TiO₂ Nanofluids: Applications, Challenges, and Future Directions – Part 2
Abstract
The field of nanofluids has experienced rapid growth due to their exceptional heat and mass transport properties, fluidity, and dispersion stability. These attributes make them attractive for a variety of energy systems, including solar collectors, refrigeration, heat pipes, and energy storage. In this second part of the review, we consolidate recent findings on TiO₂ nanofluids, highlighting their applications, identifying prevailing challenges, and proposing directions for future research. Together, these two comprehensive reviews aim to serve as a practical reference for researchers seeking to stay current with the state of TiO₂ nanofluid technology and to inspire new investigations.
Review
Background
Part 1 of this series examined the synthesis, stability, and fundamental properties of TiO₂ nanofluids. The current section builds upon that foundation by focusing on their practical deployment in energy systems. Numerous studies have explored TiO₂ nanofluids in solar collectors, refrigeration, energy storage, heat pipes, and other thermal applications such as automotive radiators, PV/T hybrid systems, and combined heat‑and‑power plants [12–34]. While prior work has summarized heat‑transfer characteristics across conduction, boiling, and natural convection [35], a holistic overview of TiO₂ nanofluid applications remains lacking. This review therefore integrates recent experimental and theoretical results to provide a comprehensive picture of TiO₂ nanofluids in energy‑related contexts.
Application in Enhancing the Thermal Conductivity
Nanofluids derive their performance advantages from the addition of nanoparticles, and thermal conductivity is a key metric for assessing their potential. While many reviews discuss thermal conductivity in the context of physical properties, its direct impact on application performance warrants dedicated attention. This section evaluates the factors that influence thermal conductivity and presents a visual synthesis of experimental data for TiO₂ nanofluids.
Experimental and theoretical investigations consistently demonstrate that incorporating TiO₂ nanoparticles can markedly increase the thermal conductivity of base fluids. Influencing parameters can be grouped into three categories:
- Internal factors – particle type, loading [36, 37], size [38], shape [39], structure [40], base‑fluid type [41], and surfactant or pH regulator [42, 43].
- External factors – temperature [40], supersonic vibration time [44], storage time [45], or operational time [46].
- Micro‑scale factors – surface charge state [47], particle clustering [48], interfacial nanolayer [49], Brownian motion [50], aggregation [51], interfacial thermal resistance, and mass‑difference scattering [52].
To provide clearer insight, Figure 1 collates the percentage increase in thermal conductivity reported across the literature, illustrating the breadth of observed effects.
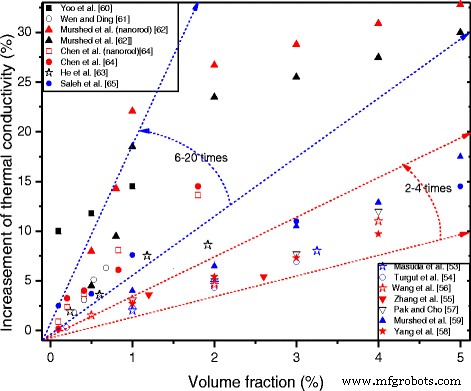
Volume‑fraction dependence of the thermal conductivity of TiO₂–water nanofluids reported in the literature.
Differences in reported enhancements arise from variations in particle size, shape, surfactant usage, pH, temperature, and other experimental conditions. Some studies report modest increases (2–4 % per volume loading) [53–59], while others observe substantial boosts (6–20 % per volume loading) [60–65]. In certain cases, TiO₂ addition yields negligible improvements, as noted by Utomo et al. [66] who found lower conductivity than predicted due to high dispersant content.
Particle Loading Effect
Figure 1 demonstrates that, regardless of the magnitude of the effect, TiO₂ nanoparticles consistently enhance base‑fluid conductivity. However, the extent of improvement varies widely across studies, underscoring the importance of controlled experimental conditions.
Particle Shape Effect
Shape and size influence is less frequently studied. Murshed et al. [62] compared rod‑shaped (10 × 40 nm) and spherical (15 nm) TiO₂ in water, finding a slightly higher enhancement for rods (≈33 % vs. 30 %). Chen et al. [64] evaluated spherical TiO₂ and TiO₂ nanotubes (10 × 100 nm) in water and ethylene glycol, reporting comparable improvements but exceeding predictions from the Hamilton–Crosser model.
Temperature Effect
Temperature is a pivotal external factor. Wang et al. [67] showed that higher temperatures amplify the conductivity benefit, aligning with models that incorporate Brownian motion and micro‑convection. Reddy et al. [68] confirmed a concurrent rise in conductivity with both particle loading and temperature (0.2–1.0 % loading, 10–60 °C). Yang et al. [58] observed a similar trend in ammonia–water nanofluids. Nevertheless, some studies report minimal temperature influence [54, 69], likely due to competing effects such as changes in particle dispersion, interfacial layers, or viscosity.
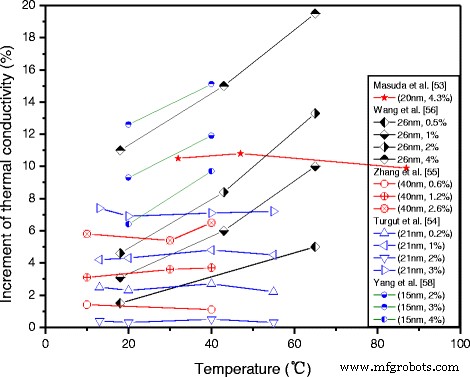
Temperature influence on the conductivity enhancement of TiO₂ nanofluids reported in different studies.
Base Fluid Effect
The base fluid’s intrinsic properties strongly affect the magnitude of conductivity enhancement. Chen et al. [64] found higher gains in ethylene glycol compared to water for both spherical and tubular TiO₂. Reddy et al. [68] reported a gradient of enhancement: water (0.649 %–5.01 %) < EG/W (40 %:60 %) < EG/W (50 %:50 %). Contrastingly, Cabaleiro et al. [41] observed lower enhancements for EG, PG, and paraffin oil relative to water, attributing the discrepancy to viscosity and interfacial effects.
Surfactant Effect
Surfactants can stabilize nanoparticle dispersion and influence conductivity. Saleh et al. [65] evaluated SDS, CTAB, and Span‑80, finding SDS yielded the largest improvement, followed by CTAB and Span‑80. Other studies note that surfactants may depress conductivity, especially at high concentrations [66]. Yang et al. [58] highlighted the role of ammonia (pH) in enhancing stability and, consequently, conductivity.
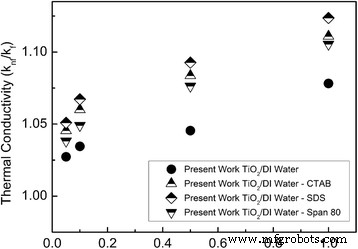
Effect of different surfactants on the conductivity of TiO₂–water nanofluids [65].
Sonication Effect
Ultrasonic treatment enhances dispersion and can influence conductivity. Ismay et al. [71] reported maximal conductivity when the pH approached 7 and after 2 h of sonication. Sonawane et al. [70] demonstrated that conductivity increased up to 60 min of sonication, beyond which aggregation reduced performance.
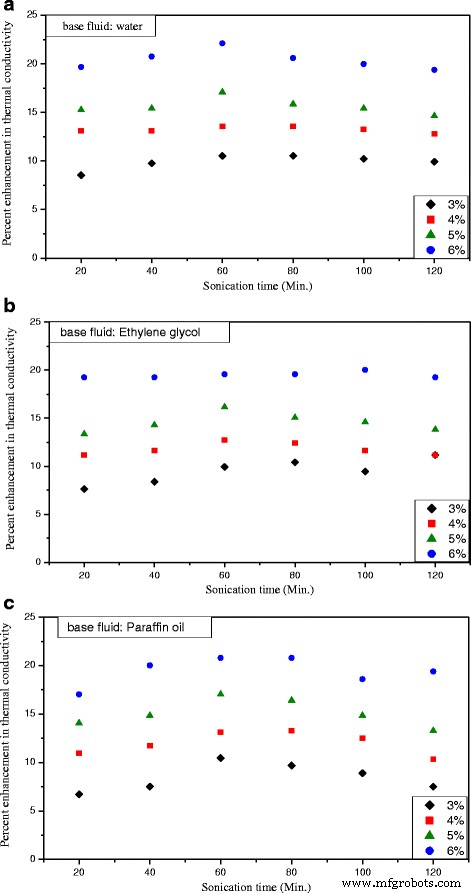
Percent enhancement in conductivity versus sonication time: (a) water, (b) ethylene glycol, (c) paraffin oil [70].
Theoretical Studies
Numerous models exist to predict nanofluid conductivity, often incorporating interfacial layers, Brownian motion, particle size, aspect ratio, and aggregation. Table 1 summarizes specialized TiO₂ nanofluid models, highlighting their reliance on experimental fitting and variance in applicability. Due to inherent experimental scatter, a pragmatic approach remains to conduct preliminary measurements for each application case.
Solar Absorption
Solar thermal collectors suffer from limited absorption efficiency when using conventional fluids. TiO₂ nanofluids offer superior thermal and optical properties, enhancing collector performance. Khullar et al. [83] illustrated a nanofluid‑based concentrating solar water‑heating system, predicting substantial emission reductions and energy savings. Chaji et al. [84] reported a 2.6–7 % efficiency increase in a flat‑plate collector using TiO₂ nanofluids. Said et al. [85] achieved a 16.9 % exergy efficiency with a 0.76 vol % TiO₂ loading at 0.5 kg min⁻¹ flow.
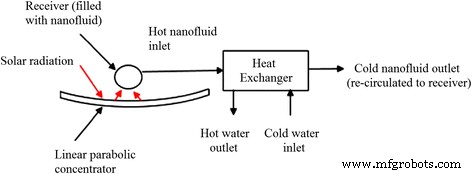
Schematic of a nanofluid‑based concentrating solar water‑heating system [83].
Numerical studies confirm that TiO₂ and other nanofluids improve collector efficiency and reduce greenhouse gas emissions [86–89]. Optical analyses show TiO₂ nanofluids exhibit higher absorptivity across visible wavelengths compared to Al₂O₃ [88], with absorption scaling linearly with volume fraction. The optimum TiO₂ loading for solar collectors is typically below 0.1 vol % to balance absorption and thermal conductivity gains [90].
Refrigeration
Nanoparticles incorporated into refrigerants—often termed nano‑refrigerants—enhance heat transfer, reduce energy consumption, and improve overall system performance. TiO₂ nanoparticles are compatible with a wide range of refrigerants (R134a, R600a, R436a, R12, R22, R410a, etc.) [94–103]. Experimental results demonstrate up to 26 % energy savings and 25 % CO₂ reduction at 0.1 wt % TiO₂ loading [96, 103]. However, long‑term stability remains a concern; alternating condensation–evaporation cycles can cause a 28–73 % degradation in nanoparticle suspension [15]. Future research must focus on real‑time re‑dispersion techniques and surfactant optimization to mitigate sedimentation.
Energy Storage
TiO₂ nanofluids can improve latent‑heat material performance by increasing thermal conductivity. Studies on BaCl₂ aqueous solutions reveal 12–16 % conductivity gains at 1 % TiO₂ loading [104, 105]. In solid PCM composites, TiO₂ addition boosts conductivity by 12–80 % while slightly reducing latent heat; optimal loadings are 0.5–5 % mass [106–109]. In paraffin, 7 wt % TiO₂ increases conductivity by 13 % but decreases latent heat by 9 % [111]. These findings suggest TiO₂‑enhanced PCMs are promising for high‑temperature energy storage, provided the trade‑off between conductivity and latent heat is acceptable.
Heat Pipes
Nanofluids improve boiling heat transfer and critical heat flux in heat pipes. Zhou et al. [114] observed reduced start‑up temperatures and shorter start‑up times for TiO₂‑filled gravity‑driven heat pipes. Saleh et al. [65] confirmed that nanofluids adhere to the Brownian motion model and achieve optimal performance at a 45° inclination and 60 % fluid charge. In thermosyphon heat‑pipe heat exchangers, 3 wt % TiO₂–methanol delivers 30–33 % energy savings under humid conditions [115].
Mass Transfer
TiO₂ nanofluids enhance mass‑transfer coefficients in gas‑liquid systems. MDEA‑based nanofluids increased CO₂ absorption by 3–14 % [116]. In falling‑film ammonia absorption, anatase TiO₂ raised the rate by 10 % [117], while rutile TiO₂ achieved 60.8 % improvement in fin‑tube configurations [118]. In electrolytes, 0.015 vol % TiO₂ and 0.01 vol % γ‑Al₂O₃ improved mass transfer by 18 % and 10 % respectively [119]. These enhancements are attributed to reduced particle clustering and increased Brownian activity.
Coolant of Milling
TiO₂–ethylene glycol nanocoolants reduce workpiece temperature by 30 % and extend tool life by 40 % during stainless‑steel milling [121, 122]. SEM/EDX analysis reveals a protective TiO₂‑rich oxide layer on the tool edge, mitigating wear. These results underscore the dual benefits of thermal and tribological performance in machining applications.
Challenges and Future Works
Challenges
Despite promising performance, TiO₂ nanofluids face several hurdles:
- Long‑term stability – sedimentation and agglomeration during extended operation degrade performance. Real‑time re‑dispersion devices and surfactant control are required.
- Pressure drop – increased viscosity leads to higher pumping power. Strategies to balance conductivity gains against flow resistance are essential.
- Surfactant side effects – foaming and potential foaming under heating or flow conditions necessitate careful surfactant selection.
- Environmental impact – waste management and potential ecological toxicity of nanoparticle‑laden fluids must be addressed.
- Standardized evaluation – unified metrics for stability, adhesion, and property sustainability are lacking.
Future Works
Key research priorities include:
- Developing high‑quantity, high‑temperature, long‑term stable TiO₂ nanofluids.
- Designing real‑time re‑dispersion systems (ultrasonic or mechanical).
- Quantifying surfactant effects on thermophysical properties.
- Optimizing pressure drop while maximizing heat‑transfer enhancement.
- Establishing environmental safety protocols for nanoparticle disposal.
- Creating comprehensive performance indices covering stability, adhesion, and sustainability.
Conclusions
This review consolidates recent advances in TiO₂ nanofluid applications and highlights the critical challenges that must be addressed for industrial adoption. While particle loading positively influences thermal conductivity, the magnitude varies across studies due to complex micro‑scale mechanisms. TiO₂ nanofluids show strong potential in solar collectors, refrigeration, energy storage, heat pipes, and machining. However, long‑term performance, pressure‑drop penalties, and stability concerns remain pivotal barriers. Future research should focus on stability, real‑time re‑dispersion, surfactant optimization, and robust evaluation frameworks to unlock the full potential of TiO₂ nanofluids.
Nanomaterials
- Molybdenum and Its Alloys: Key Applications in Material Processing
- Fluorescent Nanomaterials: Advances, Synthesis, and Biomedical Applications
- TiO₂ Nanofluids: Part 1 – Preparation and Key Properties
- Optimizing Gold Nanoparticle Placement in TiO₂ Enhances Dye‑Sensitized Solar Cell Efficiency
- Chromium‑Doped Titanium Dioxide: A Novel Colored Cool Pigment with High Near‑Infrared Reflectance
- Influence of Peptizing Acid on TiO₂ Phase Composition and Photocatalytic Efficiency: A Comparative Study of Sulfuric, Nitric, and Acetic Acids
- Titania Nanotube Arrays via Electrochemical Anodization: Synthesis, Modifications, and Biomedical Applications
- Enhanced Up‑Conversion TiO₂ Nanomaterial Boosts Perovskite Solar Cell Efficiency to 16.3 %
- Enhanced TiO₂ Catalysis and Antimicrobial Efficacy via Nitrogen and Carbon Nitride Co‑Doping
- Unlocking Blockchain’s Potential in Industrial Automation: Opportunities and Obstacles



