Fluorescent Nanomaterials: Advances, Synthesis, and Biomedical Applications
Abstract
Over the past two decades, nanotechnology has propelled the emergence of diverse fluorescent nanomaterials (FNMs) with unique optical properties, enabling breakthroughs in bioimaging and fluorescence‑based detection. Despite their promise, challenges such as limited photoluminescence efficiency and suboptimal selectivity continue to restrict clinical translation. This review surveys key FNM classes—quantum dots, carbon‑based dots, nanotubes, graphene derivatives, noble metal nanoparticles, silica particles, phosphors, and organic frameworks—highlighting recent synthesis innovations, biomedical applications, and prevailing limitations. We also outline future directions for overcoming synthesis and application hurdles, with an eye toward clinical deployment.
Introduction
Traditional organic dyes suffer from cytotoxicity, poor biocompatibility, and limited photostability, driving the development of FNMs as next‑generation imaging agents. The physicochemical attributes of FNMs—size, shape, surface chemistry—dictate their optical behavior and biological performance, making controlled synthesis a research priority. Recent advances in doping strategies, surface functionalization, and green synthesis methods have improved biocompatibility and photoluminescence, yet challenges remain in achieving high quantum yield (QY) and selective tissue targeting. This review focuses on state‑of‑the‑art synthesis techniques and biomedical uses of FNMs, providing insight into their developmental trajectory.
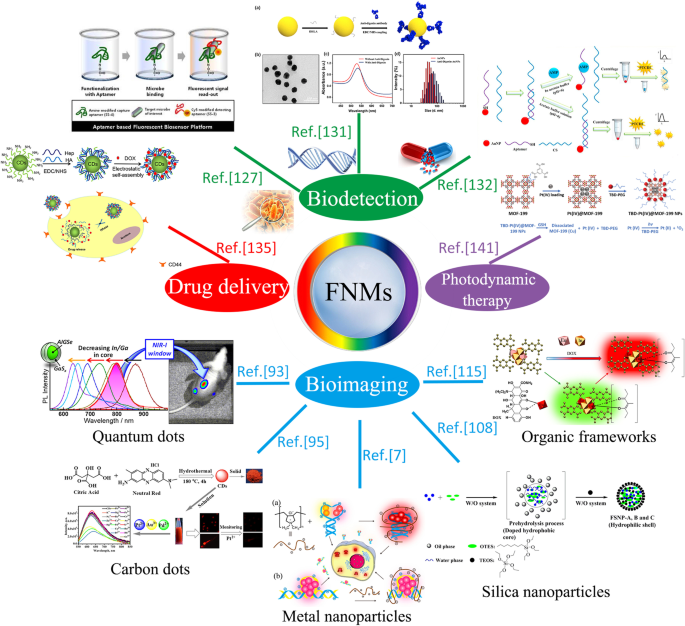
The overview diagram of biomedical applications of fluorescent nanomaterials
Synthesis of Fluorescent Nanomaterials
Quantum Dots (Semiconductor Crystals)
Quantum dots (QDs) exhibit broad absorption, narrow emission, high quantum yield, and resistance to photobleaching, making them ideal for imaging. Quantum confinement arises when electron–hole pairs are restricted to dimensions smaller than the Bohr radius. Early syntheses (1982) used glass matrix growth; modern approaches include direct adsorption, linker‑assisted adsorption, and in‑situ methods. Surface coatings—such as inorganic shells or ligand exchange—enhance colloidal stability and reduce toxicity. Recent work on Ga³⁺‑doped Ag–In–Se QDs eliminated defect‑state emission, enabling clear in‑vivo imaging with high signal‑to‑background ratios. HgTe QDs are promising for IR bioimaging due to low dark currents and high detectivity.
Carbon Dots
Carbon dots (CDs) are sub‑10 nm carbon nanostructures discovered in 2004. They offer water solubility, low cytotoxicity, excitation‑dependent multicolor emission, and facile surface chemistry, positioning them as superior to semiconductor QDs for many biomedical tasks. Synthesis routes are broadly divided into chemical (electrochemical, acidic oxidation, hydrothermal, microwave, solution chemistry, supported) and physical (arc discharge, laser ablation, plasma). For instance, electrochemical oxidation of graphite in phosphate buffer produces 3–5 nm graphene quantum dots with green PL. Hydrothermal carbonization of chitosan yields highly amino‑functionalized CDs with 7.8% QY without harsh acids. Co‑doping with nitrogen and sulfur can raise QY to >80%, as demonstrated by microwave‑assisted L‑cysteine/citric acid CDs. Functionalization strategies (e.g., sulfoxide or carbonyl groups) enable near‑infrared (NIR) emission, expanding tissue‑penetration depth.
Chemical Synthetic Methods
Electrochemical synthesis, acidic oxidation, hydrothermal, microwave, and solution‑chemistry approaches dominate CD production due to their tunability and scalability. Electrochemical routes (e.g., oxidizing a graphitic column electrode) produce CDs with distinct blue/yellow PL and low cytotoxicity. Acidic oxidation of carbon nanotubes yields 3–4 nm CDs emitting bright yellow fluorescence in saline, with excellent photostability. Microwave or ultrasonic methods enable rapid, one‑step synthesis; for example, ionic‑liquid‑derived CDs emerge in a few minutes with simultaneous functionalization. Hydrothermal carbonization of biopolymers (glucose, chitosan, citric acid) offers green, additive‑free production, preserving biocompatibility.
Physical Synthetic Methods
Arc discharge, laser ablation, and plasma treatment produce CDs from carbon precursors with controlled size and defect density. Arc‑discharged soot can be purified to isolate highly fluorescent CDs. Laser ablation in aqueous media yields monodisperse CDs, while oxygen plasma can activate graphene layers, converting them into fluorescent nanoparticles.
Carbon Nanoparticles
Carbon nanoparticles (>20 nm) combine photostability and low cytotoxicity with ease of synthesis via hydrothermal, microwave, or laser ablation routes. Recent studies demonstrate that controlled carbonization of resorcinol or hyaluronic acid yields red‑emissive, biocompatible particles suitable for in‑vivo imaging.
Carbon Nanotubes
Single‑walled (SWCNTs) and multi‑walled (MWCNTs) nanotubes possess high aspect ratios and strong NIR absorption, ideal for deep‑tissue imaging. Functionalization strategies—covalent or non‑covalent—improve water solubility and preserve fluorescence. Recent reports show that dithiothreitol enhances QY of SWCNTs to levels comparable with QDs, while amphiphilic polymers enable high‑contrast imaging in live brain tissue.
Graphene‑Based Nanomaterials
Graphene oxide (GO) and reduced GO (rGO) exhibit weak intrinsic fluorescence, but conjugation with fluorescent dyes or intrinsic doping can generate bright, biocompatible emitters. Bottom‑up synthesis (epitaxial growth, CVD) and top‑down exfoliation (mechanical, solvent, electrochemical) allow size control. Intrinsically photoluminescent graphene derivatives functionalized with amines or dodecylamine provide tunable emission and low cytotoxicity, enabling bioimaging applications.
Metal Nanomaterials
Noble metal nanoparticles—Au, Ag, Cu—combine low cytotoxicity with plasmonic or fluorescent properties. Citrate‑stabilized AuNPs can be functionalized with peptides or antibodies for targeted imaging. DNA‑templated Ag nanoclusters (DNA‑Ag NCs) offer tunable emission but suffer from negative surface charge; complexation with cationic polyelectrolytes dramatically boosts fluorescence and cellular uptake. Copper nanoclusters (Cu NCs) synthesized via microwave‑assisted polyol methods show high stability and can serve as selective sensors for RDX.
Silica Nanoparticles
Silica cores protect encapsulated fluorophores from quenching and enable high photostability. The Stöber method and reverse microemulsion produce monodisperse silica NPs, which can be doped with dyes or quantum dots. Dye‑doped silica NPs exhibit up to 12× brighter emission compared to free dyes, while QD‑embedded silica assemblies enhance biocompatibility and imaging contrast.
Phosphors
Y₂O₃‑based phosphors doped with lanthanides (Eu³⁺, Gd³⁺, Tb³⁺) provide high photostability and low background. Hydrothermal, flame‑spray pyrolysis, sol–gel, and co‑precipitation methods enable size control and high QY. Upconversion phosphors (e.g., Er³⁺:Y₂O₃, Ho³⁺/Yb³⁺:GdVO₄) convert NIR excitation to visible emission, offering deep‑tissue imaging with minimal photodamage.
Organic Frameworks
Covalent‑organic frameworks (COFs) and metal‑organic frameworks (MOFs) present high porosity, tunable photoluminescence, and low toxicity. Functional MOFs (e.g., Eu‑pyromellitic acid) exhibit strong, excitation‑dependent emission, enabling selective detection of doxycycline. DNA‑aptamer‑functionalized Zr‑MOFs provide target‑induced bioimaging with high specificity.
Biomedical Applications of Fluorescent Nanomaterials
Bioimaging
Quantum Dots
QDs offer high QY, spectral tunability, and photostability, enabling sub‑cellular resolution imaging. Surface functionalization with thiols or polymers enhances water solubility and biocompatibility. Core–shell engineering eliminates defect emission, producing bright, narrowband QDs suitable for in‑vivo imaging, as demonstrated by AIGSe@GaSₓ QDs in mouse models.
Carbon Dots
Red‑emissive CDs overcome short‑wavelength excitation limitations, enabling deep‑tissue imaging of metal ions in cells and zebrafish. NIR‑emissive CDs, achieved by sulfoxide/carbonyl surface modification, provide high‑contrast imaging with minimal photodamage.
Carbon Nanoparticles
Size‑tunable carbon nanoparticles fabricated via laser ablation or hydrothermal methods emit green or blue fluorescence without cytotoxicity, enabling safe in‑vitro and in‑vivo imaging.
Carbon Nanotubes
Functionalized SWCNTs emit in the 1160 nm NIR window, achieving high signal‑to‑noise imaging in live brain tissue. CNTs conjugated with perylene bisimide fluorophores or NIR dyes allow real‑time tumor imaging during surgery.
Graphene‑Based Nanomaterials
GO functionalized with fluorescent probes (e.g., RACD) exhibits monodispersity, hydrophilicity, and photostability, while intrinsically luminescent graphene derivatives provide tunable NIR emission for deep‑tissue imaging.
Metal Nanomaterials
AuNPs functionalized with bifunctional peptides enable nucleus‑targeted imaging with high photostability. DNA‑Ag NCs modified with cationic polyelectrolytes show 3‑fold fluorescence enhancement and improved cellular uptake, suitable for real‑time cellular imaging.
Silica Nanoparticles
Dye‑doped silica NPs protect fluorophores from quenching and exhibit 12× higher brightness than free dyes. QD‑embedded silica NPs combine excellent optical properties with enhanced biocompatibility, achieving superior in‑vivo imaging contrast.
Phosphors
Y₂O₃:Eu³⁺/Gd³⁺ phosphors (~65 nm) provide high PL and low toxicity, enabling safe bioimaging. Upconversion phosphors (Er³⁺:Y₂O₃, Ho³⁺/Yb³⁺:GdVO₄) deliver NIR‑to‑visible emission for deep‑tissue imaging with minimal background.
Organic Frameworks
MOF‑based probes emit across 614–1350 nm, offering broad NIR coverage for in‑vivo imaging. DNA‑aptamer‑conjugated Zr‑MOFs enable target‑induced fluorescence activation, providing high specificity in tumor imaging.
Biodetection
Fluorescent nanomaterials amplify detection signals, shortening assay times. QDs, CDs, and MOFs have been employed for pathogen, nucleic acid, and drug detection with limits of detection in the picomolar to nanomolar range. Techniques such as strand‑exchange amplification (SEA) coupled with lateral flow assays (LFA) enable visual, rapid nucleic acid detection suitable for low‑resource settings.
Drug Delivery
Fluorescent nanoparticles serve dual roles as carriers and real‑time trackers. pH‑responsive CD‑based systems release doxorubicin in acidic tumor microenvironments while remaining stable at physiological pH. Carbon nanotube carriers achieve high drug loading via π–π interactions, releasing therapeutics upon environmental triggers. Fluorescent labeling allows monitoring of drug distribution and release kinetics in vivo.
Photodynamic Therapy
FNMs can serve as photosensitizers or delivery vehicles for PDT agents. QDs enhance ROS generation under NIR irradiation, improving therapeutic efficacy while minimizing photobleaching. Nanoparticle carriers stabilize hydrophobic photosensitizers and preserve singlet‑oxygen production, broadening PDT applicability.
Challenges
Synthesis Challenges
Achieving Uniform Distribution
Aggregation of nanoscale particles hampers optical performance. Surface functionalization—e.g., silanization of QDs or surfactant coating of CDs—stabilizes colloids and ensures uniform size distribution. Advances in hydrothermal and microwave methods now enable narrow size dispersity (<10 nm).
Fluorescence Quantum Yield
High QY is essential for sensitive imaging. Nitrogen or sulfur co‑doping, high‑temperature microwave synthesis, and surface passivation (e.g., b‑PEI) can raise QY up to 85 %. Continued optimization of dopant concentration and synthesis conditions is needed.
Aggregation‑Caused Quenching
ACQ limits brightness in concentrated or solid states. Strategies such as aggregation‑induced emission (AIE) design, small‑molecule ionic isolation lattices (SMILES), and surface encapsulation (e.g., silica shells) mitigate quenching and preserve fluorescence.
Application Challenges
Drawbacks of UV Light FNMs
Emission in the UV/visible range restricts deep‑tissue imaging and can damage cells. NIR‑emissive FNMs are required for clinical translation, but necessitate specialized detectors and filters.
Interference in Biological Environment
Autofluorescence, pH, and temperature variations in tissues reduce signal‑to‑background ratios. FNMs must maintain high QY at 37 °C and physiological pH, and exhibit pH‑responsive fluorescence for tumor targeting.
Biocompatibility
Heavy‑metal QDs pose toxicity risks; core–shell engineering and the use of carbon or silica matrices reduce adverse effects. Green synthesis (e.g., cacao‑derived gold NPs) and selection of biocompatible metals (Fe, Ti, Zr) in MOFs further enhance safety profiles.
Conclusions
Fluorescent nanomaterials have transcended the limitations of conventional dyes, offering high brightness, tunable spectra, and multifunctionality. Advances in synthesis—chemical, physical, and green routes—yield FNMs with optimized morphology, size, and surface chemistry, propelling their adoption in bioimaging, diagnostics, drug delivery, and photodynamic therapy. Addressing synthesis uniformity, QY, quenching, and biocompatibility will accelerate the transition from laboratory to clinic, unlocking new diagnostic and therapeutic possibilities.
Nanomaterials
- Nanoparticle-Based Cancer Therapy: Advances, Mechanisms, and Clinical Translation
- TiO₂ Nanofluids: Applications, Challenges, and Future Directions – Part 2
- Titania Nanotube Arrays via Electrochemical Anodization: Synthesis, Modifications, and Biomedical Applications
- Biomedical Applications of Gold Nanoclusters: Recent Advances and Future Directions
- Neoglycoprotein‑Functionalized Fluorescent Gold Nanoclusters for Plant Lectin Detection and Dendritic Cell Imaging
- Magnetic Core‑Shell Nanoparticles: Advancing Targeted Drug Delivery, Imaging, and Antimicrobial Therapies
- Advances in Carbon Nanotube Assembly and Integration for Next‑Generation Applications
- Ultrasmall Superparamagnetic Iron Oxide Nanoparticles for Stem Cell Tracking in Parkinson’s Therapy
- Mastering 2D Semiconductor Nanomaterials: Precision Synthesis & Breakthrough Applications
- Unlocking Blockchain’s Potential in Industrial Automation: Opportunities and Obstacles



