Advanced Li‑Sulfur Batteries: Polycarboxylate‑Functionalized Graphene Cathodes and PDAAQ‑Coated Separators Deliver 1100 mAh g⁻¹
Abstract
To bridge the gap between the theoretical (1672 mAh g⁻¹) and practical performance of Li‑S batteries, we engineered composite cathodes comprising polycarboxylate‑functionalized graphene (PC‑FGF) doped with TiO₂ nanoparticles or poly‑1,5‑diaminanthraquinone (PDAAQ) and sulfur. PC‑FGF/S acts as an efficient polysulfide trap while maintaining high electronic and ionic conductivity. Two cathode–separator combinations were tested: TiO₂/PC‑FGF/S with PDAAQ/PC‑FGF‑coated glass‑fiber separators and PDAAQ/PC‑FGF/S with the same separators. The TiO₂/PC‑FGF/S cathode paired with a PDAAQ/PC‑FGF separator achieved 1100 mAh g⁻¹ at 0.5 C after 100 cycles, highlighting superior cycling stability and rate capability for next‑generation Li‑S cells.
Background
Li‑S batteries promise ~2600 Wh kg⁻¹ energy density, but practical performance is limited by the shuttle effect of soluble polysulfides. Strategies to confine polysulfides include chemically active hosts, porous carbon matrices, and functionalized separators. Graphene derivatives, especially those with oxygen or nitrogen functionalities, have shown promise in immobilizing polysulfides and enhancing conductivity. Quinone polymers like PDAAQ offer redox‑active sites and can chemically bind polysulfides, improving utilization and cycling. Metal oxides (TiO₂, MnO₂, NiO, etc.) have been incorporated into graphene or sulfur hosts to trap polysulfides through strong chemisorption.
In this work, we combine TiO₂ nanoparticles or PDAAQ with PC‑FGF to create cathodes that combine physical confinement, chemical binding, and high conductivity. Separators are modified with PDAAQ/PC‑FGF, PDAAQ/CTAB/PC‑FGF, or TiO₂/MWCNT/PC‑FGF to further suppress polysulfide migration.
Methods
Materials and Chemicals
PC‑FGF (Sigma‑Aldrich), CTAB (≥99%, Sigma‑Aldrich), PDAAQ (85%, Sigma‑Aldrich), TiO₂ nanopowder (21 nm, ≥99.5%, Sigma‑Aldrich), MWCNT (>98%, Sigma‑Aldrich), sulfur (99.5–100.5%, Sigma‑Aldrich), LiTFSI (99.95%, Sigma‑Aldrich), LiNO₃ (99.99%, Sigma‑Aldrich), PVDF (Mw 1000–1200 kg mol⁻¹, Solvay), NMP (99%, Sigma‑Aldrich), DOL/DME (1:1), and glass microfiber filters (Whatman GF/C) were used without further purification.
Separator Coating
For PDAAQ/CTAB/PC‑FGF, the components were ground in a 2:2:1 mass ratio, mixed with PVDF (1:4), and dispersed in NMP (1:9, 30 wt % PVDF) to form a homogeneous slurry. The slurry was cast on one side of a glass‑fiber separator and dried at 60 °C for 2 h. PDAAQ/PC‑FGF and TiO₂/MWCNT/PC‑FGF coatings followed the same procedure with respective mass ratios. Coating thicknesses were 53 µm, 57 µm, and 61 µm.
Cathode Preparation
TiO₂/PC‑FGF/S cathodes contained 60 wt % S, 15 wt % PC‑FGF, 15 wt % TiO₂, and 10 wt % PVDF in NMP, cast onto Al foil (16 µm, 1.32 cm²). PDAAQ/PC‑FGF/S cathodes used 60 wt % S, 15 wt % PC‑FGF, 15 wt % PDAAQ, and 10 wt % PVDF. Cells were assembled in an Ar glove box with Li metal anodes and 1 M LiTFSI/0.5 M LiNO₃ in DME/DOL (1:1). Electrolyte loading was 20 µl mg⁻¹ S; S loading ranged 1.8–2.4 mg cm⁻².
Electrochemical Performance Characterization
CR2032 cells were cycled between 1.5 and 3 V at room temperature. Specific capacities were calculated from sulfur mass. SEM with EDS examined cathodes and separators. CV was performed with a 0.2 mV s⁻¹ scan rate (3–1.4 V vs. Li⁺/Li). EIS spanned 1 MHz to 1 Hz (10 mV AC).
Results and Discussion
Figure 1 illustrates the polysulfide migration in TiO₂/PC‑FGF/S and PDAAQ/PC‑FGF/S cells with PDAAQ/PC‑FGF‑coated separators. PDAAQ’s benzoquinone groups form reversible redox pairs that trap polysulfides, while TiO₂ nanoparticles provide strong chemisorption sites.
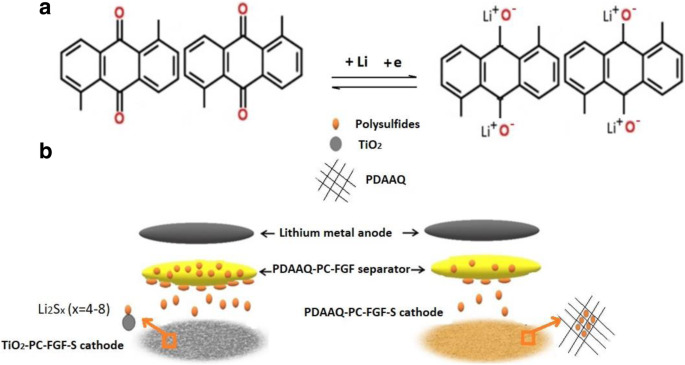
a Benzoquinone redox in PDAAQ. b Polysulfide pathways with two cathode types.
SEM images (Figure 2) confirm uniform TiO₂ dispersion in PC‑FGF/S and homogeneous PDAAQ distribution in PDAAQ/PC‑FGF/S. Separator coatings also show even nanoparticle or polymer dispersion, enhancing polysulfide confinement.

SEM of (a) TiO₂/PC‑FGF/S and (b) PDAAQ/PC‑FGF/S cathodes; (c) TiO₂/MWCNT/PC‑FGF, (d) PDAAQ/PC‑FGF, (e) PDAAQ/CTAB/PC‑FGF‑coated separators.
Cycling performance (Figure 3) shows TiO₂/PC‑FGF/S paired with PDAAQ/PC‑FGF separator achieves 1241 mAh g⁻¹ at 0.5 C and 1100 mAh g⁻¹ after 100 cycles. PDAAQ/PC‑FGF/CTAB separator yields 1040 mAh g⁻¹ (0.5 C) with 730 mAh g⁻¹ retention. PDAAQ/PC‑FGF/TiO₂/MWCNT separator delivers 800 mAh g⁻¹ (0.5 C) and 700 mAh g⁻¹ after 100 cycles, highlighting the role of TiO₂ in trapping polysulfides at the separator.
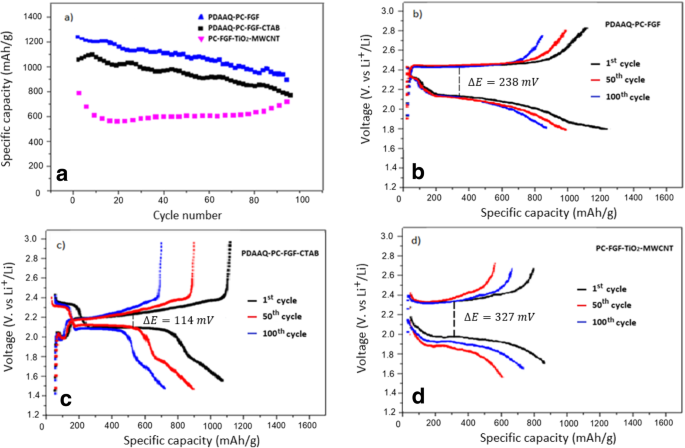
Performance of PDAAQ/PC‑FGF/S with (a) PDAAQ/PC‑FGF, (b) PDAAQ/CTAB/PC‑FGF, and (c) TiO₂/MWCNT/PC‑FGF separators.
TiO₂/PC‑FGF/S with PDAAQ/PC‑FGF separator (Figure 4) shows 1241 mAh g⁻¹ (0.5 C) and 1100 mAh g⁻¹ after 100 cycles. Polarization potentials are 261 mV (PDAAQ/PC‑FGF) and 134 mV (PDAAQ/CTAB/PC‑FGF), indicating lower kinetic barriers when PDAAQ is present. TiO₂/MWCNT/PC‑FGF separator shows higher resistance, with capacity dropping from 1011 to 697 mAh g⁻¹ (0.5 C) after 100 cycles.
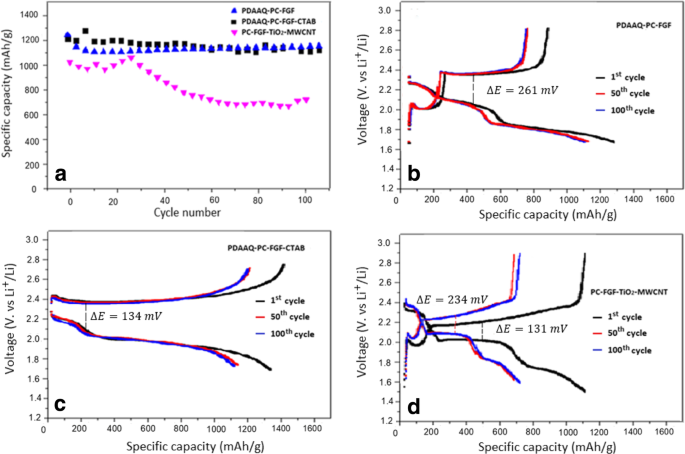
Performance of TiO₂/PC‑FGF/S with (a) PDAAQ/PC‑FGF, (b) PDAAQ/CTAB/PC‑FGF, and (c) TiO₂/MWCNT/PC‑FGF separators.
Rate capability tests (Figure 5) confirm that PDAAQ/PC‑FGF/S delivers 1367 mAh g⁻¹ at 0.2 C, retaining 1358 mAh g⁻¹ at 0.5 C and 1298 mAh g⁻¹ at 1 C. PDAAQ/CTAB/PC‑FGF shows slightly lower values but remains above 1200 mAh g⁻¹ at 0.5 C. TiO₂/PC‑FGF/S with TiO₂/MWCNT/PC‑FGF separator experiences rapid capacity loss, indicating the importance of separator design.
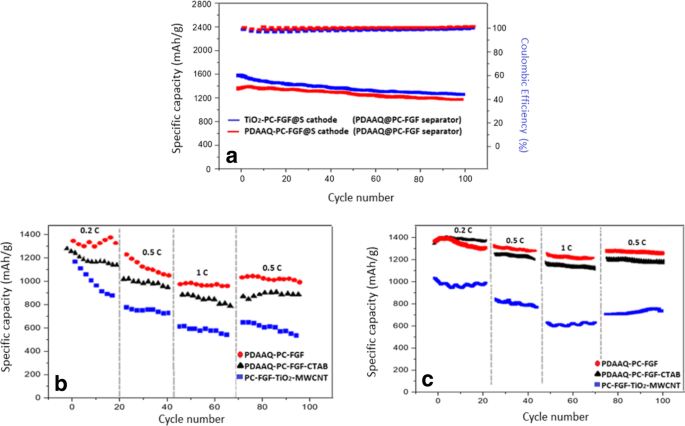
Rate performance of (a) PDAAQ/PC‑FGF/S with three separators; (b) TiO₂/PC‑FGF/S with the same separators.
CV curves (Figure 6) show cathodic peaks at 2.20/1.80 V and 2.19/1.83 V for TiO₂/PC‑FGF/S and PDAAQ/PC‑FGF/S, respectively, corresponding to long‑chain polysulfide reduction. Anodic peaks at 2.30/2.36 V indicate oxidation of short‑chain polysulfides. PDAAQ/CTAB/PC‑FGF separator shifts peaks to higher potentials, reducing polarization.
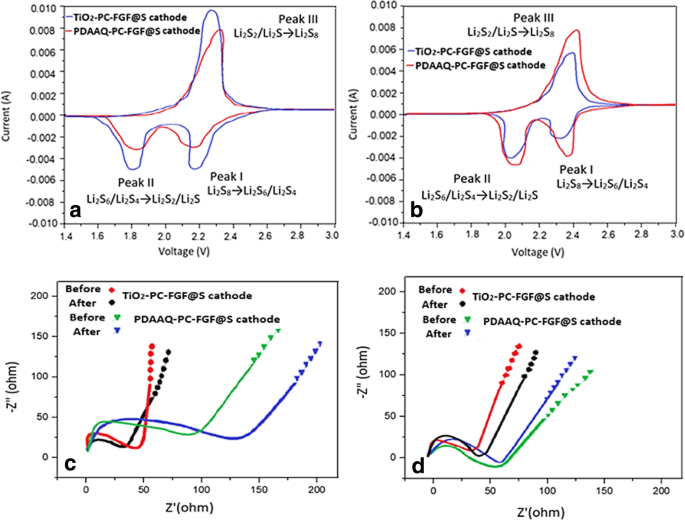
CV (a) PDAAQ/PC‑FGF separator, (b) PDAAQ/CTAB/PC‑FGF separator; (c) EIS for PDAAQ/PC‑FGF separator; (d) EIS for PDAAQ/CTAB/PC‑FGF separator.
EIS results (Figure 6c,d) reveal lower charge‑transfer resistance for TiO₂/PC‑FGF/S, confirming faster ion transport and better interfacial stability. The TiO₂/MWCNT/PC‑FGF separator exhibits higher resistance due to slower electrolyte infiltration.
After 100 cycles, SEM and EDS mapping (Figure 7) confirm that PDAAQ/PC‑FGF‑coated separators accumulate sulfur uniformly, effectively trapping polysulfides and mitigating the shuttle effect. PDAAQ/CTAB/PC‑FGF shows similar but slightly lower sulfur loading, while TiO₂/MWCNT/PC‑FGF retains minimal sulfur, explaining its poorer performance.

SEM (a‑c) and EDS sulfur maps (d‑f) of separators after 100 cycles with TiO₂/PC‑FGF/S cathode.
Conclusions
TiO₂/PC‑FGF and PDAAQ/PC‑FGF composite cathodes, prepared via a single‑step slurry method, deliver high capacity (≈1100 mAh g⁻¹) and excellent cycling (≥100 cycles) when paired with PDAAQ‑coated separators. The synergistic effect of TiO₂’s chemisorption, PC‑FGF’s conductivity, and PDAAQ’s redox activity minimizes polysulfide shuttle and reduces interfacial resistance. Future Li‑S research should focus not only on cathode design but also on tailored separator coatings to further enhance active‑material reutilization and long‑term stability.
Availability of Data and Materials
All data used within this manuscript are available upon request.
Abbreviations
- CTAB:
Cetyltrimethylammonium bromide
- CV:
Cyclic voltammogram
- DME:
1,2‑dimethoxyethane
- DOL:
1,3‑dioxolane
- LiNO3:
Lithium nitrite
- LiTFSI:
Lithium bis(trifluoromethane) sulfonamide lithium
- NMP:
N‑methyl‑2‑pyrrolidone
- PC‑FGF:
Polycarboxylate functionalized graphene
- PDAAQ:
1,5‑poly‑diaminanthraquinone
- PVDF:
Polyvinylidene fluoride
- SEM:
Scanning electron microscopy
- TiO2:
Titanium dioxide
Nanomaterials
- Graphene‑Polymer Composites for High‑Performance Supercapacitors: A Comprehensive Review
- LiNi0.5Mn1.5O4 Cathodes with Cr3+ and F− Composite Doping: Synthesis, Stability, and Electrochemical Performance
- Solid‑State Synthesized PEDOT/Au/GO Composite for Ultra‑Sensitive Electrochemical Detection of Nitrite and Iodate
- Hybrid Graphene/WO₃ and Graphene/CeOx Electrodes for High‑Performance Supercapacitors
- High‑Capacity Si/Graphene Composite Anode Fabricated via Magnesium‑Thermal Reduction for Li‑Ion Batteries
- Polyaniline‑Coated Activated Carbon Aerogel/Sulfur Composite Yields Superior Lithium‑Sulfur Battery Performance
- CuGeO3 Nanowires: A High‑Capacity, Stable Anode for Advanced Sodium‑Ion Batteries
- Enhanced Gas Separation Using Covalently Functionalized Graphene Oxide in PIM‑1 Mixed‑Matrix Thin‑Film Composite Membranes
- FeF3·0.33H2O Cathode Enhanced by CNTs and Graphene: A High‑Performance Solution for Lithium‑Ion Batteries
- Revolutionary Thermoelectric Cooling System Enhances Performance of Computers & Batteries



