Neoglycoprotein‑Functionalized Fluorescent Gold Nanoclusters for Plant Lectin Detection and Dendritic Cell Imaging
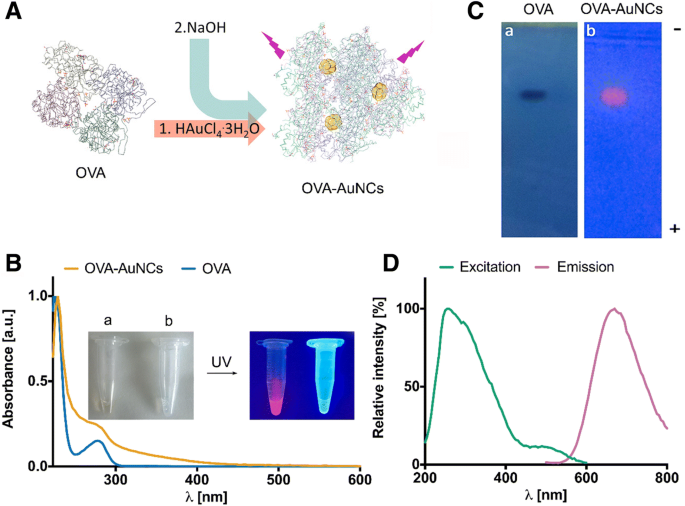
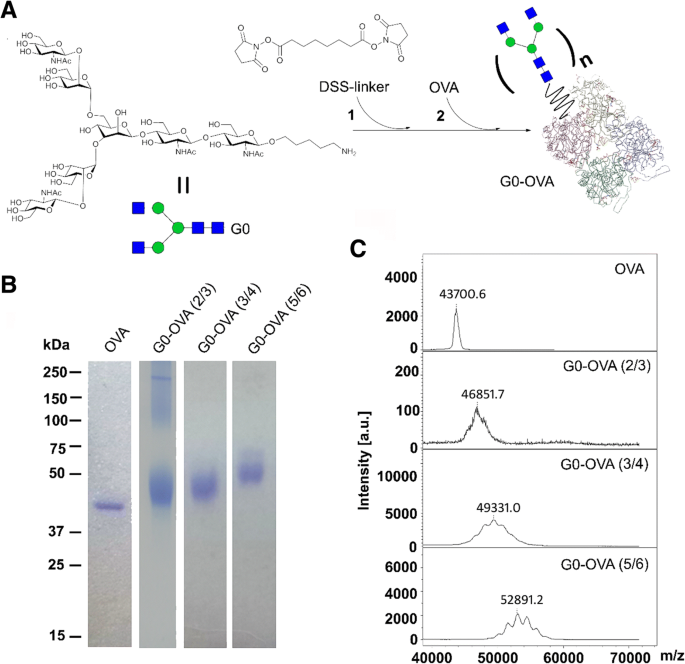
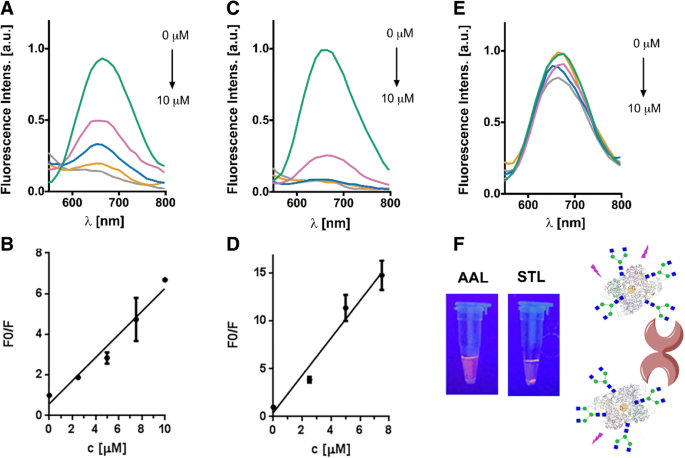
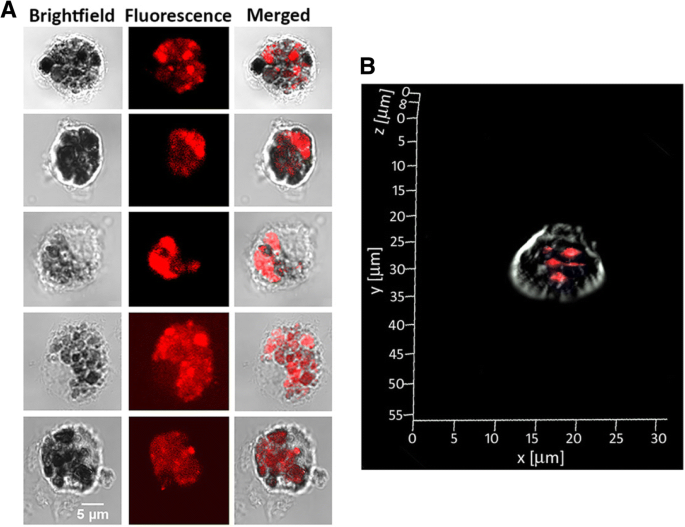
Carbohydrate–protein interactions underpin essential biological processes such as fertilization, signal transduction, and host–pathogen communication. The intricate nature of glycan recognition demands advanced analytical tools. Here we report the first synthesis of neoglycoprotein‑functionalized fluorescent gold nanoclusters (G0‑OVA‑AuNCs) that combine a biantennary N‑glycan (G0) as targeting ligand, ovalbumin as carrier protein, and a self‑emissive gold core. We demonstrate that G0‑OVA‑AuNCs serve as selective sensors for plant lectins and as potent imaging probes for murine dendritic cells. Gold nanoclusters (AuNCs), comprising 10–100 gold atoms, exhibit size‑dependent fluorescence that makes them attractive for bio‑sensing and imaging. Unlike conventional organic dyes or quantum dots, AuNCs possess a large Stokes shift, red‑to‑near‑IR emission, and superior photochemical stability, enabling applications in the tissue transparency window. Protein‑templated AuNCs were first reported with bovine serum albumin in 2009 and have since become a versatile platform for designing water‑soluble, biocompatible fluorophores. Glycosylated proteins, however, remain underexplored as nanocluster scaffolds, despite the critical role of glycans in modulating protein folding, stability, and receptor binding. Synthetic neoglycoproteins—proteins chemically conjugated with defined carbohydrates—offer a strategy to bestow proteins with new functional properties while preserving their structural integrity. In this study we harnessed a synthetic biantennary G0 glycan, conjugated to ovalbumin (OVA) via a C5 amino linker and disuccinimidyl suberate, to generate G0‑OVA‑AuNCs. We investigated their physicochemical characteristics, assessed glycan accessibility through lectin agglutination assays, and evaluated their capacity to label dendritic cells (DCs) in vitro. The results establish G0‑OVA‑AuNCs as robust, multivalent lectin sensors and potent imaging agents for immune cells. We optimized the synthesis of G0‑OVA‑AuNCs by adapting a protocol for unconjugated OVA‑AuNCs. A 15 mg mL⁻¹ OVA solution was mixed with HAuCl₄·3H₂O (4.2 mM) and NaOH (150 mM) under microwave irradiation (100 °C, 6 min), yielding pale brown clusters that emit bright red fluorescence at 670 nm upon 350 nm excitation. UV‑Vis spectra lack a plasmon resonance band, confirming particle sizes below 5 nm; TEM revealed an average core diameter of 1.9 ± 0.7 nm, and dynamic light scattering showed a hydrodynamic diameter of 8.7 ± 2.5 nm, consistent with a single protein encapsulating the core. The clusters exhibited a quantum yield of ~4 % and remained photostable across pH 3–11 and in fetal bovine serum, indicating suitability for biological assays. a Schematic of OVA‑AuNC synthesis. b UV‑visible spectra of OVA (blue) and OVA‑AuNCs (orange). Insert: visible (left) and UV (365 nm, right) images. c Agarose gel of OVA (Coomassie Blue) and OVA‑AuNCs (UV). d Excitation (green) and emission (pink) spectra of OVA‑AuNCs. For G0‑functionalized clusters, G0 glycans were conjugated to OVA using DSS, yielding neoglycoproteins with 2–3, 3–4, or 5–6 glycans per protein (verified by SDS‑PAGE and MALDI‑TOF). Subsequent AuNC formation under identical conditions produced G0‑OVA‑AuNCs whose optical properties matched those of OVA‑AuNCs: absorbance at 278 nm and a 670 nm emission peak. TEM confirmed a core diameter of 1.6 ± 0.5 nm. CD spectra indicated a random coil secondary structure, likely due to the alkaline synthesis, yet the glycan moieties remained accessible for lectin binding. a G0‑OVA neoglycoprotein synthesis (n = valency). b SDS‑PAGE of OVA and G0‑OVA variants. c MALDI‑TOF spectra of the same samples. Lectin agglutination assays with Bandeiraea simplicifolia lectin‑II (BSL‑II) and Solanum tuberosum lectin (STL) revealed concentration‑dependent fluorescence quenching for G0‑OVA‑AuNCs (5/6), while a control lectin (Aleuria aurantia, AAL) produced no effect. The quenching followed linear calibration curves, yielding limits of detection of 2.83 µM (BSL‑II) and 2.35 µM (STL). G0‑OVA‑AuNCs bearing fewer glycans (2/3) did not agglutinate, underscoring the necessity of multivalent glycan presentation. Agarose gel electrophoresis in cell‑culture media confirmed that lectin binding remained efficient in complex biological environments. Lectin agglutination of G0‑OVA‑AuNCs (5/6). a Fluorescence spectra after BSL‑II incubation. b F₀/F vs BSL‑II concentration. c Spectra after STL incubation. d F₀/F vs STL concentration. e Spectra after AAL incubation. f UV images of samples; precipitation observed with STL. Confocal microscopy of murine splenic DCs (CD11c⁺) incubated with G0‑OVA‑AuNCs (3/4) demonstrated rapid internalization within 40 min, with fluorescence localized intracellularly and absent in untreated controls. 3‑D reconstructions confirmed internal cluster distribution. DC uptake of G0‑OVA‑AuNCs (3/4). a Representative images of labeled cells. b Z‑stack illustrating intracellular fluorescence. In summary, we present the first neoglycoprotein‑protected AuNCs, demonstrate their selective lectin recognition via multivalent glycans, and establish their capacity to image dendritic cells. Their robust fluorescence, aqueous solubility, and pH stability make them promising alternatives to conventional dyes for carbohydrate‑protein interaction studies and immunological imaging. We successfully synthesized and characterized G0‑OVA‑AuNCs, confirming glycan accessibility and lectin‑specific agglutination. The multivalent glycan display was essential for efficient cross‑linking, while the clusters maintained high photostability and aqueous solubility. In vitro imaging of murine dendritic cells validated the clusters as potent, self‑fluorescent probes. These findings support further exploration of neoglycoprotein‑functionalized AuNCs as versatile tools for carbohydrate‑protein interaction assays, immune cell imaging, and potential therapeutic applications. All solutions were prepared in nanopure water (18 MΩ cm). Gold (III) chloride trihydrate, NaOH, and triethylamine were from Sigma‑Aldrich. Ovalbumin was purchased from Hyglos. DSS and DMSO were from Thermo Fisher. G0 glycan was synthesized as previously described. 15 mg mL⁻¹ protein in nanopure water was mixed with 4.2 mM HAuCl₄·3H₂O, stirred 5 min, then 150 mM NaOH was added. The mixture was heated at 100 °C for 6 min in a microwave reactor. Clusters were dialyzed (10 kDa MWCO) against nanopure water. Fluorescence was recorded on a Varioskan Flash plate reader (λₑₓ=350 nm). Agarose gel (0.75 %) and TEM (200 kV) were used for size characterization. CD assessed secondary structure. XPS determined gold oxidation states. G0‑OVA‑AuNCs (0.2 mg mL⁻¹) were mixed with lectins (AAL, STL, BSL‑II) at 0–10 µM in TSM buffer. After overnight incubation, samples were centrifuged and supernatant fluorescence measured. Precipitation was observed under UV for STL. Spleens from C57BL/6J mice were processed to single‑cell suspensions. CD11c⁺ cells were isolated by magnetic‑activated cell sorting (Miltenyi) and cultured on poly‑D‑lysine–coated coverslips. DCs (2 × 10⁶ cells) were incubated with 150 µg mL⁻¹ G0‑OVA‑AuNCs (3/4) for 40 min at 37 °C, washed, fixed with 3 % PFA, and imaged on a Zeiss LSM 510 confocal microscope (365 nm laser, 63× oil objective). Aleuria aurantia lectin Gold nanoclusters Bovine serum albumin Bandeiraea simplicifolia lectin‑II Circular dichroism C‑type lectin receptors Dendritic cells Dimethyl sulfoxide Disuccinimidyl suberate ester Fetal bovine serum Biantennary N‑glycan Neoglycoprotein‑functionalized gold nanoclusters Gold tetrachloroauric (III) acid Iscove’s modified Dulbecco’s medium Infrared Limit of detection Magnetic‑activated cell separation Ovalbumin Quantum yield Solanum tuberosum lectin Transmission electron microscopy X‑ray photoelectron spectroscopyAbstract
Introduction
Results and Discussion
Conclusions
Methods/Experimental
Materials
AuNCs Synthesis. General Procedure
Lectin Agglutination Assay
Isolation of Mouse Splenic DCs
DC Uptake of G0‑OVA‑AuNCs
Abbreviations
Nanomaterials
- Gold Nanoparticles: Advancing Diagnostic and Therapeutic Applications in Medicine – A Comprehensive Review
- Fluorescent Nanomaterials: Advances, Synthesis, and Biomedical Applications
- High‑Performance Au/Ce‑La Nanorod Catalysts for Low‑Temperature CO Oxidation: Synthesis, Characterization, and Mechanistic Insights
- Seed‑Mediated Synthesis of Tunable‑Aspect‑Ratio Gold Nanorods for Enhanced Near‑Infrared Photoacoustic Imaging
- Biomedical Applications of Gold Nanoclusters: Recent Advances and Future Directions
- Dual‑Mode MRI/Fluorescence Nanoprobe cRGD‑Gd‑Cy5.5 for Targeted Imaging of Integrin αvβ3‑Positive Tumors
- Magnetic Core‑Shell Nanoparticles: Advancing Targeted Drug Delivery, Imaging, and Antimicrobial Therapies
- Efficient One-Step Green Synthesis of Multifunctional Gold Nanoparticles for Targeted Tumor Imaging and Therapy
- Ultrasmall Superparamagnetic Iron Oxide Nanoparticles for Stem Cell Tracking in Parkinson’s Therapy
- Mastering 2D Semiconductor Nanomaterials: Precision Synthesis & Breakthrough Applications



