Nanotechnology in Battery Anodes: From Silicon to LTO and Beyond
Abstract
Enhancing anode performance is central to next‑generation lithium‑ion batteries. This review focuses on high‑capacity alloy anodes—particularly silicon—and the challenges of volume expansion, pulverization, and solid‑electrolyte interphase (SEI) growth. We demonstrate how reducing these materials to the nanoscale mitigates fragmentation, improves electron and ion transport, and enables novel storage mechanisms such as quasi‑capacitive behavior. Various nanostructures—including nanowires, nanotubes, nanoparticles, hollow shells, and composite coatings—are evaluated for their ability to preserve capacity, extend cycle life, and reduce stress. The discussion also covers titanium‑based anodes (LTO, TiO₂) and conversion‑type (exchange) anodes, highlighting how nanoscale engineering enhances performance and safety across all classes.
Introduction
Graphite, with a layered structure and a 35.3 Å interlayer spacing, serves as the traditional intercalation anode. During lithiation, lithium ions intercalate between the layers, expanding the spacing to 3.5 Å, while delithiation restores the original structure. Each graphite unit can accommodate one lithium ion per six carbon atoms, giving a theoretical capacity of ≈ 350 mAh g⁻¹. Although graphite lacks dendrite formation, its low capacity necessitates alternative high‑energy anodes. Alloy anodes—formed by lithium alloying with metals such as silicon, antimony, or tin—offer much higher capacities (silicon ≈ 4000 mAh g⁻¹, tin ≈ 900 mAh g⁻¹) but suffer from drastic volume changes (up to 400 %) that lead to pulverization and loss of electronic contact. Table 1 lists the theoretical capacities and key attributes of representative anode materials.
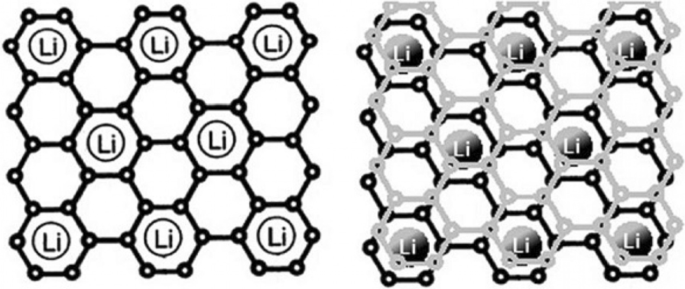
Illustration of lithium storage in graphite: one Li⁺ per six C atoms.
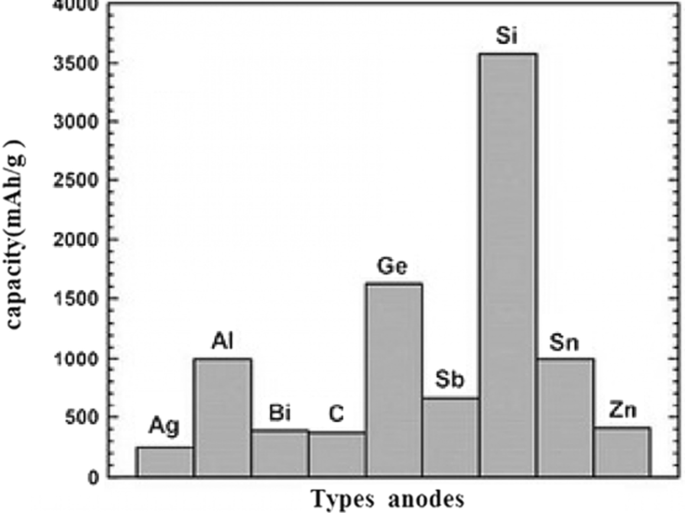
Comparative capacities of graphite, silicon, tin, and antimony anodes.
Problems of Alloy Anodes
Alloy anodes undergo large volumetric expansions during lithiation—up to 400 % for silicon—producing severe mechanical stresses that fracture the active material. Fragmentation severs electrical pathways between the active phase, conductive additives, and current collectors, leading to capacity fade and reduced coulombic efficiency. Figure 3 depicts the pulverization phenomenon and its impact on electrochemical performance. Conventional electrodes, composed of micron‑sized particles, binders, and carbon, cannot accommodate these stresses, resulting in rapid degradation. In contrast, graphite experiences only a 3 % volume change, allowing it to retain capacity over thousands of cycles.
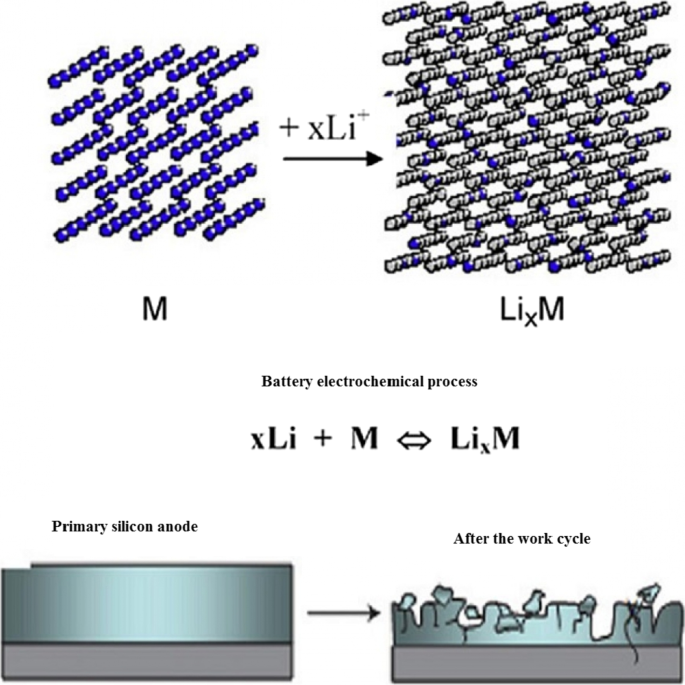
Pulverization and loss of electrical contact in silicon anodes.

Charge–discharge curves of 10‑µm silicon particles versus graphite.
Nanotechnology Solution
Reducing silicon dimensions below 150 nm eliminates pulverization. TEM images (Figure 5) show silicon nanoparticles expanding during lithiation without fracturing. However, isolated nanoparticles still suffer from poor electronic connectivity. Vertically aligned silicon nanowires grown directly on current collectors (Figure 6) provide continuous pathways for electrons and ample void space for volume expansion, preserving capacity and preventing stress‑induced cracking. The high surface area of nanostructures also shortens lithium‑ion diffusion paths, reducing concentration polarization and enhancing power density.
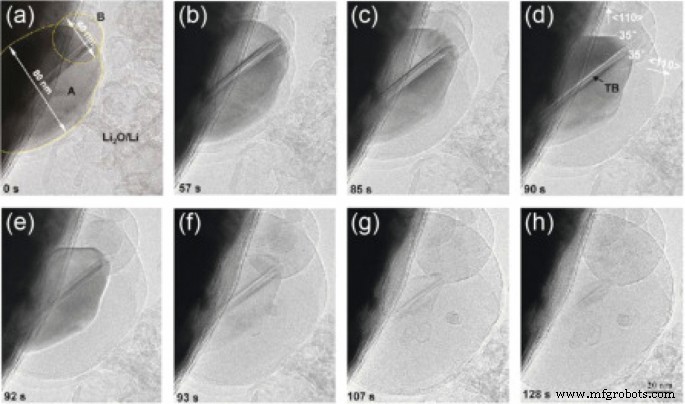
TEM images of silicon nanoparticles at different lithiation stages.

SEM of silicon nanowires grown on a current collector.
Different Nano Morphologies
Silicon nanotubes offer additional internal voids that accommodate expansion from both inner and outer walls. However, their thin walls can become amorphous during cycling, limiting electronic conductivity. Hybrid structures—e.g., silicon cores wrapped in conductive carbon shells—restore electrical pathways while preserving volume buffering capacity. Carbon nanocomposites with tin (Figure 7) exemplify how a carbon matrix can provide mechanical support and electronic conduction, extending cycle life to ~ 1000 cycles despite a slightly reduced theoretical capacity. Nanoparticle synthesis routes vary from sol–gel to electrospinning and CVD, each influencing particle size, crystallinity, and scalability.
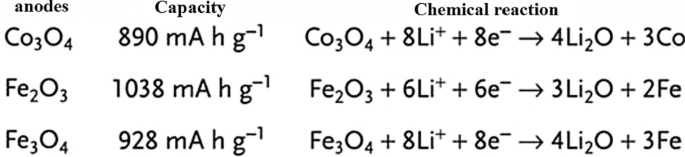
TEM of tin–carbon nanocomposite: tin particles encapsulated in carbon.
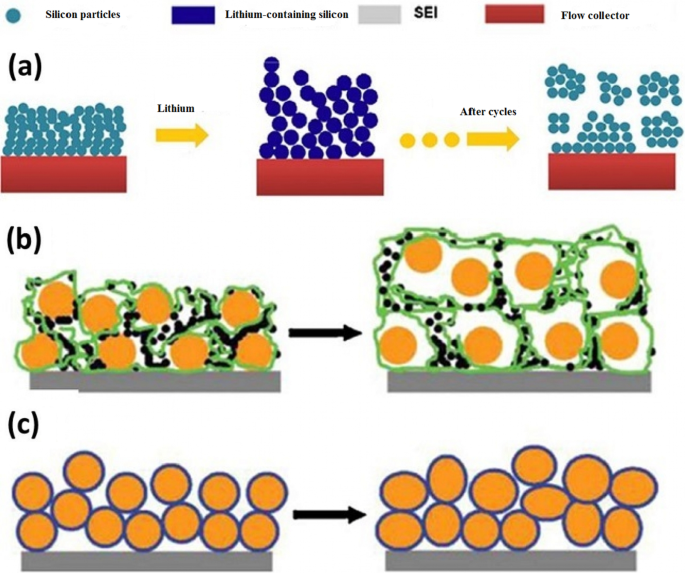
(a) Electrical contact loss in nanoparticles; (b) conventional slurry; (c) amorphous silicon binder restores connectivity.

Hollow nanoparticles mitigate volume‑induced stress.
Electrolyte Decomposition in the Anode
Electrolyte reduction occurs when the electrode potential drops below the electrolyte’s stability window. In graphite anodes, a thin, lithium‑ion‑permeable solid electrolyte interphase (SEI) forms, consuming lithium and increasing internal resistance. The SEI composition is complex but typically includes Li⁺, C, and organic fragments. While the SEI protects the electrode from further reduction, it also impedes electron flow and increases diffusion distances. Figure 10 shows the SEI structure, while Figure 11 compares the stability windows of common anodes and cathodes, highlighting why high‑voltage cathodes rarely form SEI while graphite and silicon do.
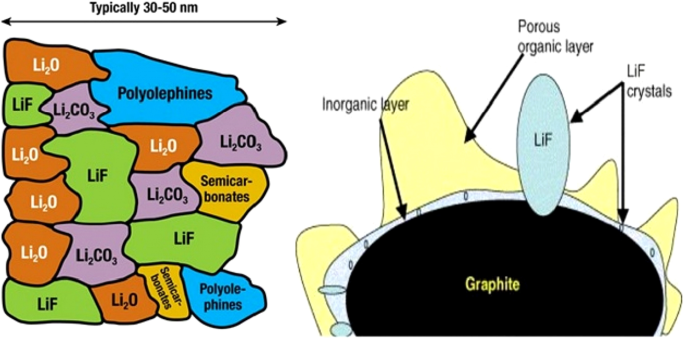
Schematic of SEI formation and its layered composition.
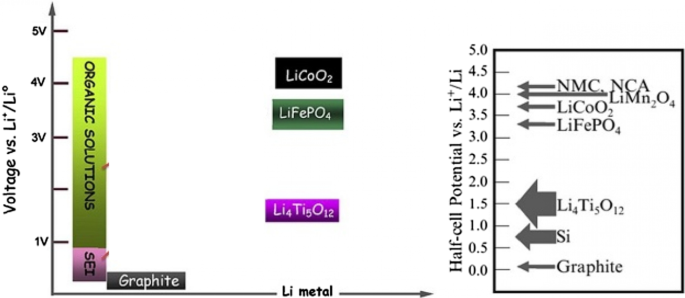
Potential windows of anodes and cathodes relative to SEI formation.
SEI Problem in Silicon
Silicon’s anode potential (0.3–0.4 V vs Li⁺/Li) lies below the electrolyte stability range, triggering continuous SEI growth during cycling. As silicon expands and contracts, fresh surfaces are exposed to the electrolyte, forming new SEI layers that consume additional lithium and degrade capacity. Figure 12 illustrates how SEI layers grow on nanowires and nanoparticles, while Figure 13 shows strategies—such as silicon oxide coatings and core–shell architectures—to suppress SEI evolution by preventing direct electrolyte contact with the active silicon. These designs enable high cycle life (> 900 cycles) even under deep‑discharge conditions.
Progressive SEI growth on silicon nanostructures.
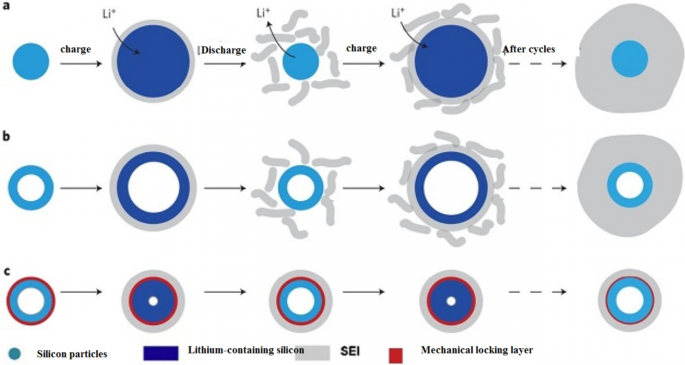
Mechanical locking layers and core–shell designs that suppress SEI formation.
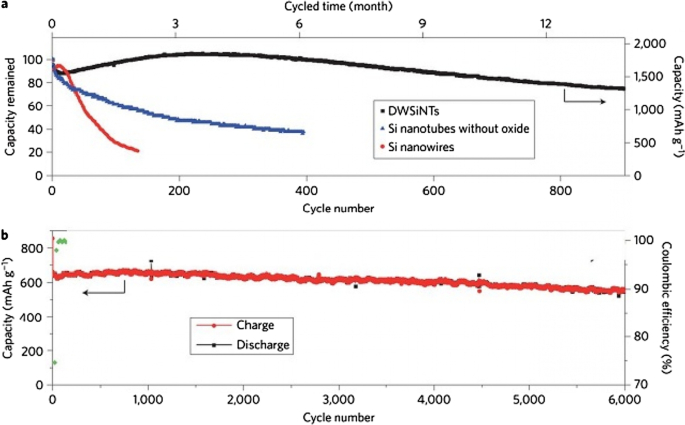
Comparative cycle life of oxide‑coated versus uncoated silicon nanotubes.
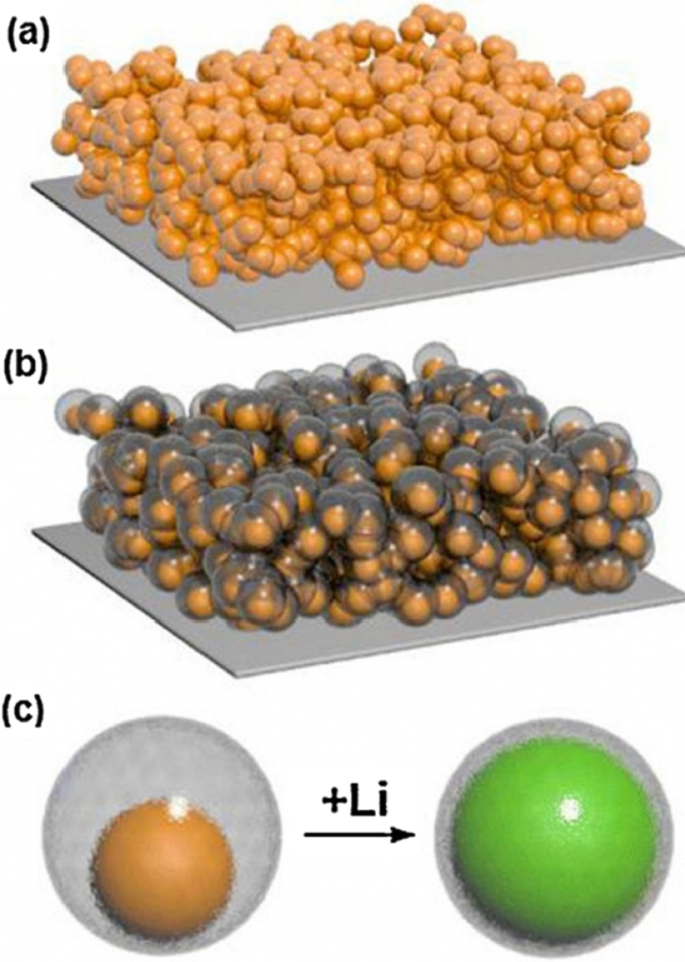
Core–shell silicon nanoparticles: (a) bare, (b) carbon‑coated, (c) hollow‑core design.
Introducing LTO Anode
Li₄Ti₅O₁₂ (LTO) is an intercalation anode that operates at ~1.5 V vs Li⁺/Li, well above the electrolyte stability window, thus eliminating SEI formation. Its reversible capacity (~175 mAh g⁻¹) is modest, but LTO offers exceptional safety, thermal stability, and a projected cycle life of > 20,000 cycles. The Ti⁴⁺/Ti³⁺ redox reaction occurs without significant volume change, preserving electrode integrity. Nanostructuring LTO (10‑nm primary particles aggregated into micron‑sized secondary spheres) dramatically shortens lithium‑ion diffusion paths, enabling rapid charge–discharge rates (up to 12 C) while maintaining capacity retention.
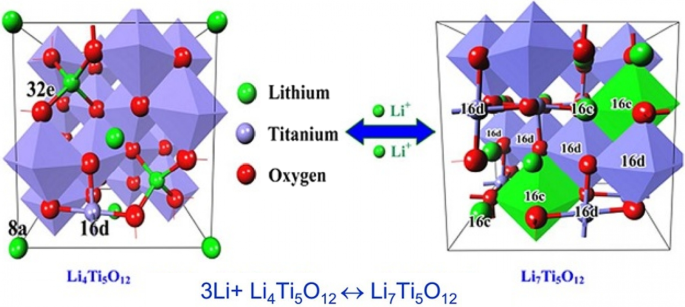
Structural evolution and Li⁺ insertion mechanism in LTO.
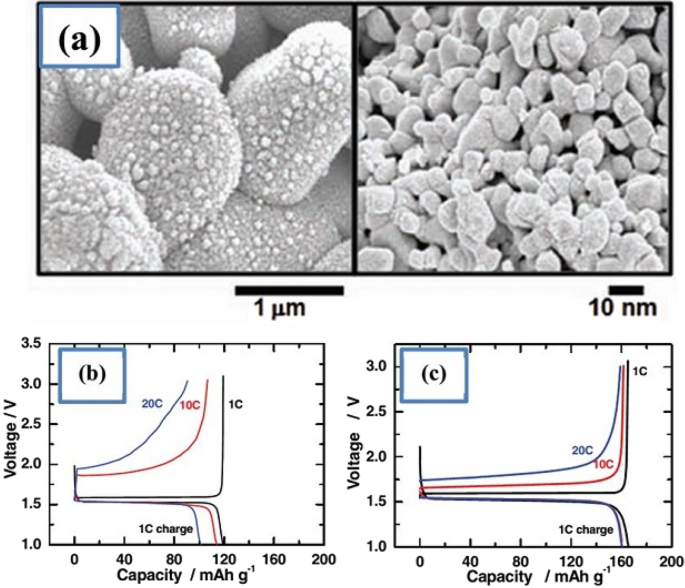
(a) Nanostructured LTO morphology; (b) charge–discharge curves for micro‑ vs nano‑particles; (c) effect of particle size on capacity.
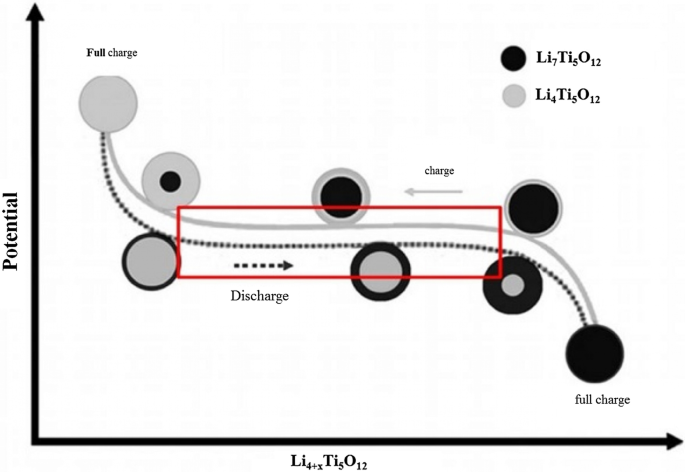
Linear voltage plateau of LTO and its reduction with decreasing particle size.
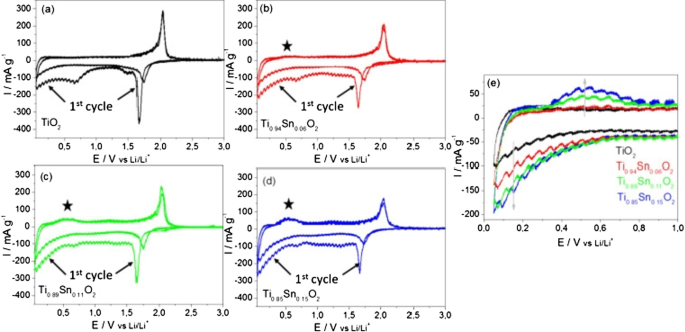
TiO₂ Anode
TiO₂ anodes (LTO family) offer high safety and no volume change. Four polymorphs exist—brockite, anatase, rutile, and TiO₂(B)—each with distinct lithium‑ion transport channels. The TiO₂(B) phase, in particular, provides open channels that facilitate rapid Li⁺ diffusion, yielding high specific capacity (~335 mAh g⁻¹) and superior power density. Nanoscale TiO₂, especially in nanowire or hollow‑tube form, reduces diffusion distances and mitigates the c‑axis bottleneck, enabling reversible capacities up to 0.8 mol Li⁺ mol⁻¹ and cycle life exceeding 1000 cycles.
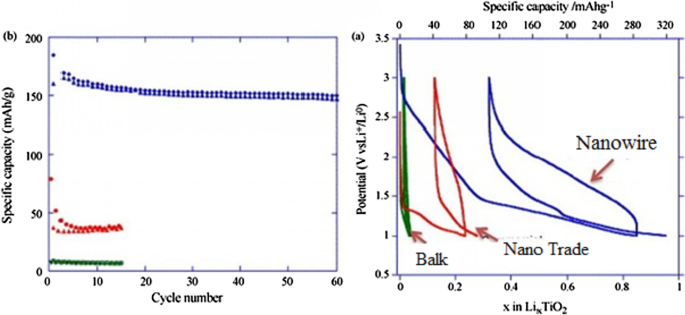
(a) Charge–discharge of rutile TiO₂; (b) cycle life comparison.
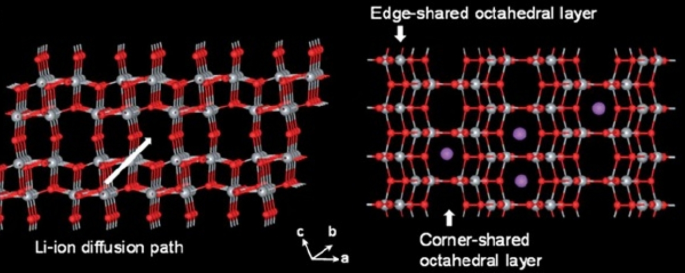
Atomic structure of TiO₂(B) phase and its lithium‑ion pathways.
Quasi‑capacitive Capacity
At the nanoscale, a significant fraction of lithium storage occurs at the electrode surface due to the high surface‑to‑volume ratio, yielding a fast, capacitive‑like charge transfer that is independent of bulk diffusion. This quasi‑capacitive behavior provides high power density and excellent cycle life, though the energy density is lower than bulk intercalation. Figure 21 compares the surface‑controlled capacity of LTO nanoparticles of varying sizes, showing a pronounced decline as particle size increases beyond the few‑nanometre regime.
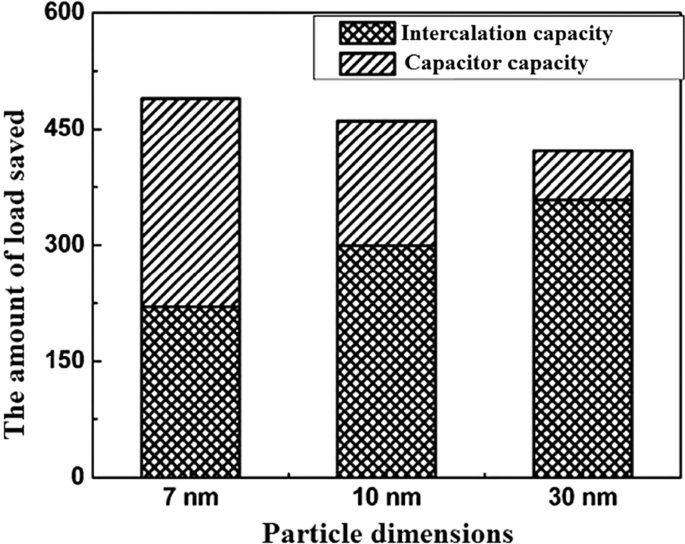
Surface‑controlled capacity of LTO nanoparticles vs particle size.
Introduction of Exchange Anodes
Conversion or exchange anodes (e.g., CoO, FeO, Co₃O₄) operate via a redox reaction that liberates multiple Li⁺ per formula unit (n > 1), offering theoretical capacities far beyond intercalation materials. However, these reactions involve significant volume changes and sluggish electron/ion transport, leading to high voltage hysteresis and rapid capacity fade. Figure 22 presents the general mechanism and key reactions for representative exchange anodes.
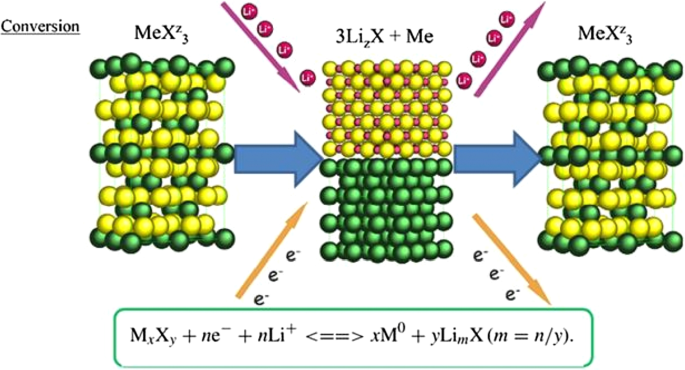
Mechanism of lithium insertion/extraction in conversion anodes.
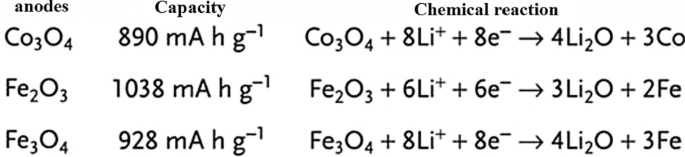
Capacities and reaction pathways of various conversion oxides.
Exchange Anode Problems
Exchange anodes face challenges analogous to alloy anodes: large volume changes, mechanical pulverization, and SEI growth. Their intrinsically low electronic and ionic conductivity further slows charge/discharge, producing a pronounced voltage hysteresis (up to 1 V) as shown in Figure 24. Hysteresis reflects activation polarization and limits practical energy density. Strategies such as nanosizing, conductive coatings, and composite architectures are essential to reduce these effects.
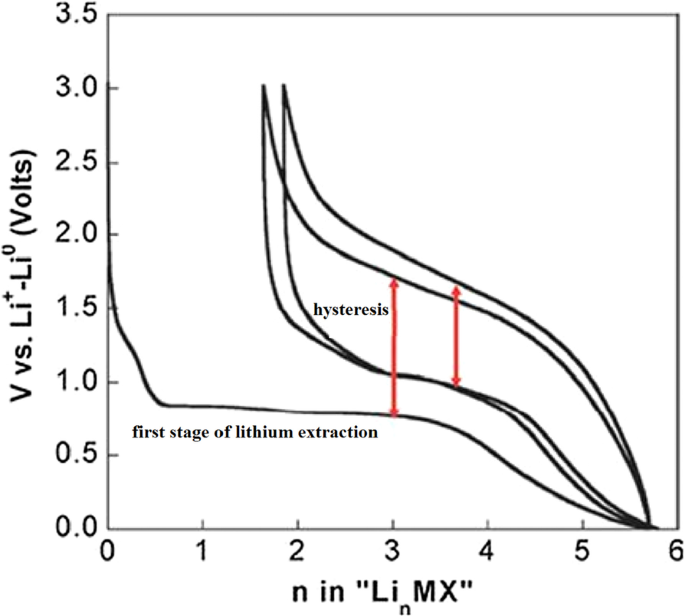
Charge–discharge curves of exchange anodes illustrating voltage hysteresis.
Nano Sizing Effects
Figure 25 compares the lithium‑ionization behavior of 20‑nm iron oxide nanoparticles with 500‑nm micron particles. The nanoscale sample shows a higher reversible lithium uptake and a steeper charge–discharge profile, attributed to the enlarged surface area (~ 60 m² g⁻¹ vs 2 m² g⁻¹) and reduced diffusion path. Consequently, the nanoscale material retains more capacity over successive cycles and exhibits lower volume expansion (< 1 % vs > 10 % for bulk).
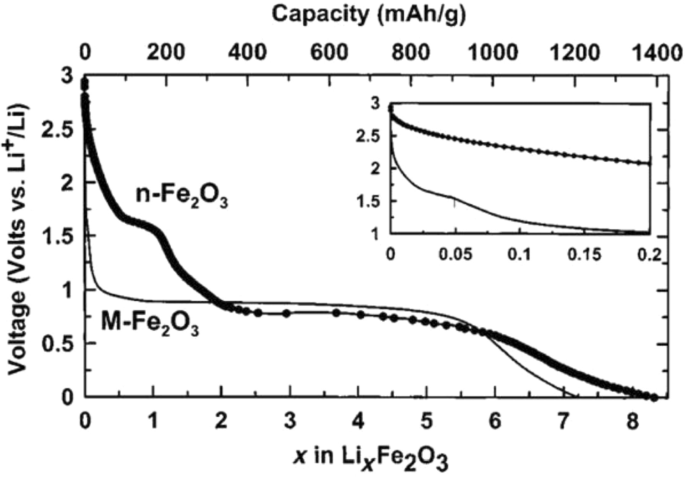
Li⁺ ionization curves for nano‑ and micro‑Fe₂O₃ particles.
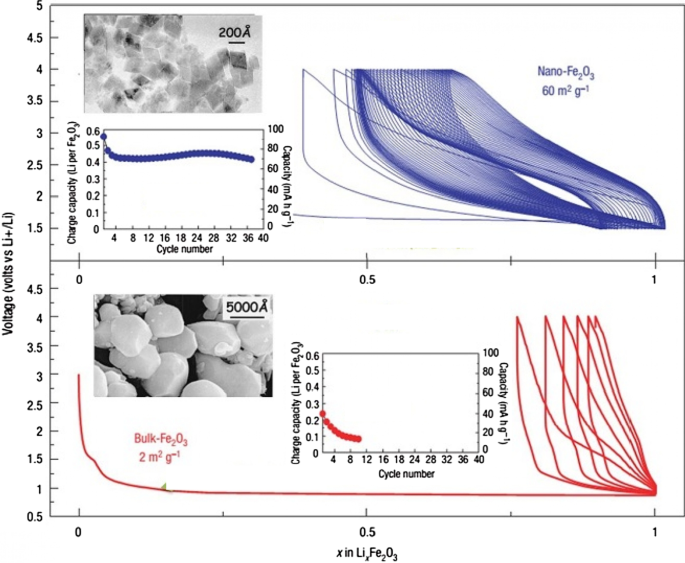
SEM, charge–discharge curves, and cycle life of nano‑ vs bulk Fe₂O₃.
Nanomaterials in Batteries
Nanostructuring active electrode materials offers three main advantages: (1) new reactions inaccessible to bulk forms, (2) increased electrode–electrolyte interface area, and (3) shortened electron and Li⁺ transport paths. These benefits translate into higher charge/discharge rates and better cycle life, crucial for electric vehicles and grid storage. Table 2 summarizes common synthesis routes—ball milling, CVD, template methods, hydrothermal, sol–gel, etc.—and their impact on particle size, crystallinity, and scalability.

Core–shell and core–wall architectures for LiFePO₄ using carbon nanotubes.
Silicon continues to be a focus of nanomaterial research due to its high theoretical capacity (≈ 4000 mAh g⁻¹) and abundance. Nonetheless, its large (~ 300 %) lithiation expansion demands robust mechanical buffering and high electronic conductivity. Composite designs such as Si@C, Si–metal alloys, and porous silicon structures combine silicon’s capacity with conductive matrices that absorb strain and provide pathways for charge transfer. Continuous hydrothermal flow synthesis (CHFS) is emerging as a scalable method to produce high‑quality nanocrystals with narrow size distributions (< 10 nm), essential for reproducible electrode performance.

Cyclic voltammograms of anatase TiO₂ and Sn‑doped variants.
Conclusion
- Silicon alloy anodes exhibit unparalleled capacity, but pulverization limits cycle life. Nanostructuring—especially nanowires, nanotubes, and core–shell composites—provides mechanical resilience and efficient charge transport, enabling near‑theoretical performance.
- SEI growth on alloy anodes is a primary degradation driver. Nanoscale designs that isolate active silicon from direct electrolyte contact, such as silicon oxide shells or hollow structures, suppress continuous SEI formation and preserve capacity.
- Carbon coatings and dopants elevate electronic conductivity in both silicon and titanium‑oxide anodes. For LTO and TiO₂, nanosizing reduces Li⁺ diffusion distances and eliminates the need for high‑temperature stability, improving power and energy density.
- Exchange (conversion) anodes offer high theoretical capacities but suffer from volume change and hysteresis. Nanomaterials mitigate these issues by enhancing conductivity and buffering strain, shifting the dominant storage mechanism toward faster, surface‑controlled processes.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article.
Nanomaterials
- Understanding Resistance, Reactance, and Impedance in AC Circuits
- Tin Nanocrystals: The Next Frontier for High‑Capacity Lithium‑Ion Batteries
- Biosensors and Nanosensors for Monitoring Agroecosystems: A Comprehensive Review
- Graphene‑Polymer Composites for High‑Performance Supercapacitors: A Comprehensive Review
- From Photovoltaic Si Sludge to High‑Performance Li‑Ion Battery Anodes via Oxygen Diffusion
- Efficient Solution-Scale Synthesis of Red Phosphorus Nanoparticles for High‑Performance Lithium‑Ion Battery Anodes
- Comprehensive Review of Luminescent Silica Nanoparticles: Classification, Synthesis, and Practical Applications
- Enhancing Lithium Metal Anode Cycling with Atomic-Scale Interlamellar Ion Channels for Li‑S Batteries
- Safe Charging & Storage of Drone Lithium Batteries: A Comprehensive Guide
- Breaking Down Lithium Battery Costs: Why They’re More Expensive Than You Think



