High‑Performance NiO Flakes@CoMoO₄ Nanosheets Core‑Shell Electrode on Ni Foam for Advanced Supercapacitors
Abstract
Transition‑metal oxides (TMOs) have attracted attention as pseudocapacitor electrodes because of their high theoretical capacitance, abundance, and environmental friendliness. However, their practical specific capacitance often falls short of theory, and volume changes during cycling compromise stability. A core‑shell architecture can synergistically combine the fast electron pathways of the core with the abundant redox sites of the shell, mitigating these issues. Here, we report a two‑step hydrothermal synthesis of NiO flakes@CoMoO₄ nanosheets (NSs) grown on nickel foam (NF) to form a 2‑D branched core‑shell architecture. The vertically aligned CoMoO₄ NSs provide ample active sites, efficient ion channels, and enhanced electron transport. As a positive electrode, the NiO flakes@CoMoO₄ NSs/NF delivers an impressive specific capacitance of 1,097 F g⁻¹ at 1 A g⁻¹ and retains 97.5 % after 2,000 cycles. An asymmetric supercapacitor (ASC) assembled with AC/NF as the negative electrode achieves 25.8 Wh kg⁻¹ at 894.7 W kg⁻¹, while preserving 100 % of its capacity over 3,000 cycles. These results highlight the 2‑D branched core‑shell design as a powerful strategy for high‑performance TMO electrodes.
Introduction
With the rapid adoption of renewable energy, the demand for safe, high‑power, and long‑life energy storage solutions is escalating. Supercapacitors, offering fast charge–discharge cycles, high power density, and robust safety, are increasingly relevant. They are traditionally divided into electric double‑layer capacitors (EDLCs) and pseudocapacitors; the latter store charge via surface redox reactions, enabling higher energy densities.
TMOs are attractive pseudocapacitor materials because of their high theoretical capacitance, low cost, and environmental compatibility. Yet, practical capacitance is often limited by incomplete material utilization and volume expansion during cycling. Two effective strategies address these challenges: (1) directly growing active materials on conductive substrates (e.g., Ni foam) to eliminate “dead” layers and improve conductivity; (2) engineering nanostructures, such as core‑shell architectures, to harness synergistic electronic and ionic transport pathways.
Traditional core‑shell designs, however, suffer from shell shielding that restricts core accessibility. To overcome this, we developed a 2‑D branched core‑shell structure where CoMoO₄ nanosheets grow vertically on NiO flakes. This configuration maximizes surface contact, enhances electron pathways, and creates porous channels that accommodate electrolyte diffusion and relieve stress during cycling. The resulting NiO flakes@CoMoO₄ NSs/NF electrode exhibits exceptional electrochemical performance, demonstrating the promise of branched core‑shell architectures for next‑generation supercapacitor electrodes.
Methods Section
Synthesis of NiO Flakes/NF
Nickel foam (NF, 1.5 × 3.5 cm²) was first etched in 3 M HCl for 2 h to remove surface oxides, then dried at 60 °C for 12 h. The pretreated NF was immersed in 32 mL deionized water and transferred to a 40 mL stainless‑steel autoclave. The autoclave was sealed and heated to 140 °C for 24 h (step 1). After natural cooling, the product was washed, dried at 60 °C, and annealed at 400 °C for 2 h (step 2) to convert β‑Ni(OH)₂ into NiO. The procedure is illustrated in Fig. 1.
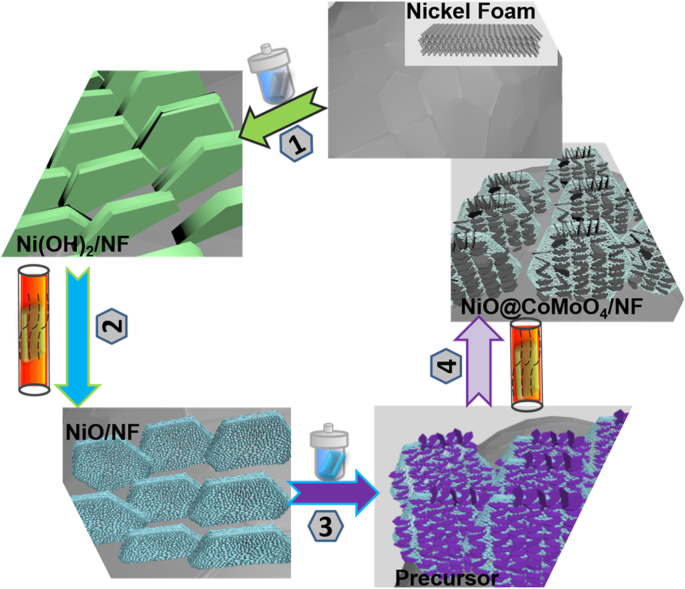
The synthesis illustration of NiO flakes@CoMoO₄ NSs electrode
Synthesis of NiO Flakes@CoMoO₄ NSs/NF
A solution of CoCl₂·6H₂O (65.1 mg) and Na₂MoO₄·2H₂O (50.8 mg) in 23 mL deionized water was prepared under stirring. NiO flakes/NF was immersed for 30 min, then placed in a 40 mL autoclave and heated to 160 °C for 6 h (step 3). Post‑processing involved ultrasonic cleaning, vacuum drying at 60 °C for 12 h, and final calcination at 400 °C for 2 h (step 4) to form NiO flakes@CoMoO₄ NSs/NF. CoMoO₄ flakes/NF were fabricated similarly but using bare NF.
Materials Characterization
X‑ray diffraction (Rigaku D/Max‑02400, Cu Kα) confirmed phase purity. FESEM (Zeiss Gemini, Hitachi SU8100) and HRTEM (JEM‑2100F) revealed morphology and crystal structure. XPS (Thermo ESCALAB 250Xi) identified elemental states. Brunauer–Emmett–Teller (BET) analysis (BELSORP‑max) measured specific surface area and porosity.
Electrochemical Measurements
All electrochemical tests used a μIII Autolab workstation in 6 M KOH. Three‑electrode tests employed saturated Ag/AgCl (reference) and Pt foil (counter), with NiO flakes@CoMoO₄ NSs/NF as working electrode (1 cm²). GCD and CV measured capacitance; EIS covered 100 kHz–0.01 Hz. Mass loadings were determined from weight changes before and after synthesis (0.79 mg cm⁻² for NiO flakes, 1.14 mg cm⁻² for CoMoO₄ NSs, 1.93 mg cm⁻² for the composite). Asymmetric supercapacitor (ASC) cells used 6 M KOH with NiO flakes@CoMoO₄ NSs/NF as positive and AC/NF as negative electrode. Mass balance followed equation (2) to match charge capacities of each electrode.
Results and Discussion
Characterizations
XRD patterns (Fig. 2a) confirm NiO formation after calcination and the emergence of CoMoO₄ peaks at 26.5°, 29.1°, 32.1°, and 33.7°, corresponding to (002), (310), (±1 31), and (±2 22) planes. No impurity peaks appear, confirming phase purity. XPS spectra (Fig. 2b‑f) show Co²⁺/Co³⁺, Mo⁶⁺, Ni²⁺/Ni³⁺, and lattice oxygen, verifying successful composite formation.
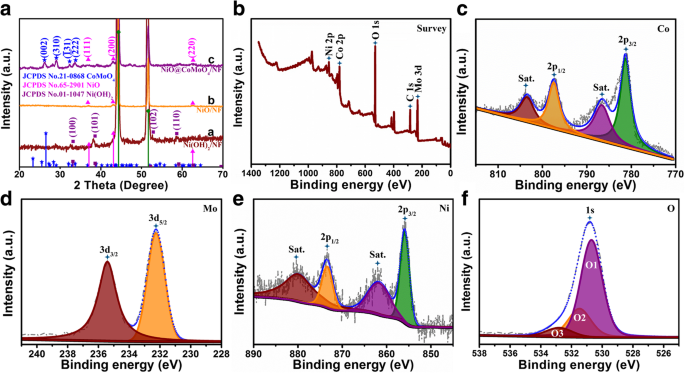
a XRD patterns of Ni(OH)₂ flakes/NF (curve a), NiO flakes/NF (curve b), and NiO flakes@CoMoO₄ NSs/NF (curve c). b XPS survey. c Co 2p. d Mo 3d. e Ni 2p. f O 1s.
SEM images (Fig. 3a‑i) reveal the evolution from porous Ni(OH)₂ flakes to NiO flakes with mesoporous features, and finally to branched CoMoO₄ nanosheets densely covering the flakes. HRTEM confirms crystal lattice spacings of 0.242 nm and 0.148 nm for NiO (111 and 220) and 0.199 nm/0.196 nm for CoMoO₄ (± 4 03 and ± 5 11). BET analysis (Fig. 4g‑i) shows specific surface areas of 28.2, 45.3, and 53.5 m² g⁻¹ for Ni(OH)₂, NiO, and the composite, respectively, with mean pore sizes of 6.13, 6.57, and 4.16 nm. The higher surface area and smaller pores in the composite facilitate electrolyte access and ion transport.

The SEM images of a–c Ni(OH)₂ flakes/NF, d–f NiO flakes/NF, and g–i NiO flakes@CoMoO₄ NSs/NF with different magnifications.
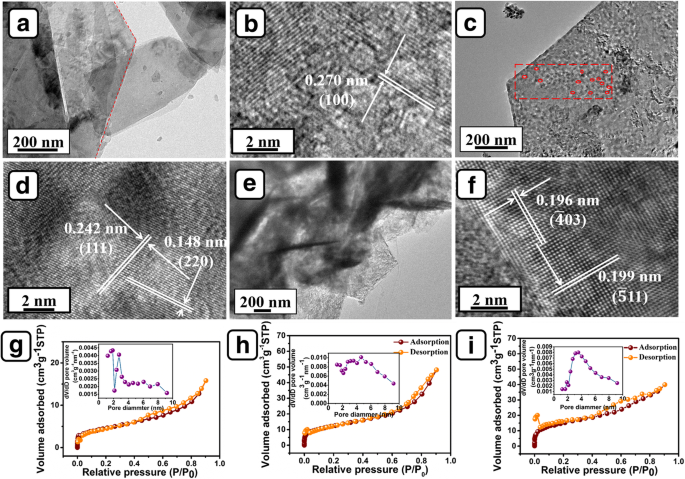
HRTEM images of a, b Ni(OH)₂ flakes, c, d NiO flakes, e, f NiO flakes@CoMoO₄ NSs; g–i nitrogen adsorption–desorption isotherms of Ni(OH)₂ flakes/NF, NiO flakes/NF, and NiO flakes@CoMoO₄ NSs/NF, respectively. Insets of (g–i) are the corresponding pore size distributions.
Electrochemical Performance of NiO@CoMoO₄/NF
Specific capacitance was calculated using Eq. (3). The composite achieves 1,097 F g⁻¹ at 1 A g⁻¹, significantly higher than NiO flakes (349 F g⁻¹) and CoMoO₄ flakes (316 F g⁻¹). Capacitance remains above 500 F g⁻¹ even at 10 A g⁻¹, indicating excellent rate capability. CV curves (Fig. 5b) show pronounced redox peaks and a large enclosed area, confirming pseudocapacitive behavior. GCD curves (Fig. 5c) exhibit symmetric shapes and long discharge times. The 2‑D branched structure provides abundant active sites and short ion diffusion paths, boosting performance (Fig. 5f).
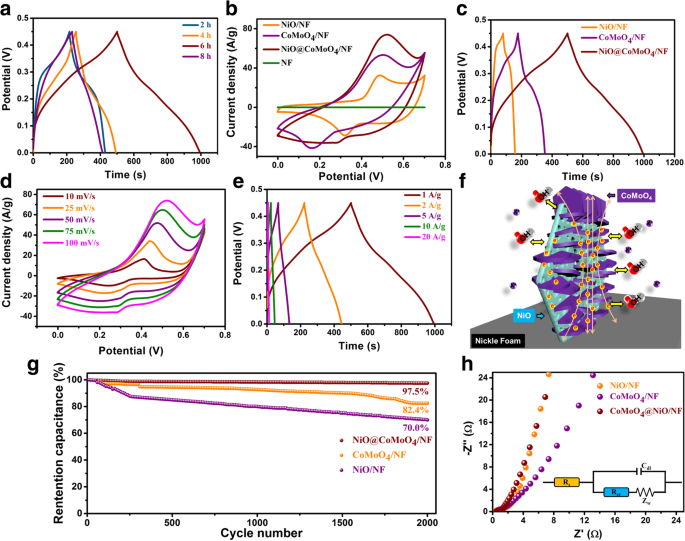
a GCD curves at different reaction times (2 h, 4 h, 6 h, 8 h) at 1 A g⁻¹. b CV curves of NiO flakes/NF, CoMoO₄ flakes/NF, and NiO flakes@CoMoO₄ NSs/NF at 100 mV s⁻¹. c GCD curves of the three electrodes at 1 A g⁻¹. d CV curves of the composite at various scan rates. e GCD curves at different current densities. f Schematic of structural advantages. g Cycling stability up to 2,000 cycles. h EIS spectra.
After 2,000 cycles at 2 A g⁻¹, the composite retains 97.5 % of its initial capacitance, whereas NiO flakes and CoMoO₄ flakes drop to 82.4 % and 70 %, respectively. EIS analysis shows lower series resistance (0.4 Ω) and charge‑transfer resistance (0.21 Ω) for the composite compared to its constituents, underscoring superior electron kinetics.
ASC Performance (NiO Flakes@CoMoO₄ NSs/NF//AC/NF)
The ASC operates over a 1.8 V window (Fig. 6a). CV curves (Fig. 6b) reveal EDLC behavior of AC and pseudocapacitance of the composite, and the device maintains shape at high scan rates. GCD curves (Fig. 6d) at 1–5 A g⁻¹ produce an energy density of 25.8 Wh kg⁻¹ at 894.7 W kg⁻¹, and still deliver 16.8 Wh kg⁻¹ at 4,500 W kg⁻¹. A single LED powered for 10 min demonstrates practical applicability. The ASC shows 100 % capacity retention after 3,000 cycles at 5 A g⁻¹, with negligible morphological change (Fig. 6f).
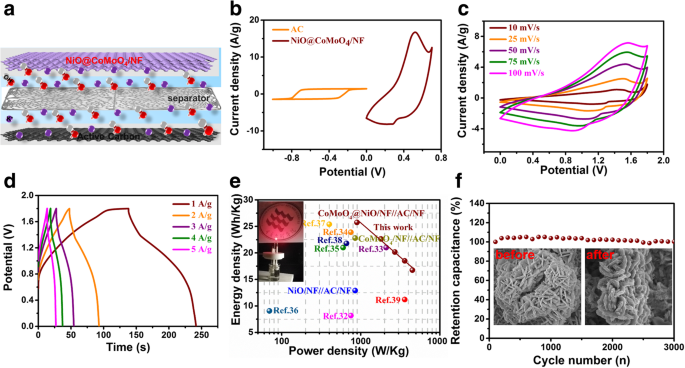
a ASC structure. b CV of NiO flakes@CoMoO₄ NSs/NF and AC. c CV of the ASC at various scan rates. d GCD of the ASC at different current densities. e Ragone plot compared to other NiO/CoMoO₄ electrodes. f Cycling stability over 3,000 cycles. Inset: SEM before/after cycling.
Conclusion
We have successfully fabricated a NiO flakes@CoMoO₄ nanosheet core‑shell electrode on Ni foam via a two‑step hydrothermal route. The 2‑D branched architecture delivers a high specific capacitance of 1,097 F g⁻¹, low charge‑transfer resistance, and outstanding cycling stability (97.5 % after 2,000 cycles). When paired with AC, the resulting ASC achieves 25.8 Wh kg⁻¹ at 894.7 W kg⁻¹ and retains full capacity over 3,000 cycles. These findings confirm that branched core‑shell designs can unlock the full potential of TMOs for high‑performance supercapacitors.
Availability of Data and Materials
All data are fully available without restriction.
Abbreviations
- 2D:
Two‑dimensional
- AC:
Active carbon
- ASC:
Asymmetric supercapacitor
- CV:
Cyclic voltammetry
- EDLCs:
Electric double‑layer capacitors
- EIS:
Electrochemical impedance spectroscopy
- FESEM:
Field emission scanning electron microscopy
- GCD:
Galvanostatic charge‑discharge
- HRTEM:
High‑resolution transmission electron microscopy
- NF:
Ni foam
- NSs:
Nanosheets
- PTFE:
Polytetrafluoroethylene
- TMOs:
Transition metal oxides
- XPS:
X‑ray photoelectron spectroscopy
- XRD:
X‑ray diffractometer
Nanomaterials
- Sol‑Gel Fabricated SiO₂@C/MWNT Nanocomposites Deliver Superior Li‑Ion Battery Anodes
- Enhancing Supercapacitor Energy Density with Anionic Surfactant‑Intercalated Thermally Reduced Graphene Oxide in Ionic Liquid Electrolytes
- Ni(OH)₂ Hollow‑Porous Architecture Enables Ultra‑Sensitive, Enzyme‑Free Glucose Detection
- Ultra‑High Capacity Supercapacitor Electrodes from One‑Step Hydrothermal Synthesis of Chlorine‑Doped Carbonated Cobalt Hydroxide Nanowires on Nickel Foam
- 3D Honeycomb‑Like SnS₂ Quantum Dot / rGO Composites: A High‑Performance Anode for Lithium and Sodium‑Ion Batteries
- One‑Step In Situ Assembly of Cypress‑Leaf‑Like Cu(OH)₂/Graphene Nanosheets for Ultra‑Stable, High‑Performance Supercapacitor Electrodes
- Cubic Ni(OH)₂ Nanocage Synthesis via Coordinated Etching–Precipitation for Superior Supercapacitor Performance
- Engineered Nickel‑Cobalt Layered Double Hydroxides: Tunable Thin‑Layer Nanosheets for Superior Supercapacitor Performance
- Bakelite FoamSet: Lightweight, Heat‑Stable, Flame‑Retardant Foams for Injection & Compression Molding
- High‑Voltage PCB Design: Materials, Safety, and Best Practices



