Monodisperse Carbon Nanospheres with Hierarchical Porosity Deliver Superior Supercapacitor Performance
Carbon nanospheres with distinct microstructures were synthesized by carbonizing F108/resorcinol‑formaldehyde composites followed by KOH activation. The amount of triblock copolymer Pluronic F108 determines the resulting morphology: polydisperse carbon nanospheres (PCNS) with a microporous network, monodisperse carbon nanospheres (MCNS) featuring a hierarchical porous architecture, and agglomerated carbon nanospheres (ACNS). Comparative analyses show that MCNS deliver the highest electrochemical performance, achieving a specific capacitance of 224 F g⁻¹ at 0.2 A g⁻¹, retaining 73 % at 20 A g⁻¹, and preserving 93 % of the initial capacitance after 10 000 cycles. These results establish MCNS as a promising electrode material for high‑performance supercapacitors. Supercapacitors are emerging as key energy‑storage devices because they combine high power density, rapid charge‑discharge capability, and exceptional cycle life. The performance of a supercapacitor is fundamentally linked to the microstructure of its electrode material. Carbon materials—owing to their large surface area, tunable pore architecture, and robust chemical and mechanical stability—have become the cornerstone of advanced supercapacitor electrodes. Recent efforts have focused on engineering nanostructured carbons, including fibers, films, and spheres, to maximize accessible surface area and facilitate ion transport. While carbon spheres have been widely explored, many studies report only a single synthesis route or limited structural variations. Consequently, systematic comparisons of how distinct morphologies influence electrochemical behaviour are scarce. In this work, we employ a uniform synthetic protocol and vary the dosage of Pluronic F108 to produce three types of carbon nanospheres—PCNS, MCNS, and ACNS—each with a unique microstructure. We then evaluate how these structural differences affect capacitance, rate capability, and cycling stability. F108/resorcinol‑formaldehyde composites were prepared by hydrothermal reaction. Pluronic F108 (Mw = 14 600, PEO₁₃₂–PPO₅₀–PEO₁₃₂) served as a template, while phenolic resin acted as the carbon source. For MCNS, 0.9 g of F108 was dissolved in 30 mL deionized water, followed by the addition of 1.2 g phenol and 4.2 mL formalin (37 wt %) in 30 mL 0.1 M NaOH. The mixture was stirred at 66 °C for 10 h, then diluted three‑fold and hydrothermally treated at 130 °C for 24 h. The resulting precipitate was collected, washed, and carbonized at 700 °C for 3 h (intermediate carbonized spheres, mCNS). KOH activation (1:2 mass ratio) at 700 °C for 1 h yielded MCNS. PCNS and ACNS were obtained by adjusting the F108 loading to 0.6 g and 1.8 g, respectively, and extending the stirring times to 5.5 h and 15 h. Scanning electron microscopy (SEM; HELIOS Nanolab 600i) and transmission electron microscopy (TEM; Tecnai G2 F20 STWIX) were used to examine morphology. Nitrogen adsorption–desorption isotherms, recorded on an ASAP 2020 system at 77 K, quantified surface area and pore size distribution. Electrodes were fabricated by mixing 80 % MCNS, 10 % acetylene black, and 10 % poly( tetrafluoroethylene) (PTFE) binder, followed by pressing onto a 1 cm² area (≈ 3 mg active material). A three‑electrode cell was assembled in 6 M KOH, with platinum as counter and Hg/HgO as reference. Cyclic voltammetry (CV), chronopotentiometry (CP), and electrochemical impedance spectroscopy (EIS) were performed on a CHI660E workstation to assess capacitance, rate capability, and resistance. SEM images (Fig. 1a–c) reveal that MCNS and PCNS are well‑defined spheres, whereas ACNS form irregular aggregates. TEM confirms that MCNS are monodisperse (≈ 140 nm diameter) and possess hierarchical porosity (Fig. 1d). In contrast, PCNS display a wide size distribution (Fig. 1e) and ACNS are densely packed (Fig. 1f). These observations underscore the critical role of F108 dosage in controlling size homogeneity and dispersibility. Morphology of all samples. SEM images of a MCNS, b PCNS, and c ACNS; TEM images of d MCNS at different magnification, e PCNS, and f ACNS N₂ adsorption–desorption isotherms (Fig. 2a) exhibit type I behaviour at low relative pressure, indicating abundant micropores, while H3 hysteresis loops at higher pressures point to interparticle mesoporosity in mCNS, MCNS, and ACNS. Pore‑size distributions (Fig. 2b) show that PCNS is predominantly microporous, whereas MCNS and ACNS combine micropores with a significant mesopore fraction. Notably, KOH activation increases the pore volume from 0.423 cm³ g⁻¹ (mCNS) to 0.645 cm³ g⁻¹ (MCNS), confirming that hierarchical porosity arises from the monodisperse arrangement of the spheres. N₂ adsorption–desorption measurements of all samples. a Isotherms; b pore‑size distributions CV curves at 10 mV s⁻¹ (Fig. 3a) show quasi‑rectangular shapes with subtle humps, indicative of combined electric‑double‑layer and pseudocapacitance contributions. MCNS exhibit the largest enclosed area, reflecting superior charge storage. CP curves at 0.2 A g⁻¹ (Fig. 3b) yield a specific capacitance of 224 F g⁻¹ for MCNS, surpassing PCNS (201 F g⁻¹) and ACNS (182 F g⁻¹). Rate performance (Fig. 3c) demonstrates that MCNS retain 72.7 % of their initial capacitance at 20 A g⁻¹, compared with 70.6 % (PCNS) and 70.5 % (ACNS). The enhanced performance of MCNS is attributed to their monodisperse structure, which provides abundant mesopores for rapid ion transport, and to the hierarchical pores that offer high surface area for charge accumulation. Cycling stability (Fig. 3d) shows that MCNS maintain 93 % of the initial capacitance after 10 000 cycles, outperforming PCNS (90 %) and ACNS (93 %). EIS (Fig. 3e) reveals a low equivalent series resistance (0.76 Ω) for MCNS, lower than PCNS (1.02 Ω) and ACNS (1.08 Ω), indicating superior electronic conductivity. Bode plots (Fig. 3f) confirm that all samples approach an ideal capacitor (phase angle ≈ –90°), with MCNS exhibiting the most pronounced capacitive behaviour. Electrochemical performance of MCNS, PCNS, and ACNS. a CV curves at 10 mV s⁻¹; b CP curves at 0.2 A g⁻¹; c specific capacitance at varying current densities; d cycling test at 10 A g⁻¹; e Nyquist plots (10 mHz–10 kHz); f Bode angle plots By adjusting the Pluronic F108 loading, we successfully produced three distinct carbon nanospheres—PCNS, MCNS, and ACNS—whose porous architectures are directly linked to the template dosage. MCNS, characterized by uniform particle size and hierarchical porosity, outperform their counterparts, achieving 224 F g⁻¹ at 0.2 A g⁻¹, superior rate capability, and 93 % capacitance retention after 10 000 cycles. The synergy between monodispersity and hierarchical pore structure is the key factor driving the enhanced electrochemical performance, establishing MCNS as a compelling candidate for next‑generation supercapacitor electrodes. Agglomerated carbon nanospheres Chronopotentiometry Cyclic voltammetry Electrochemical impedance spectroscopy Equivalent series resistance Intermediate carbonized carbon nanospheres for MCNS Monodisperse carbon nanospheres Polydisperse carbon nanospheresAbstract
Background
Methods
Synthesis of Carbon Nanospheres
Microstructure Characterization
Electrochemical Measurement
Results and Discussion
Morphology

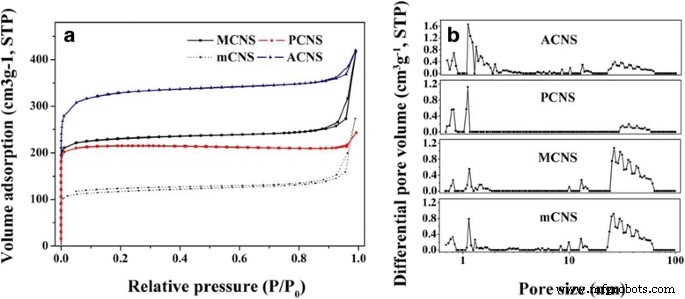
Pore Structure Analysis
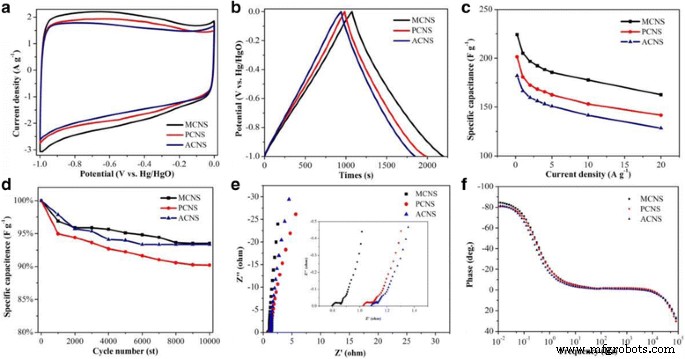
Electrochemical Performance
Conclusions
Abbreviations
Nanomaterials
- Graphene‑Polymer Composites for High‑Performance Supercapacitors: A Comprehensive Review
- Hierarchical Activated Carbon Fibers from Waste Cotton Gloves: High‑Performance Electrodes for Supercapacitors
- CoMoO4 Microspheres Synthesized Hydrothermally: Superior Electrode Performance for High‑Rate Supercapacitors
- Hierarchically Structured Kaolinite Nanospheres: Superior Methylene Blue Adsorption Performance
- Ultra‑Thin TiO₂ Nanomembranes via Atomic Layer Deposition Deliver Record‑High Capacitance for Pseudocapacitor Electrodes
- Egg‑Albumin Assisted Hydrothermal Synthesis of Mesoporous Co3O4 Quasi‑Cubes: High‑Performance Supercapacitor Electrode
- High‑Performance Hydrogen Evolution Catalysis via Nitrogen‑Doped Porous Carbon Nanosheets and Mo₂C Nanoparticles
- High-Performance Supercapacitor Electrodes from Hierarchically Porous Carbon Derived from Lignosulfonate Biowaste
- Hierarchically Porous MoS₂/C Nanospheres from Nanosheets: Exceptional Electrochemical Energy Storage
- Engineered Nickel‑Cobalt Layered Double Hydroxides: Tunable Thin‑Layer Nanosheets for Superior Supercapacitor Performance



