Hierarchical Activated Carbon Fibers from Waste Cotton Gloves: High‑Performance Electrodes for Supercapacitors
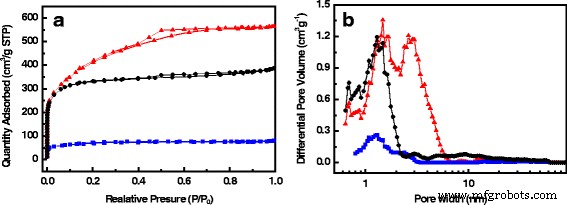
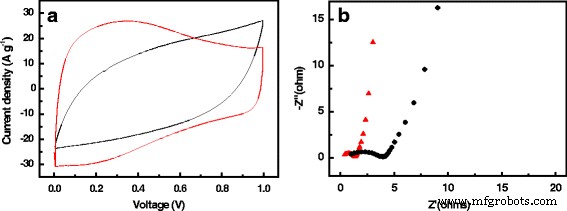
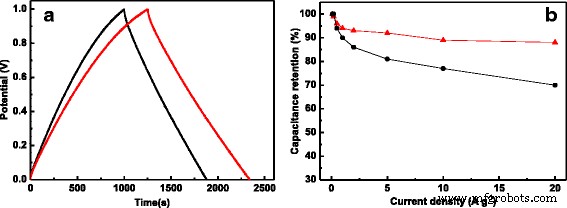
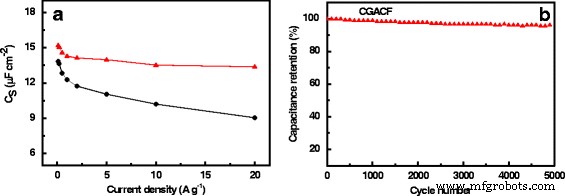
One of the most significant hurdles in leveraging biomass‑derived nanocarbons for supercapacitors is the loss of intrinsic structure during activation. In this study, we introduce a novel class of activated carbon fibers (ACFs) by using waste cotton gloves (CG) as the precursor. CG, largely composed of cellulose, naturally retains its fibrous morphology when subjected to pyrolysis and KOH activation, yielding a material with a 3‑dimensional interconnected framework and a highly hierarchical pore system. The resulting CG‑derived ACF (CGACF) exhibits an exceptional BET surface area of 1,435 m² g⁻¹, dominated by 1.3‑nm micropores and 2.7‑nm mesopores, and preserves the original fiber skeleton. This structure simultaneously minimizes electrolyte and electron diffusion resistance while maximizing charge‑storage capacity. Consequently, CGACF delivers a specific capacitance of 218 F g⁻¹ at 0.1 A g⁻¹ and maintains 88 % of its capacitance at a remarkably high current density of 20 A g⁻¹, outperforming commercial activated carbon (AC). Porous carbon materials (PCMs) have become the cornerstone of modern supercapacitors, offering high power density, rapid charge‑discharge cycles, and long‑term stability. Their performance, however, is intrinsically tied to nanostructure: well‑defined micro‑ and mesopores enable efficient electric‑double‑layer formation, while meso‑ and macropores facilitate swift ion transport. Conventional high‑performance PCMs such as carbon nanospheres, nanotubes, and nanorods require multistep syntheses and hazardous oxidants, limiting scalability. Biomass‑derived activated PCMs (APCMs) remain the most commercially viable option due to their low cost and straightforward processing. Nonetheless, many APCMs—derived from coconut shells, wood, cigarette filters, tea leaves, or fish scales—often suffer from inadequate meso/macroporosity and poor structural inheritance, leading to diminished surface‑area utilization at high rates. Selecting a precursor that inherently possesses a fibrous, interconnected architecture and can withstand activation without collapse is therefore critical. Here we exploit waste cotton gloves, a ubiquitous textile waste, as a precursor. Their cellulose fibers can be directly converted to carbon while preserving morphology through careful activation, yielding CGACF with a superior hierarchical pore network and robust conductive skeleton. CGACF was synthesized via a KOH‑assisted activation route. Cotton glove fibers were chopped to millimeter scale, pre‑carbonized at 500 °C for 3 h under N₂, then mixed with KOH (KOH/PCCG = 1.5 wt %) and carbonized at 900 °C (5 °C min⁻¹, 3 h, N₂). The product was repeatedly washed with HCl and deionized water until neutral pH, then dried at 80 °C for 12 h. A non‑activated control (CGCF) was prepared identically without KOH. Commercial AC (Kuraray) was used as a benchmark. X‑ray diffraction (D/MAX 2200 VPC) and Raman spectroscopy (Renishaw inVia 2000) confirmed the turbostratic carbon structure and increased defect density in CGACF. Morphology and nanostructure were examined by SEM (JSM‑6330F) and TEM (JEOL JEM‑2010). Nitrogen adsorption–desorption isotherms at 77 K (Micrometrics ASAP 2460) provided BET surface areas and pore size distributions via t‑plot, BJH, and DFT analyses. Electrochemical performance was evaluated in 1 M H₂SO₄ using a symmetric two‑electrode cell. The working electrode comprised active material, carbon black, and PVDF (8:1:1 wt %) pressed onto Ti foil (10 MPa) and dried at 120 °C for 12 h. Galvanostatic charge–discharge (GCD) tests were conducted from 0.1 to 20 A g⁻¹ using a Neware CT2001A. Cyclic voltammetry (CV) at 200 mV s⁻¹ and electrochemical impedance spectroscopy (EIS) (5 mV, 0.001–100 kHz) were recorded on an IM6ex workstation. Specific capacitance (C_g) was calculated from discharge curves using the standard formula. XRD patterns of CGCF and CGACF (Fig. 2a) display broad peaks at 23.5° and 44° (2θ), indicative of (002) and (100) planes of turbostratic graphite. The diminished peak intensity in CGACF reflects the extensive porosity introduced by KOH activation. Raman spectra (Fig. 2b) show a higher I_D/I_G ratio for CGACF, confirming the increased defect density that correlates with enhanced porosity. Schematic diagram for the preparation of CGACF. SEM and TEM images (Fig. 3) reveal that the raw CG exhibits ~3 µm fibers with smooth surfaces. After pre‑carbonization, fiber diameters shrink to ~2 µm, maintaining smoothness. KOH activation preserves the fibrous skeleton while creating a rough, 3‑D interconnected surface that hosts abundant micro‑ and mesopores (Fig. 3c‑d). TEM confirms numerous surface nanopores in CGACF, whereas commercial AC shows large, non‑porous carbon blocks (Fig. 3f). SEM and TEM images of (a) raw CG, (b) CGCF, (c) CGACF, (d) CGACF surface, (e) CGACF TEM, and (f) commercial AC. N₂ adsorption–desorption isotherms (Fig. 4a) demonstrate that CGACF has a significantly higher uptake at low P/P₀, reflecting an increased microporosity. A hysteresis loop at intermediate P/P₀ confirms the presence of small mesopores. DFT pore size distribution (Fig. 4b) shows peaks at 1.3 nm (micropores) and 2.7 nm (mesopores), aligning with TEM observations. In contrast, commercial AC shows a predominantly microporous surface area of 1,282 m² g⁻¹. N₂ adsorption–desorption isotherms and PSD curves for CGACF (red), CGCF (blue), and AC (black). Electrochemical evaluation confirms the advantageous structure of CGACF. CV curves at 200 mV s⁻¹ exhibit a near‑rectangular shape for CGACF, whereas AC shows distortion (Fig. 5a), indicating superior ion accessibility. EIS Nyquist plots (Fig. 5b) reveal a lower series resistance (0.42 Ω vs. 1.03 Ω) and charge‑transfer resistance (0.94 Ω vs. 2.90 Ω) for CGACF, underscoring efficient electrolyte/electron transport. CV at 200 mV s⁻¹ and EIS of CGACF (red) and AC (black). GCD tests (Fig. 6) demonstrate that CGACF achieves 218 F g⁻¹ at 0.1 A g⁻¹, surpassing AC (175 F g⁻¹). Even at an extreme current density of 20 A g⁻¹, CGACF retains 88 % of its capacitance (192 F g⁻¹), whereas AC drops to 70 %. GCD curves at 0.1 A g⁻¹ and capacitance retention vs. current density for CGACF (red) and AC (black). Capacitance per surface area (C_S) serves as an indicator of surface‑area utilization. Both CGACF and AC show similar C_S (~13–15 µF cm⁻²) at low current density, but CGACF maintains higher C_S (>13 µF cm⁻²) at 20 A g⁻¹, whereas AC falls to 9 µF cm⁻². Cycling stability tests over 5,000 cycles at 1 A g⁻¹ reveal 96.3 % capacitance retention for CGACF (Fig. 7). C_S vs. current density and cycling stability for CGACF (red) and AC (black). In summary, CGACF’s exceptional performance arises from two synergistic features: (1) a hierarchical micro‑/mesoporous network delivering a 1,435 m² g⁻¹ surface area, and (2) a robust, fiber‑like skeleton with 3‑D interconnected surfaces that facilitate rapid ion/electron transport, especially at high rates. We have successfully fabricated a new class of activated carbon fibers with hierarchical nanostructure from waste cotton gloves. The CG‑derived ACF preserves the original fiber morphology, achieving a surface area of 1,435 m² g⁻¹ composed of 1.3‑nm micropores and 2.7‑nm mesopores, and a conductive 3‑D framework. As a result, CGACF delivers a specific capacitance of 218 F g⁻¹ at 0.1 A g⁻¹ and maintains 88 % of this value at 20 A g⁻¹, outperforming commercial AC. Moreover, it retains 96.3 % capacitance after 5,000 cycles at 1 A g⁻¹. This work demonstrates that waste cotton gloves are a viable, low‑cost precursor for high‑performance, biomass‑derived supercapacitor electrodes.Abstract
Background
Experimental
Preparation of CGACF
Structure Characterization
Electrochemical Measurements
Results and Discussions


Conclusions
Nanomaterials
- Carbon Fiber: Composition, Manufacturing, and Future Applications
- Monodisperse Carbon Nanospheres with Hierarchical Porosity Deliver Superior Supercapacitor Performance
- Polyaniline‑Coated Activated Carbon Aerogel/Sulfur Composite Yields Superior Lithium‑Sulfur Battery Performance
- Nitrogen‑Doped Porous Carbon Networks Derived from Graphitic Carbon Nitride for High‑Performance Oxygen Reduction
- Hemp‑Stem‑Derived Nanoporous Carbon Anodes for High‑Capacity Lithium‑Ion Batteries
- High-Performance Supercapacitor Electrodes from Hierarchically Porous Carbon Derived from Lignosulfonate Biowaste
- Sustainable Activated Carbon from Kusha Grass (Desmostachya bipinnata) Enhances Supercapacitor Performance
- High‑Performance Date Palm Fiber: Sustainable & Cost‑Effective from Agricultural Waste
- Vision Composite Products Partners with IDI Composites for High‑Performance Carbon‑Fiber Forged Wheels
- Hexcel & Metyx Forge Alliance to Deliver High‑Performance Carbon Fiber Pultrusion for Wind Energy



