TiO₂ Nanofluids: Part 1 – Preparation and Key Properties
Abstract
Nanofluids, suspensions of sub‑100‑nm particles, have become a leading research frontier over the past three decades. While broad reviews cover many nanofluid systems, a focused analysis of TiO₂ nanofluids—renowned for their exceptional dispersibility, chemical stability, and non‑toxicity—offers actionable insights for heat‑transfer and energy‑related applications. This first installment synthesizes recent advances in TiO₂ nanofluid preparation, stability, and core physical properties, setting the stage for the forthcoming discussion on thermal conductivity.
Review
Background
Development of Nanofluids
Conventional liquid coolants exhibit thermal conductivities far below those of solid metals. Introducing solid nanoparticles into a base fluid can elevate heat‑transfer performance, yet millimeter‑scale particles suffer from poor dispersibility, aggregation, and sedimentation, compromising efficiency and increasing pumping energy. The breakthrough came with Choi’s 1995 proposal of nanofluids—dilute suspensions of <100‑nm particles—promising enhanced thermal properties while maintaining a single‑phase behavior.
Nanofluid research spans preparation & stability, thermal conductivity, viscosity, heat‑transfer experiments, engineering applications, and theoretical modeling. Publication rates have surged, with 2016 accounting for 21.9% of the past two decades’ output, underscoring the field’s rapid expansion and the challenge of synthesizing comprehensive reviews.
Advantages of TiO₂ Nanofluids
TiO₂ nanofluids have attracted particular interest due to several key attributes:
- Non‑toxic, widely used in printing, cosmetics, and air purification.
- Excellent chemical stability—resistant to acids, bases, and most organic solvents.
- Industrial‑grade production makes TiO₂ nanoparticles cost‑effective.
- Superior dispersibility in both polar and non‑polar media, especially when combined with appropriate dispersants.
Studies such as Taghizadeh‑Tabari et al. [63] and Yang et al. [65] confirm TiO₂ nanofluids’ stability and applicability in heat exchangers and absorption refrigeration. The next sections systematically review the preparation techniques, stability mechanisms, and key physical properties that underpin TiO₂ nanofluid performance.
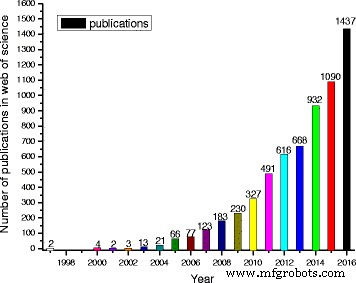
Number of publications containing “nanofluids or nanofluid” in title retrieved from “web of science”
Preparation of TiO₂ Nanofluids
One‑Step vs. Two‑Step Methods
Preparation strategies are typically categorized as one‑step or two‑step. One‑step methods—vapor deposition, laser ablation, submerged arc, or chemical synthesis—generate nanoparticles directly within the base fluid, eliminating drying and re‑dispersion steps that can trigger agglomeration. However, these techniques often require high temperatures, specialized equipment, or specific chemical environments, limiting their practicality for many systems.
Two‑step methods, conversely, synthesize dry TiO₂ powders (via coprecipitation or precursor conversion) and subsequently disperse them in the target fluid. This approach benefits from industrial‑scale production, versatile base fluid compatibility, and well‑established dispersion protocols (surfactants, pH adjustment, mechanical or ultrasonic agitation). Figure 6 illustrates the standard two‑step workflow.
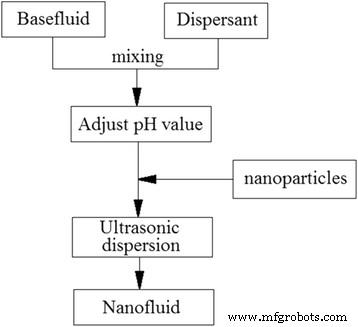
A typical procedure of the two‑step preparation of TiO₂ nanofluids [35]. Reproduced with permission from Elsevier
Vapor Deposition
In vapor deposition, bulk TiO₂ is evaporated under reduced pressure and condensed onto a swirling liquid film. Lee et al. [68] demonstrated a one‑step pulsed‑wire evaporation (PWE) system that produced TiO₂–EG nanofluids by direct plasma interaction with the base fluid.
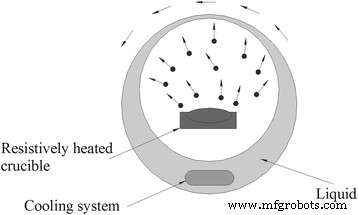
A typical device of vapor deposition for nanofluid synthesis. Redrawn based on reference [67]
Submerged Arc Method
The submerged arc technique achieves even higher temperatures by vaporizing TiO₂ in vacuum and rapidly cooling the vapor with an isolated liquid. Chang et al. [69] introduced a system that yielded highly stable, Newtonian TiO₂ nanofluids. Zhang et al. [70] refined this setup, improving particle size control and reproducibility.
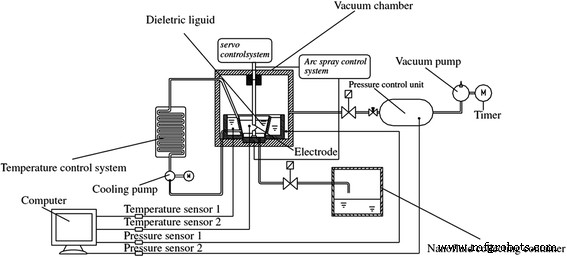
Schematic of the improved submerged arc nanofluid synthesis system (ISANSS) [40]. Reproduced with permission from The Japan Institute of Metals and Materials
Chemical One‑Step
Chemical routes—coprecipitation or precursor conversion—produce TiO₂ nanoparticles in situ by reacting titanic salts with ammonia or other bases. While these methods can generate highly stable nanofluids, the need for precise pH, temperature, and reactant control, as well as potential by‑products, often limits their suitability for industrial applications. Typically, the powders are then dried and re‑dispersed, which is effectively a two‑step process.
Two‑Step Dispersal Strategies
Successful dispersion hinges on three complementary techniques:
- Surfactant addition—CTAB, SDBS, SDS, PVP, oleic acid, PEG, and others create electrostatic or steric barriers that mitigate aggregation. Mo et al. [77] reported 286‑h stability for SDS‑stabilized TiO₂‑water nanofluids.
- pH adjustment—Tuning the base fluid to pH ≈ 11 generates a high zeta potential (~45 mV) that promotes electrostatic repulsion. He et al. [81] and Sen et al. [83] achieved multi‑month stability at this pH.
- Physical agitation—mechanical stirring, ultrasonic cavitation, or bead milling can break agglomerates. Continuous‑wave sonication (25 kHz, 48 h) outperformed intermittent pulses for water‑based TiO₂ nanofluids [87].
Combined treatments—surfactant, pH, and ultrasound—often yield the most robust dispersions, as demonstrated by Ghadimi et al. [90], who achieved one‑year stability at 0.1 wt.% TiO₂ loading.
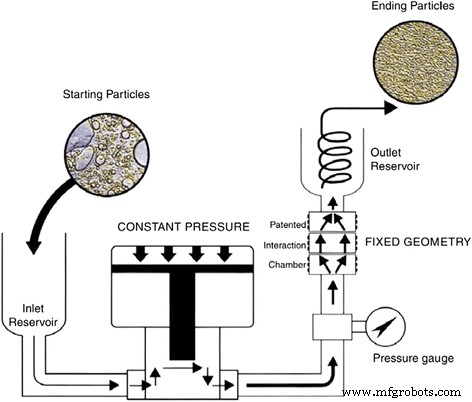
Schematic diagram of the high‑pressure homogenizer for producing nanofluids [99]. Reproduced with permission from Elsevier
Stability of Nanofluids
Stability assessment is critical: unstable suspensions lead to sedimentation, fouling, increased pumping power, and reduced heat‑transfer efficiency. Evaluation methods include visual sedimentation, zeta potential, particle size distribution, absorbance changes, and microscopy. Studies consistently show that surfactant‑assisted, pH‑optimized, and sonicated nanofluids exhibit the longest shelf lives—ranging from several hours to one year.
Key findings:
- Ghadimi et al. [90] achieved 1‑year stability at 0.1 wt.% TiO₂ using a surfactant–pH–ultrasonic combo.
- He et al. [81] and Longo et al. [87] reported multi‑month stability at pH 11 without surfactants.
- Physical agitation alone can sustain stability for up to 6 months when particle loading is low and the base fluid has high viscosity (e.g., R134a, mineral oil) [101, 102].
Physical Properties of TiO₂ Nanofluids
Viscosity
Viscosity dictates pumping power and flow dynamics. TiO₂ nanofluid viscosities increase with particle loading, but the magnitude varies widely across studies due to differing base fluids, particle sizes, and preparation protocols. Some works report linear growth, while others observe quadratic behavior.
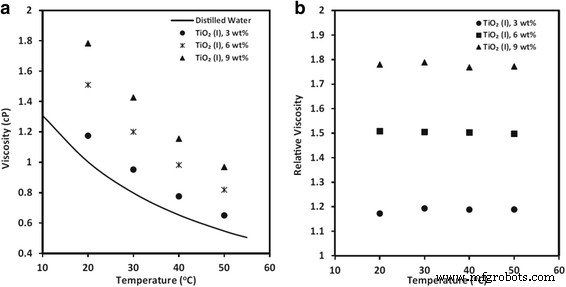
Dynamic and relative viscosity of TiO₂‑water nanofluids at various temperatures [93]. Reproduced with permission from Springer
Temperature generally reduces viscosity, yet the relative viscosity remains nearly constant across a range of temperatures for many formulations. Particle shape influences viscosity more strongly than size: rod‑like TiO₂ particles elevate viscosity by up to 82 % at low loadings, whereas spherical particles increase it by 0.5–23 % [64, 119].
Surfactants can either raise or lower viscosity. Trioxadecane acid and poly‑carboxylate surfactants increased viscosity significantly at high loading, whereas PEG600 actually reduced viscosity at low concentrations [93, 120]. The effect depends on surfactant type, concentration, and the underlying base fluid.
Base fluid choice matters: water‑based nanofluids typically exhibit higher relative viscosity increases than ethylene glycol or organic oils. High‑viscosity bases dampen the relative rise, making them attractive for applications where low pumping losses are critical [110, 123].
Newtonian behavior is common but not universal; rheology depends on particle loading, base fluid, temperature, and shear rate. Non‑Newtonian characteristics appear in water‑based TiO₂ systems at low shear rates or elevated temperatures [118].
Surface Tension
Surface tension studies are comparatively sparse. Most reports find that TiO₂ additions cause only minor changes (≤ 2 %) in surface tension, whereas surfactants exert a much larger effect. Liu et al. [125] observed a 1.6 % increase at 1 wt.% loading, and Hu et al. [126] noted a slight rise that diminishes with temperature. Chinnam et al. [130] provided a correlation showing surface tension decreases with both temperature and volume loading, with smaller particles contributing to lower tension.
Surfactant‑induced surface tension reduction can enhance boiling performance in nano‑refrigerants [131], highlighting the importance of careful surfactant selection.
Conclusions
For TiO₂ nanofluids, the two‑step preparation route—combined with post‑treatments such as surfactant addition, pH optimization, and ultrasonic or mechanical agitation—offers the most practical and scalable approach. While one‑step methods can yield superior stability in theory, their operational constraints (high temperature, specialized equipment, or complex chemistry) limit widespread adoption.
Viscosity increases positively with particle loading, but the effect is modulated by particle morphology, surfactant chemistry, base fluid viscosity, and temperature. No universal model reliably predicts viscosity across all systems; experimental calibration remains essential. Surface tension is primarily influenced by surfactants rather than nanoparticles; low‑concentration surfactants can reduce tension without significantly raising viscosity or causing foaming.
Future work should focus on standardizing stability metrics, exploring surfactant‑nanoparticle synergies that balance dispersion and rheology, and integrating TiO₂ nanofluids into real‑world heat‑transfer devices to validate laboratory findings.
Nanomaterials
- Cobalt‑Doped FeMn₂O₄ Spinel Nanoparticles: Size‑Controlled Synthesis and Magnetic Behavior
- TiO₂ Nanofluids: Applications, Challenges, and Future Directions – Part 2
- Chromium‑Doped Titanium Dioxide: A Novel Colored Cool Pigment with High Near‑Infrared Reflectance
- Engineering n-Type GeBi Thin Films by Molecular Beam Epitaxy: Crystalline Control and Infrared/THz Optical Performance
- Optimizing Zinc Oxide/Porous Anodic Alumina Composite Films for Superior Antibiofilm Performance
- Pomegranate‑Shaped Fe₂O₃/C Core‑Shell Nanocomposites: A One‑Step Hydrothermal Route for High‑Performance Li‑Ion Battery Anodes
- Fabrication and Magnetic Behavior of Nd/Ferromagnet/PA66 Three-Layer Coaxial Nanocables
- High‑Quality In₂S₃ Quantum Dots Synthesized at Room Temperature for Ultra‑Sensitive UV Photodetectors
- Synthesis, Structure, and Superior Capacitance of Sulfur‑Nitrogen Co‑Doped Graphene Quantum Dots
- Hydrothermally Synthesized Nano-Copper Sulfide: Superior Adsorption of 17α-Ethynyl Estradiol



