Cubic Ni(OH)₂ Nanocage Synthesis via Coordinated Etching–Precipitation for Superior Supercapacitor Performance
Abstract
Transition‑metal hydroxides can reach their full electrochemical potential when engineered into cage‑like architectures. Here, we report a one‑pot Coordinated Etching–Precipitation (CEP) strategy that transforms cubic Cu₂O templates into hollow Ni(OH)₂ nanocages (NCs). The resulting NCs exhibit abundant active sites, high porosity, and a thin, uniform shell that facilitates rapid electron and ion transport. As a positive electrode, Ni(OH)₂ NCs/Ni‑foam deliver a remarkable 539.8 F g⁻¹ at 1 A g⁻¹ and retain 96.9 % of their capacitance after 2 000 cycles. In an asymmetric supercapacitor (ASC) paired with activated carbon, the device reaches 23.3 Wh kg⁻¹ at 800 W kg⁻¹, outperforming conventional broken‑nanocage counterparts by an order of magnitude. These findings underscore the importance of cage‑like nanostructures for next‑generation energy‑storage materials.
Background
Environmental sustainability demands high‑performance, low‑carbon energy storage. Supercapacitors, with their high power density, rapid charge–discharge, and benign chemistry, satisfy these criteria. Pseudocapacitive materials based on transition‑metal hydroxides, especially Ni(OH)₂, are attractive due to the reversible Ni³⁺/Ni²⁺ redox couple in alkaline media. However, bulk Ni(OH)₂ suffers from limited active surface area and sluggish ion diffusion, leading to specific capacitances far below theoretical limits.
Engineering the micro‑ and nanoscale morphology of Ni(OH)₂ offers a pathway to overcome these limitations. Hollow, cage‑like structures maximize surface area, expose inner and outer facets, and create straight ion channels, thereby accelerating charge transfer and enhancing cycling stability. Coordinated etching and precipitation—where a sacrificial template is simultaneously dissolved and the target material is precipitated—has emerged as a versatile route to such architectures.
Methods/Experimental
Cu₂O Template Synthesis
Cu₂O cubes (∼500 nm edge) were prepared by a one‑step aqueous reaction: CuCl₂·2H₂O (0.01 M) was stirred in 2 M NaOH at 55 °C, followed by dropwise addition of 0.6 M ascorbic acid. The precipitate was centrifuged, washed, and dried under vacuum.
Ni(OH)₂ NC Fabrication
In a 1:1 water–ethanol mixture, Cu₂O templates (400 mg) were combined with NiCl₂·6H₂O (80–320 mg, controlling the Cu₂O:NiCl₂ ratio). After 10 min sonication and addition of 13.2 mg PVP, 160 mL of 1 M Na₂S₂O₃ was introduced dropwise. The mixture was stirred for 3 h at room temperature, then centrifuged and dried. The broken‑nanocage control (BNC) was produced by sonication of the NCs in alcohol for 2 h.
Characterization
XRD (Rigaku D/Max‑2400), XPS (ESCALAB 250Xi), FESEM (Zeiss Gemini 300), TEM (FEI F20), and BET surface area (Belsort‑max) were employed to confirm phase purity, morphology, and porosity.
Electrochemical Testing
All measurements were conducted in 3 M KOH using a three‑electrode cell (Pt counter, Ag/AgCl reference). The working electrode consisted of 80 % Ni(OH)₂, 15 % acetylene black, and 5 % PTFE on Ni‑foam (1 cm²) with a loading of 3.4 mg cm⁻². Cyclic voltammetry (CV), galvanostatic charge–discharge (GCD), and electrochemical impedance spectroscopy (EIS, 0.01–100 kHz) were performed on an Autolab μIII system. ASC devices were assembled with Ni(OH)₂ NCs/NF as the positive electrode and activated carbon (AC)/NF as the negative electrode, separated by a cellulose paper and immersed in 3 M KOH.
Results and Discussions
Structural Analysis
XRD patterns confirm hexagonal β‑Ni(OH)₂ (JCPDS 14‑0117). XPS shows Ni²⁺ signals at 856.1 eV (Ni 2p₃/₂) and 873.7 eV (Ni 2p₁/₂), with corresponding satellites, and an O 1s peak at 531.2 eV indicative of Ni–O–Ni bonds.
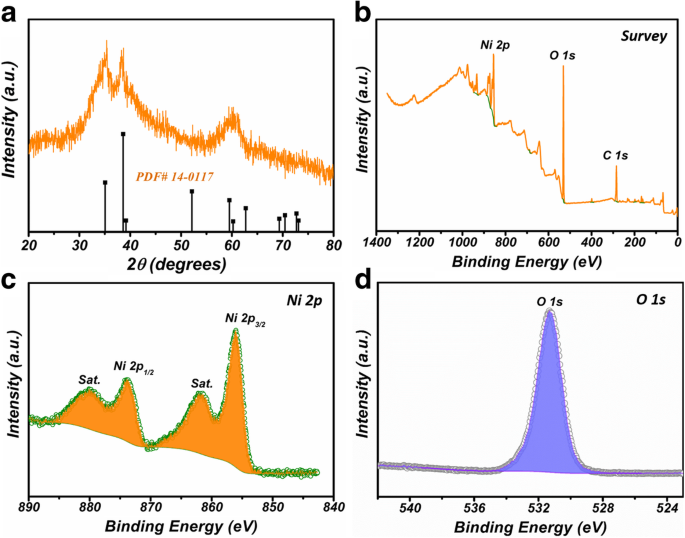
a XRD of Ni(OH)₂ NCs; b–d XPS spectra (survey, Ni 2p, O 1s).
SEM/TEM reveal cubic cages (∼500 nm edge) with a 50‑nm thick, porous shell and an internal void, confirming the hollow architecture. BET analysis shows a surface area of 54.7 m² g⁻¹ and a mesoporous distribution centered at 2.7–6.1 nm, superior to the 38.1 m² g⁻¹ of the BNCs.
Electrochemical Performance
At 1 A g⁻¹, Ni(OH)₂ NCs/NF deliver 539.8 F g⁻¹, compared with 87.3 F g⁻¹ for BNCs/NF. Capacitance remains 96.9 % after 2 000 cycles at 8 A g⁻¹. The NCs exhibit a lower charge‑transfer resistance (120.8 Ω vs. 976.5 Ω) and a steeper Warburg slope, indicating rapid ion diffusion.
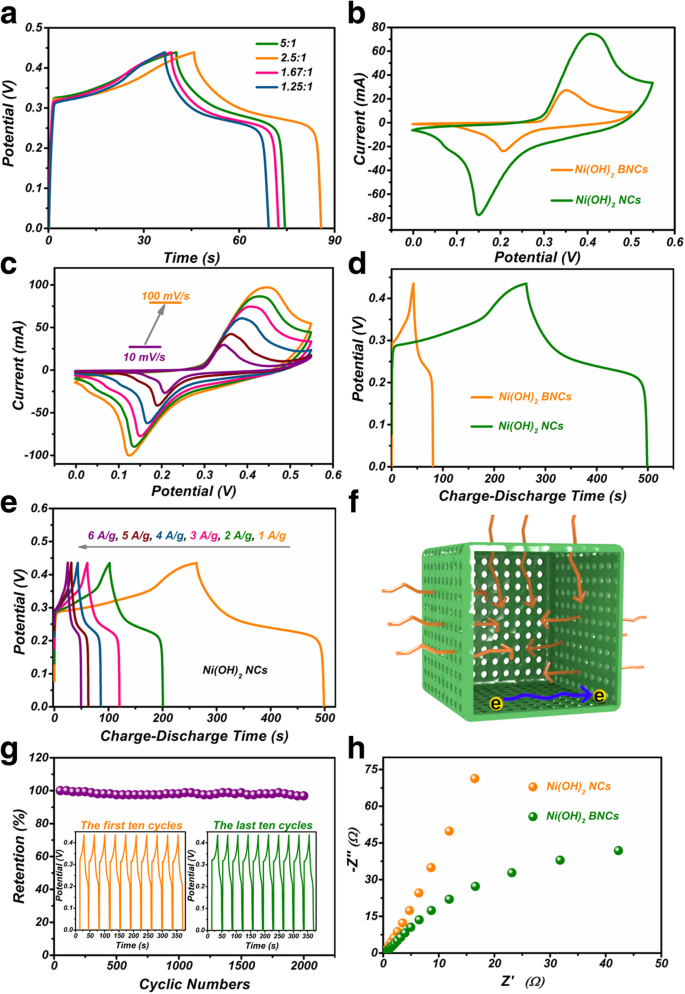
a GCD curves; b CVs at 60 mV s⁻¹; c CVs at varying scan rates; d GCD comparison; e GCD at different currents; f Charge‑storage schematic; g Cycling stability; h EIS spectra.
ASC Device Performance
The asymmetric cell (Ni(OH)₂ NCs/NF//AC) operates at 1.6 V. It delivers 23.3 Wh kg⁻¹ at 800 W kg⁻¹ and retains 9.6 Wh kg⁻¹ at 8000 W kg⁻¹. After 2 000 cycles at 4 A g⁻¹, the capacitance is 90.1 % of the initial value. SEM images confirm the preservation of the cage morphology post‑cycling.
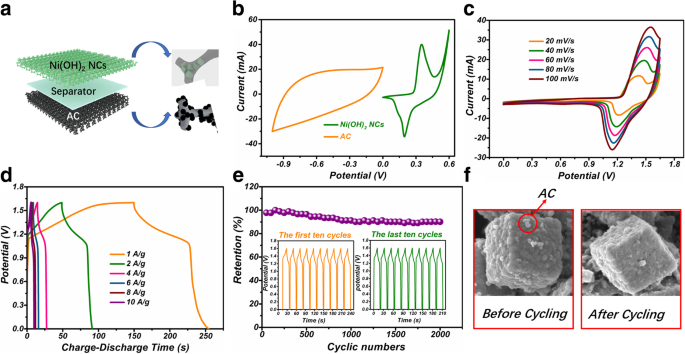
a ASC schematic; b CVs of individual electrodes; c ASC CV; d GCD curves; e Cycling stability; f SEM before/after cycling.
Conclusions
The CEP‑derived cubic Ni(OH)₂ nanocages combine high surface area, thin porous shells, and a well‑defined cavity to achieve record‑setting specific capacitance and energy density for asymmetric supercapacitors. Their excellent cycling stability and scalable synthesis position them as strong candidates for next‑generation energy‑storage systems.
Nanomaterials
- Fluorescent Nanomaterials: Advances, Synthesis, and Biomedical Applications
- CuGeO3 Nanowires: A High‑Capacity, Stable Anode for Advanced Sodium‑Ion Batteries
- Rapid, One‑Pot Synthesis of Nitrogen‑Doped Microporous Carbon Spheres for Superior Symmetric Supercapacitor Performance
- Ultra‑High Capacity Supercapacitor Electrodes from One‑Step Hydrothermal Synthesis of Chlorine‑Doped Carbonated Cobalt Hydroxide Nanowires on Nickel Foam
- Sea Urchin‑Shaped NiCo₂O₄ via Charge‑Driven Self‑Assembly for Superior Li‑Ion Battery Performance
- High‑Performance NiO Flakes@CoMoO₄ Nanosheets Core‑Shell Electrode on Ni Foam for Advanced Supercapacitors
- Large-Scale Silicon Nanowire Arrays on 6‑inch Mono‑ and Multi‑Crystalline Solar Cells via Enhanced Metal‑Assisted Chemical Etching
- Advanced c‑Si/c‑SiGe Wheel Test Structures for Precise Evaluation of Wet Etchant Anisotropy and Selectivity
- Eco-Friendly Microplasma Synthesis of Yellow-Emitting Carbon Quantum Dots for Cancer Cell Imaging and Photocatalytic Inactivation
- High‑Performance CNC Lathes & VMCs for Precision Machining



