles, exceeding conventional solid‑state counterparts by more than two‑fold. The high capacity and rate capability are attributed to the abundant active sites and short Li⁺ diffusion channels inherent to the porous, hollow architecture.
Nanomaterials
- TOTAL partners with SKF to craft integrated maintenance & inspection strategy for Indonesian gas plants
- Silicon‑Germanium FinFETs: Pioneering Low‑Resistance Channels for 7nm and Beyond
- Nanotechnology in Battery Anodes: From Silicon to LTO and Beyond
- Hollow‑Structured LiNb3O8 Photocatalysts: Synthesis, Characterization, and Superior Methylene Blue Degradation
- Silver‑Coated Spherical Li₄Ti₅O₁₂ Anodes: A Sol‑Gel‑Assisted Hydrothermal Route for High‑Performance Lithium‑Ion Batteries
- CuGeO3 Nanowires: A High‑Capacity, Stable Anode for Advanced Sodium‑Ion Batteries
- High‑Conductivity Nano‑Silver Ink Achieves Low‑Temperature Sintering for Paper‑Based Electronics
- Penta‑Siligraphene: A Thermally Stable, High‑Capacity Anode for Li‑Ion Batteries – A First‑Principles Study
- Machining vs. Grinding: Key Differences Explained
- Efficient Vertical Bulk Material Handling with Bucket Conveyors & Elevators



