Enhanced Gas Separation Using Covalently Functionalized Graphene Oxide in PIM‑1 Mixed‑Matrix Thin‑Film Composite Membranes
Abstract
We report the fabrication of mixed‑matrix membranes (MMMs) comprising covalently functionalized graphene oxide (FGO) and pristine graphene oxide (GO) dispersed in a polymer of intrinsic microporosity (PIM‑1). The membranes were prepared by a dip‑coating protocol, and single‑gas permeation performance was quantified. FGO was characterized by FTIR, UV‑Vis, Raman, SEM, and TGA, confirming successful grafting of amine, oxime, ferrocenyl, and phosphochloride groups. Systematic variation of FGO loading (9–84 wt %) revealed distinct effects on membrane morphology and gas transport, highlighting the role of functional group chemistry on filler‑polymer interactions, filler stiffness, and alignment relative to the membrane surface.
Introduction
Graphene’s two‑dimensional lattice and exceptional barrier properties make it a promising additive for membrane‑based separations. Even trace amounts (<0.0075 wt %) of graphene nanoplatelets in PIM‑1 reduce gas permeability by a factor of three [1, 2], while 0.4 wt % graphene in PET lowers O₂ permeability fourfold [3]. Functionalized GO has shown >99.9 % salt rejection in PVDF membranes [4] and superior organic solvent nanofiltration when incorporated into PBI [5]. In thermally rearranged polymers, reduced GO increased CO₂ permeance by 482 × while maintaining a CO₂/N₂ selectivity of 35 [6].
Most thin‑film composite (TFC) studies use <0.1 wt % graphene, exploiting the low percolation threshold of exfoliated flakes [7, 8]. However, achieving monolayer graphene remains energy intensive, and inter‑sheet van der Waals forces promote aggregation [9]. Chemical grafting of functional groups can mitigate this issue by disrupting π‑π stacking and enhancing dispersion [10]. GO, enriched with epoxide, hydroxyl, and carboxyl groups, is readily oxidized and functionalized, improving its solubility and reactivity [11, 12].
In the present work, we covalently grafted amine, oxime, ferrocenyl, and phosphochloride moieties onto GO, yielding GO‑AEDPPF, GO‑DClBAO, GO‑DMPPA, and PhChGO, respectively. We incorporated these FGOs into PIM‑1 at loadings up to 84 wt % and evaluated single‑gas permeation for CH₄, N₂, O₂, and CO₂ at 30 °C, 500 mbar feed. Key questions addressed were: (i) the maximum filler content that preserves the solution‑diffusion mechanism; (ii) the feasibility of aligning platelet‑like fillers via dip‑coating; (iii) the influence of filler chemistry on selective‑layer integrity; and (iv) any preferential gas interactions with functional groups.
Methods
Materials
Graphite (99.9995 %), 2,4‑dichlorobenzamidoxime (97 %), THF, NaNO₃ (99.5 %), DMF, Et₃N (99 %), H₂O₂ (30 %), DMPPA (98 %), AEDPPF (97 %), dClpf (≥ 96 %), KMnO₄ (99 %), H₂SO₄ (95–98 %), SOCl₂ (99 %), PCl₃ (99 %), NMP (> 99 %), and benzene (for synthesis) were obtained from Alfa Aesar, Merck, Sigma‑Aldrich, and AppliChem. All reagents were used as received.
Synthesis of Graphene Oxide
GO was produced from natural graphite via the Hummers method [15]. Briefly, 2 g graphite, 1 g NaNO₃, and 46 mL H₂SO₄ were cooled to 0 °C; 6 mg KMnO₄ was added slowly to keep the temperature below 20 °C, then stirred for 2 h. The mixture was warmed to 35 °C for 1 h, then 92 mL water was added, raising the temperature to 98 °C. After the solution turned bright yellow, it was diluted to 250 mL water and 20 mL H₂O₂ was added to quench residual permanganate. The suspension was vacuum‑filtered, washed, centrifuged, and freeze‑dried (Gamma 1‑16 LSC plus).
Synthesis of Functionalized Graphene Oxide (FGO)
Chlorinated Graphene Oxide (GO‑Cl)
GO‑Cl was prepared by refluxing 0.5 g GO with 10 mL benzene and 50 mL SOCl₂ at 70 °C for 24 h. Excess SOCl₂ was removed by vacuum distillation, and the residue was dispersed in acetone, filtered, washed, and vacuum‑dried at 60 °C.
GO‑AEDPPF
GO‑Cl (0.2 g) was reacted with 0.02 g AEDPPF and 3 mL Et₃N in 60 mL DMF at 130 °C for 3 days. The product was cooled, filtered, washed with DMF, water, and acetone, and dried at 60 °C.
GO‑DClBAO
0.2 g GO‑Cl was dispersed in 50 mL NMP; 0.02 g DClBAO and 3 mL trimethylamine were added, then heated at 157 °C for 72 h. The mixture was filtered, washed with NMP and water, and dried at 40 °C.
Synthesis of Polymer of Intrinsic Microporosity (PIM‑1)
PIM‑1 was synthesized following literature protocols [19–23], dried under vacuum at 70 °C for 48 h. SEC yielded a weight‑average molecular weight of 200 kg mol⁻¹ and a polydispersity of 4–5.
Thin‑Film Composite Membrane Preparation
PIM‑1 solutions (1 wt % in THF) were mixed with GO, GO‑AEDPPF, or GO‑DClBAO at 9, 33, 50, 76, and 84 wt % (relative to dry polymer). All mixtures were sonicated for 1 h. The selective layer was deposited onto a 22 nm pore PAN support by a modified dip‑coating method: the support was immersed, then withdrawn at 1.56 mm min⁻¹, allowing a meniscus to form. The solvent evaporated naturally; the membrane was then left to dry at ambient conditions.
Characterizations
FTIR (Bruker ALPHA, ATR, 400–4000 cm⁻¹, 4 cm⁻¹, 64 scans), UV‑Vis (Specord 210 Plus, 190–1100 nm), Raman (Senterra, 532 nm laser, 10× objective), TGA (Netzsch TG209 F1, Ar flow, 25–900 °C, 10 K min⁻¹), SEM (Merlin, Zeiss, 2–10 kV, 6 nm carbon coating) with EDX, GPC (Waters, THF), and elemental analysis (EuroEA CHNSO) were performed. Gas permeation was measured at 30 °C, 500 mbar feed, and 10 mbar permeate for CH₄, N₂, O₂, and CO₂ using the standard permeance equation (Eq. 1) and ideal selectivity (Eq. 2). Each membrane was tested at least four times, with an average of 10 data points per gas.
Results and Discussion
Synthesis and Characterization of GO and FGO
The Hummers‑derived GO exhibited typical oxygen functional groups: –OH, epoxide, and –COOH (Fig. 3). Covalent grafting of amine and oxime groups (GO‑DMPPA, GO‑AEDPPF, GO‑DClBAO) was confirmed by new FTIR bands at 1500–1650 cm⁻¹ (N–H bending) and 1240 cm⁻¹ (C–N stretching). GO‑Cl displayed characteristic C–Cl and C=O (≈1800 cm⁻¹) bands, while GO‑dClpf and PhChGO showed P–Cl and P=O vibrations. Elemental analysis (Table 1) revealed C/O ratios increasing from 1.42 (GO) to 5.61 (GO‑DMPPA) and 5.12 (GO‑AEDPPF), indicating successful functionalization and reduced oxygen content.
FTIR spectra of GO and its functionalized counterparts.
Raman spectra displayed the D (~1350 cm⁻¹) and G (~1580 cm⁻¹) bands with I_D/I_G ≈ 1 for all samples, confirming multilayer GO sheets. TGA showed that amine/imine grafting improved thermal stability, with major weight loss (≈35 %) between 200–500 °C, whereas GO‑Cl, GO‑dClpf, and PhChGO exhibited additional loss at 100–360 °C due to chlorinated or phosphorous groups (Fig. S3).
TFC Membrane Morphology
Surface Morphology
SEM images (Fig. S6) show that pure PIM‑1 surfaces are smooth, whereas membranes containing GO, GO‑AEDPPF, or GO‑DClBAO display increasing roughness with filler loading. At 9 wt % GO, flakes are well dispersed; at 50–84 wt % GO, agglomerates appear, indicating a limit for homogeneous dispersion.
Cross‑Sectional Morphology
Cross‑section images (Fig. S7) reveal a ~200 nm selective layer on the PAN support. GO particles align parallel to the membrane surface at low loadings (9 wt %) due to shear during withdrawal, but at >50 wt % significant voids and agglomerates form, compromising layer integrity.
Gas Transport Performance
Permeance data (Fig. 5) demonstrate a pronounced decrease in CH₄, N₂, O₂, and CO₂ permeance with increasing GO loading, particularly at 9 wt %. The permeance drop is most severe for CO₂, consistent with its higher diffusivity. At >50 wt % GO, the selective layer ruptures, leading to a permeance rise that approaches the Knudsen limit. In contrast, GO‑AEDPPF and GO‑DClBAO exhibit lower permeance at 33 wt % but maintain selectivity above the pure PIM‑1 membrane, indicating improved filler‑polymer compatibility. Above 50 wt %, permeance rises sharply for these FGOs, suggesting that bulky functional groups hinder efficient alignment and create non‑selective pathways.
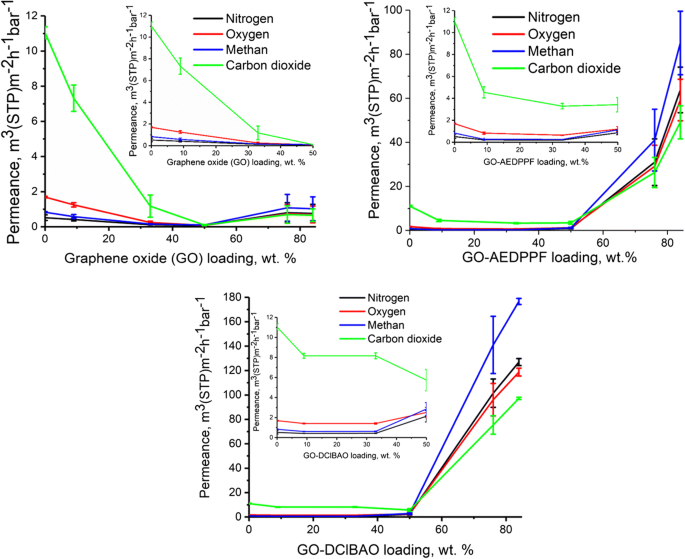
Gas permeance as a function of GO and FGO loading in PIM‑1.
CO₂/N₂ selectivity trends (Fig. 6) show that GO‑AEDPPF and GO‑DClBAO retain higher selectivity at low loadings (≤33 wt %) but drop once the selective layer is compromised. The data corroborate the hypothesis that functionalization enhances interfacial adhesion and mitigates agglomeration up to a critical filler content.
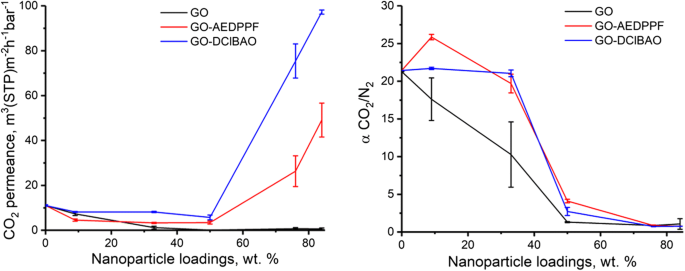
Impact of different FGO types on permeance and CO₂/N₂ selectivity.
Conclusions
We successfully grafted amine, oxime, ferrocenyl, and phosphochloride groups onto GO, producing FGOs with enhanced thermal stability and altered interlayer spacing. When incorporated into PIM‑1 MMMs, pristine GO served as an effective barrier, reducing permeance and selectivity with increasing loading. GO‑AEDPPF and GO‑DClBAO maintained high CO₂ and O₂/N₂ selectivities at 9–33 wt % loading, outperforming pure PIM‑1, but above 50 wt % the selective layer degraded and permeance increased. These findings demonstrate that functional group chemistry can tune filler–polymer interactions, control filler alignment, and define the upper filler loading limit for robust TFC membranes. Future work should probe the mechanical rigidity of functionalized GO sheets (e.g., via nano‑indentation) to correlate filler stiffness with membrane performance.
Abbreviations
- AEDPPF
(Rp)-1-[(1S)-(1‑Aminoethyl)]‑2‑(diphenylphosphino) ferrocene
- DClBAO
2,4‑Dichlorobenzamidoxime
- dClpf
1,1‑Bisdichlorophosphino‑ferrocene
- DMPPA
2,5‑Dimethyl‑6‑phenylpyrazolo(1a)-pyrimidin‑7‑amine
- FGO
Functionalized graphene oxide
- FTIR
Fourier transform infrared spectroscopy
- GO
Graphene oxide
- GO-AEDPPF
(Rp)-1-[(1S)-(1‑aminoethyl)]‑2‑(diphenylphosphino)ferrocene‑modified GO
- GO-Cl
Chlorinated graphene oxide
- GO-DClBAO
2,4‑Dichlorobenzamidoxime‑modified GO
- GO-dClpf
1,1‑Bisdichlorophosphino‑ferrocene‑modified GO
- GO-DMPPA
2,5‑Dimethyl‑6‑phenylpyrayolo(1,5‑a)-pyrimidin‑7‑amine‑modified GO
- GPC
Gel permeation chromatography
- PhChGO
Phosphochlorinated GO
- SEM
Scanning electron microscopy
- TFC
Thin‑film composite
- TGA
Thermal gravimetric analysis
Nanomaterials
- Graphene‑Polymer Composites for High‑Performance Supercapacitors: A Comprehensive Review
- Synergistic Graphene-Modified TiO₂ Composites Deliver Superior Photocatalytic Activity
- Titanate Nanotube-Enhanced Graphene Oxide Nanocomposites: Boosting Flame Retardancy and Photodegradation in Flexible PVC
- Assessing the Biosafety and Antibacterial Efficacy of Graphene and Graphene Oxide for Orthopedic Implant Applications
- High‑Performance Au/Ce‑La Nanorod Catalysts for Low‑Temperature CO Oxidation: Synthesis, Characterization, and Mechanistic Insights
- Hybrid Graphene/WO₃ and Graphene/CeOx Electrodes for High‑Performance Supercapacitors
- Spherical Graphene/SiO₂ Supports Enable Uniform In Situ Polymerization of UHMWPE for High‑Performance Nanocomposites
- Oxidant‑Free Hydrothermal Anchoring of Ultrathin PEDOT on Reduced Graphene Oxide for High‑Performance Flexible Supercapacitors
- Advanced Li‑Sulfur Batteries: Polycarboxylate‑Functionalized Graphene Cathodes and PDAAQ‑Coated Separators Deliver 1100 mAh g⁻¹
- Reducing Cytotoxicity of Silver Nanoparticle Coatings with Graphene Oxide: Enhanced Antibacterial Performance and Biocompatibility of an Ag–GO Nanocomposite



