TiO₂/Porous Carbon Coated Celgard 2400 Separators Enhance Lithium/Sulfur Battery Performance
Abstract
The practical use of lithium/sulfur batteries is limited by the migration of soluble polysulfides (Li2Sn, 4 ≤ n ≤ 8) from cathode to anode, which undermines cell stability. In this study, we fabricated a TiO2/porous carbon (TiO2/PC) composite coating on a Celgard 2400 separator, acting as a robust polysulfide barrier. The highly conductive PC, with a three‑dimensional ordered porous framework, physically confines polysulfides while also serving as an auxiliary current collector. TiO2 nanoparticles anchored on the PC surface chemically adsorb polysulfides during charge–discharge cycles. This dual physical‑chemical strategy delivers an initial discharge capacity of 926 mAh g−1 at 0.1 C and retains 75 % of the capacity after 150 cycles. Rate‑capability tests show the modified cell recovers 728 mAh g−1 at 0.1 C after high‑rate cycling, maintaining ~88 % of the initial reversible capacity.
Background
Lithium/sulfur (Li/S) batteries have attracted attention as next‑generation energy storage devices due to their exceptionally high theoretical energy density (2,600 Wh kg−1) and specific capacity (1,675 mAh g−1)1. Their additional advantages include low toxicity, inexpensive raw materials, and abundant natural sulfur2. Nonetheless, several intrinsic challenges hinder commercial deployment: (i) the insulating nature of elemental sulfur (σ298 = 5 × 10−30 S cm−1) leads to poor active‑material utilization; (ii) the volumetric expansion when Li2S forms from S8 causes capacity fade; and (iii) the dissolution and migration of polysulfides in the electrolyte reduce Coulombic efficiency and accelerate capacity decay3,4. Extensive research has focused on confining sulfur within cathode hosts such as porous carbon, inorganic oxides, and polymers5–13, yet high loading of trapping materials inevitably diminishes overall energy density. Consequently, strategies that modify the internal architecture of the cell, particularly the separator, have gained traction. Coating interlayers on separators have been shown to impede polysulfide shuttling through physical absorption14,15 and chemical interactions with polar metal oxides like CeO2 or layered double hydroxides18–22. The TiO2 surface, in particular, has demonstrated strong polar–polysulfide interactions both experimentally and theoretically23,24. Here, we present a TiO2/PC composite coating on Celgard 2400 that synergistically combines physical confinement and chemical binding to suppress the shuttle effect.
Methods
Preparation of Li/S Battery with TiO2/PC-Modified Separator
Preparation of Porous Carbon
Figure 1 illustrates the fabrication route of the TiO2/PC‑modified Celgard 2400 separator. Monodisperse silica microspheres were synthesized via TEOS hydrolysis in ammonia, followed by ethanol centrifugation. After natural drying, the silica opal was impregnated with a resol solution and pyrolyzed at 600 °C for 2 h under Ar (ramp 2 °C min−1). The process yielded a carbonized template with ~11 % weight loss. Subsequent HF etching removed the silica framework, producing a three‑dimensional ordered porous carbon (PC) scaffold.
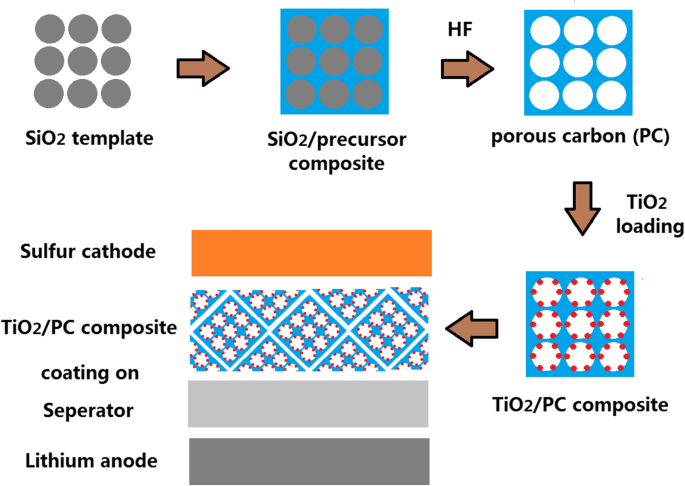
Synthesis of the TiO2/PC-modified Celgard 2400 separator for the Li/S battery
Deposition of TiO2 on PC
The TiO2 precursor was prepared via a sol–gel route. TTIP (2.84 g, 0.1 mol), HCl (2.4 g), and ethyl alcohol (4.0 g) were mixed and stirred for 1.5 h, forming a transparent gel. The PC template was soaked in this solution for 24 h, then dried for 3 days and calcined at 450 °C for 1 h under N2 to anchor TiO2 nanoparticles onto the carbon matrix.
Preparation of the TiO2/PC-Modified Separator
A slurry of 0.7 g TiO2/PC, 0.2 g carbon black, and 0.1 g PVDF in NMP was coated onto commercial Celgard 2400 and dried at 50 °C overnight in vacuum. The resulting TiO2/PC layer was 37 µm thick, with an areal loading of ~0.5 mg cm−2. Disks of 1 cm diameter were punched for cell assembly.
Material Characterizations
Crystalline phases were identified by powder XRD (Cu–Kα, λ = 1.5406 Å, 2θ = 10–90°). Morphology was examined via SEM (JSM‑7100F) and TEM (JEM‑2100F, 200 kV). Contact angles were measured with a JGW‑360Y meter. XPS (Kratos AXIS Ultra DLD, Al–Kα) assessed surface chemistry before and after cycling.
Electrochemical Measurements
Sulfur cathodes were prepared by mixing 0.8 g S, 0.1 g carbon black, and 0.1 g PVDF in NMP, coated onto Al foil, and dried at 60 °C under vacuum. Sulfur loading was ~2.0 mg cm−2. Coin cells (CR2025) were assembled in an Ar glove box (≥ 99.9995 %) with 1 M LiTFSI in a 1:1 (v/v) DOL/DME electrolyte. Charge–discharge cycling was conducted between 1.5 and 3 V using a Neware BTS‑5V5mA tester at room temperature.
Results and Discussion
Figure 2 displays the XRD pattern of the TiO2/PC-modified separator, confirming anatase TiO2 (JCPDS No.21‑1272) and characteristic carbon peaks at ~23° (002) and ~44° (100).
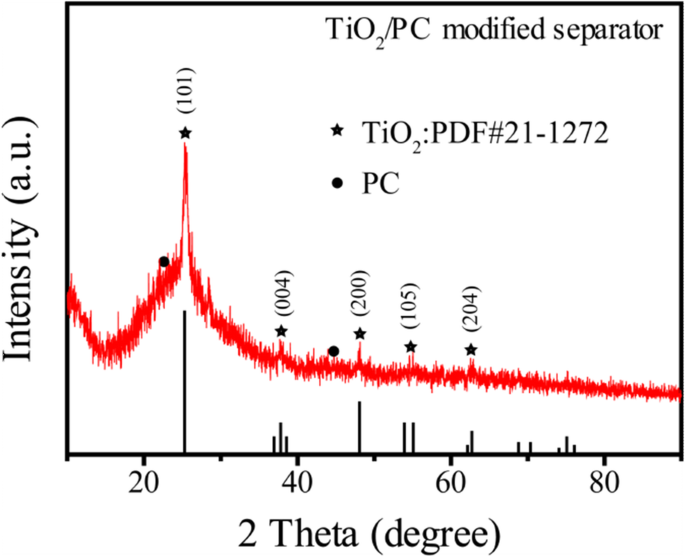
XRD pattern of the TiO2/PC-modified separator
SEM and TEM images (Figure 3) reveal a uniform, ordered porous network (~110 nm pores) with TiO2 nanoparticles evenly dispersed on the PC matrix. A lattice spacing of 0.35 nm, matching the (101) facet of anatase TiO2, confirms the crystalline nature of the embedded nanoparticles.

SEM (a,b) and TEM (c,d) images of the TiO2/PC interlayer
Figure 4a shows the nitrogen adsorption–desorption isotherm of TiO2/PC, yielding a BET surface area of 263 m2 g−1. The pore size distribution (Figure 4b) highlights micropores around 1 nm and a broad mesoporous range, reinforcing the physical confinement capability.
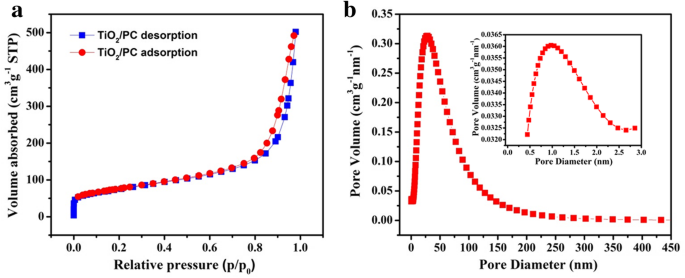
a N2 adsorption–desorption isotherms. b Pore diameter distribution of TiO2/PC. Inset: magnification of pore diameter distribution between 0 and 3 nm
Figure 5a presents the XPS survey spectrum after cycling, confirming the presence of O, Ti, C, and S. High‑resolution spectra (Figure 5b–d) identify C–C/C=C (284.6 eV), O–C=O (290.4 eV), S–Ti (162.9 eV), S 2p2/3 (163.9 eV), S 2p1/2 (165.0 eV), sulfate (170.4 eV), –SO3 (167.0 eV), C–S (169.0 eV), Ti–S (458.25 eV), Ti 2p2/3 (459 eV), and Ti 2p1/2 (464.7 eV). The Ti–S bond indicates a strong chemical interaction between TiO2 and polysulfides.
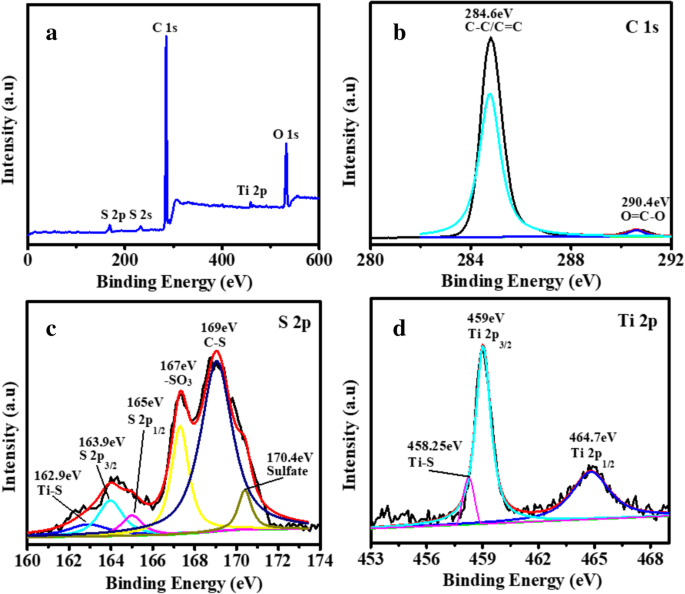
Wide spectrum (a) and high-resolution XPS spectra of the TiO2/PC-modified separator after charge/discharge: C 1s (b), S 2p (c), Ti 2p (d)
Figure 6a demonstrates the mechanical flexibility of the TiO2/PC-coated separator. Contact‑angle measurements (Figure 6b) show a complete wetting (0°) on the modified surface versus 38° on the pristine separator, reflecting the enhanced electrolyte infiltration afforded by the polar TiO2/PC composite.

Digital images of the TiO2/PC-modified separator with excellent flexibility. (a) The contact angle of the electrolyte on the TiO2/PC-modified separator and the unmodified separator (b)
CV curves (Figure 7) for cells with and without the TiO2/PC separator were recorded at 0.1 mV s−1. The modified separator shifts the cathodic peaks to higher potentials (2.27 V and 1.97 V) and reduces the anodic peak (2.44 V), indicating suppressed polarization and enhanced reaction kinetics.
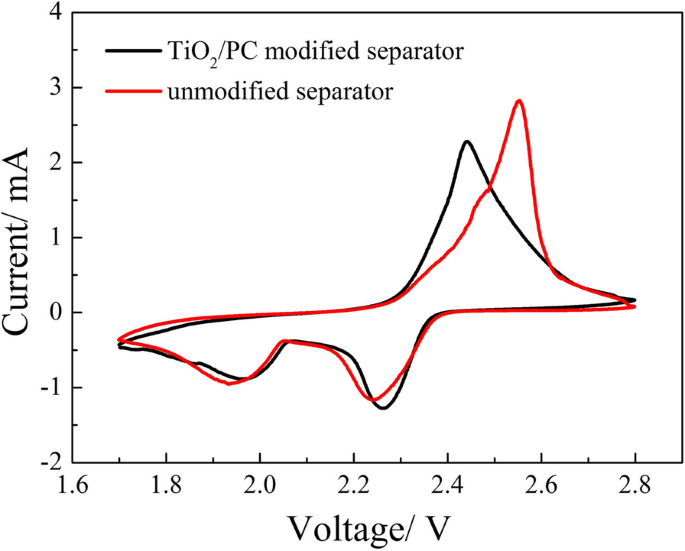
CV curves of the cells with and without TiO2/PC-modified separator
Galvanostatic charge/discharge curves at 0.1 C (Figure 8) display two plateaus at 2.27 V and 1.97 V, corresponding to the two‑step reduction of S8 and subsequent conversion to Li2S2 / Li2S. The first cycle delivers 1,060 mAh g−1; subsequent cycles maintain 926 mAh g−1 (second cycle) and 853 mAh g−1 (third cycle), evidencing robust cycling stability.
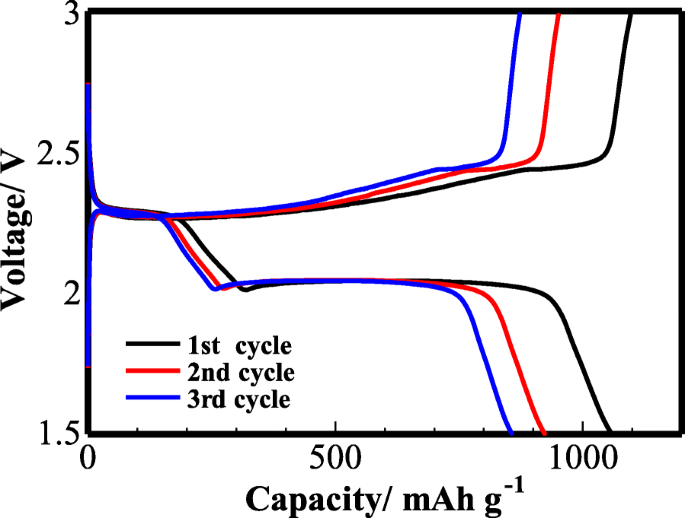
The charge/discharge curves of the cell with TiO2/PC-modified Celgard 2400 separator at 0.1 C
Cycling performance (Figure 9) shows the modified cell retains ~75 % (708 mAh g−1) of its initial reversible capacity after 150 cycles at 0.1 C, whereas the pristine separator degrades rapidly. At 1 C (Figure 10), the modified separator delivers 788 mAh g−1 initially and 564 mAh g−1 after 300 cycles, underscoring its superior long‑term stability.
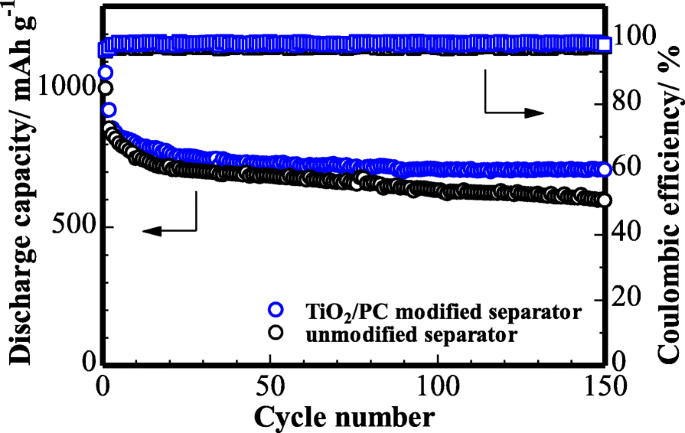
Cycling stability of the cell (with TiO2/PC-modified separator and unmodified) at 0.1 C
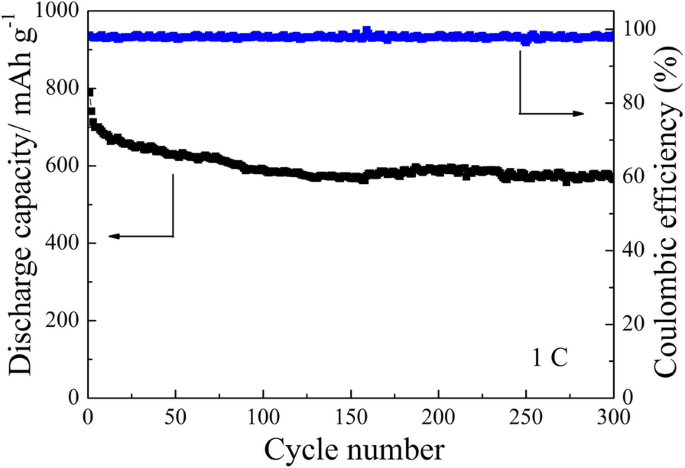
Long‑term cycling stability of the cell with TiO2/PC-modified separator at 1 C
Rate capability (Figure 11) demonstrates reversible capacities of 823, 672, 578, and 455 mAh g−1 at 0.1, 0.5, 1, and 2 C, respectively. After high‑rate cycling, the capacity recovers to 728 mAh g−1 at 0.1 C, preserving ~88 % of the initial capacity. The pristine separator exhibits markedly lower capacities, confirming the effectiveness of the TiO2/PC layer in enhancing sulfur utilization and curbing polysulfide migration.
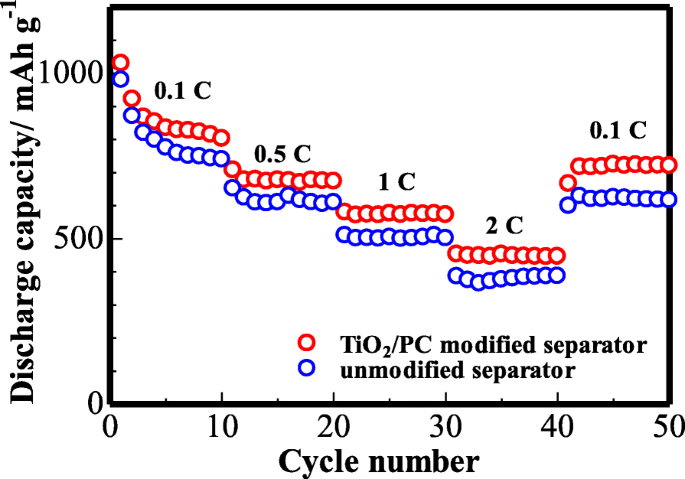
The rate performance of the cell (with unmodified and TiO2/PC-modified Celgard 2400 separator) at various current densities
Self‑discharge behavior was examined after 72 h rest following the initial three cycles at 0.1 C. The unmodified separator shows a 0.21 V drop (2.28→2.07 V), indicating significant polysulfide migration. In contrast, the TiO2/PC-modified separator experiences only a 2.6 % voltage decline (2.30→2.24 V), reflecting its superior ability to trap polysulfides and mitigate self‑discharge.
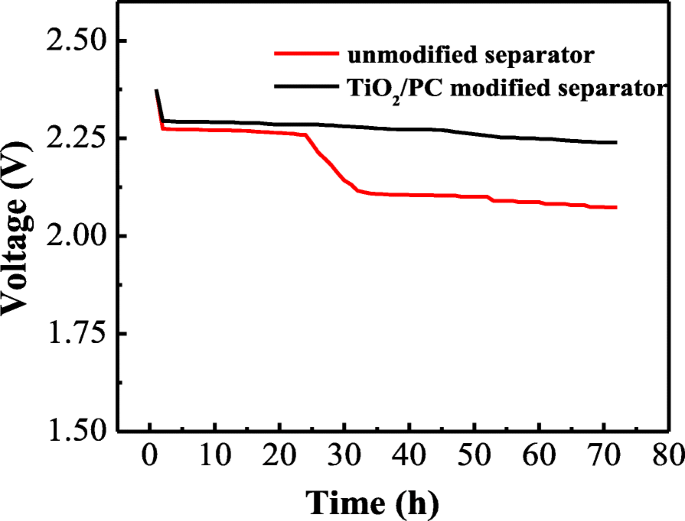
Open‑circuit voltage profiles of the cells with unmodified and TiO2/PC-modified separator during 72 h rest time
Conclusions
We have demonstrated that a TiO2/PC composite coating on Celgard 2400 effectively suppresses the polysulfide shuttle in Li/S batteries. The TiO2 nanoparticles provide strong electrostatic attraction (S–Ti–O), while the ordered porous carbon matrix offers high electrical conductivity and physical confinement. The resulting cell delivers an initial specific capacity of 926 mAh g−1 and retains 75 % after 150 cycles, achieving superior cycling stability and rate performance. This separator‑modification strategy presents a practical route toward high‑performance Li/S energy storage devices.
Abbreviations
- DME:
1,2-Dimethoxyethane
- DOL:
1,3-Dioxolane
- Li/S:
Lithium/sulfur
- LiTFSI:
Lithium bis (trifluoromethanesulfonyl)imide
- NMP:
N-methyl pyrrolidone
- PC:
Porous carbon
- PVDF:
Polyvinylidene fluoride
- SEM:
Scanning electron microscope
- TEM:
Transmission electron microscope
- TEOS:
Hydrolyzing tetraethyl orthosilicate
- TiO2:
Titanium dioxide
- TTIP:
Tetraisopropyl titanate
- XPS:
X-ray photoelectron spectroscopy
- XRD:
X-ray diffraction
Nanomaterials
- Tin Nanocrystals: The Next Frontier for High‑Capacity Lithium‑Ion Batteries
- Monodisperse Carbon Nanospheres with Hierarchical Porosity Deliver Superior Supercapacitor Performance
- Polyaniline‑Coated Activated Carbon Aerogel/Sulfur Composite Yields Superior Lithium‑Sulfur Battery Performance
- Facile Thermal Decomposition Loading of CeO₂ Nanoparticles onto Anodic TiO₂ Nanotube Arrays
- Efficient Solution-Scale Synthesis of Red Phosphorus Nanoparticles for High‑Performance Lithium‑Ion Battery Anodes
- High‑Performance Li/S Batteries Enabled by CeO₂/RGO Composite‑Coated Separators
- Nitrogen‑Doped Porous Carbon Networks Derived from Graphitic Carbon Nitride for High‑Performance Oxygen Reduction
- Self‑assembled SnIn4S8/TiO₂ Nanotube Photoanode for Enhanced Visible‑Light Photocathodic Protection of Q235 Steel
- Enhancing Lithium Metal Anode Cycling with Atomic-Scale Interlamellar Ion Channels for Li‑S Batteries
- Enhanced TiO₂ Catalysis and Antimicrobial Efficacy via Nitrogen and Carbon Nitride Co‑Doping



