One‑Step In Situ Assembly of Cypress‑Leaf‑Like Cu(OH)₂/Graphene Nanosheets for Ultra‑Stable, High‑Performance Supercapacitor Electrodes
Abstract
Transition‑metal hydroxides combined with graphene are emerging as next‑generation electrode materials for energy storage, offering high capacity, excellent conductivity, and robust cycling life. Here we present a facile, single‑step, in‑situ synthesis of a cypress‑leaf‑shaped Cu(OH)₂ nanostructure anchored on graphene nanosheets, grown directly on copper foam in a two‑electrode setup. The electrostatic attraction between negatively charged graphene and Cu(OH)₂ drives a self‑assembly process that yields a highly porous, open framework. This architecture promotes rapid ion transport and mitigates volume changes during charge–discharge, resulting in a peak specific capacitance of 317 mF cm⁻² at 1 mA cm⁻² and unprecedented stability—100 % retention after 20 000 cycles—and excellent rate capability even at high current densities.
Introduction
The depletion of fossil fuels and the growing environmental concerns demand sustainable energy storage solutions for electronics and hybrid vehicles. Supercapacitors (SCs) have attracted attention for their high power density, rapid charge‑discharge, long life, and safety. Two main SC types exist: pseudocapacitors, relying on fast Faradaic redox reactions, and electric double‑layer capacitors (EDLCs), which store charge electrostatically. Carbon materials are typical EDLC electrodes due to their abundance, low toxicity, large surface area, and conductivity, yet their specific capacitance is comparatively low. Transition‑metal hydroxides—Ni(OH)₂, NiO, MnO₂, Co₃O₄—offer higher pseudocapacitance through surface redox but suffer from poor conductivity and large volume changes, limiting cycle life. Combining these hydroxides with conductive carbon, especially graphene, can enhance conductivity, stabilize the structure, and improve overall performance. Cu(OH)₂ stands out as a promising candidate because it is naturally abundant, environmentally benign, and exhibits a fast Cu²⁺/Cu⁺ redox couple. Graphene, with its exceptional surface area and intrinsic conductivity, can further boost capacity and stability. Prior studies reported Cu(OH)₂/graphene composites with capacitances up to 602 F g⁻¹ and good retention, but the synthesis routes were complex and cycling stability still needed improvement. Here we introduce a straightforward, single‑step, in‑situ self‑assembly approach that produces a cypress‑leaf‑like Cu(OH)₂/graphene composite directly on Cu foam, yielding a binder‑free electrode with outstanding electrochemical performance.
Methods Section
Sample Preparation
Commercial Cu foam (10 × 15 × 1.6 mm³, Xiamen Yongchangshuo) and graphite foil (10 × 15 × 1.0 mm³, Shanghai Alfa Aesar) were cleaned by ultrasonic baths in absolute ethanol and deionized water for 15 min, then rinsed and dried. The two‑electrode cell consisted of the graphite foil as anode and Cu foam as cathode. The electrolyte was a 0.1 M (NH₄)₂SO₄ (100 mL) solution with 3 mL NH₃·H₂O. A constant 7 V DC was applied for 1 h, during which the graphite foil was electrochemically exfoliated into graphene nanosheets and the Cu foam was oxidized into cypress‑leaf‑shaped Cu(OH)₂ under the influence of NH₃. The electrostatic attraction between negatively charged graphene edges and Cu(OH)₂ surfaces drives the in‑situ assembly into a porous hybrid network, which is then air‑dried.
Reactions:
Cu + 6 NH₃ + 2 H₂O → [Cu(NH₃)₆]²⁺ + 2 OH⁻ + H₂↑ (1)
Cu²⁺ + 2 OH⁻ → Cu(OH)₂ (2)
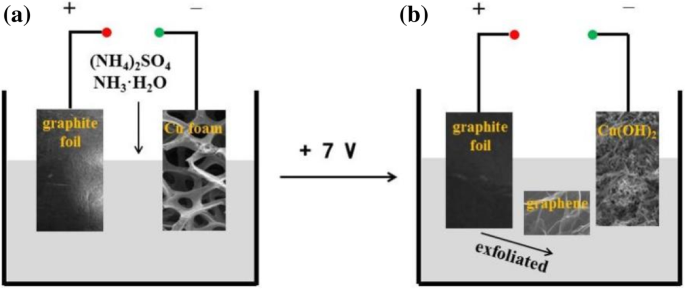
Schematic of the one‑step in‑situ self‑assembly of Cu(OH)₂/graphene composite
Characterizations
X‑ray diffraction (XRD) was performed on a Rigaku Ultima IV with Cu Kα radiation, scanning 10°–80° (2θ) at 2° min⁻¹. Raman spectra were recorded on a Renishaw Via‑reflex with a 532 nm laser. Morphology and structure were examined by FESEM (Zeiss Ultra Plus) and TEM (JEOL JEM‑2100F, 200 kV). X‑ray photoelectron spectroscopy (XPS) assessed surface chemistry and valence states.
Electrochemical Measurements
The Cu(OH)₂/graphene electrode on Cu foam was tested in a three‑electrode cell with Ag/AgCl reference and Pt counter electrodes in 1 M KOH. Cyclic voltammetry (CV), galvanostatic charge–discharge (GCD), and electrochemical impedance spectroscopy (EIS) were carried out on a PARSTAT 4000 and LAND CT‑2001A. CV and GCD operated from 0 V to 0.6 V; EIS spanned 0.01–100 kHz. Area capacitance was calculated by
C = J t / ΔV (3)
Results and Discussions
XRD confirmed the presence of crystalline Cu(OH)₂ (JCPDS 01‑080‑0656) and metallic Cu from the foam substrate (JCPDS 04‑0836). Sharp peaks indicate high crystallinity and phase purity. Raman spectra displayed the characteristic D (1349 cm⁻¹), G (1579 cm⁻¹), and 2D (2715 cm⁻¹) bands of graphene, verifying successful exfoliation.
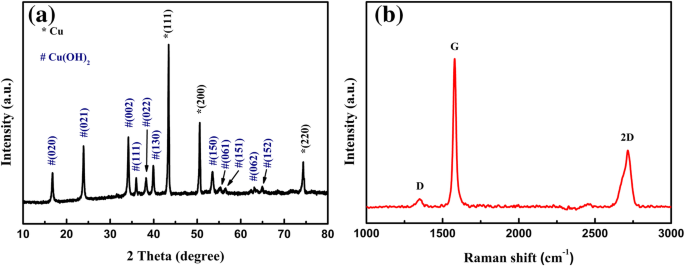
a XRD pattern and b Raman spectrum of the Cu(OH)₂/graphene composite
FESEM images reveal a highly porous, interconnected network where cypress‑leaf‑shaped Cu(OH)₂ nanorods (~300 nm length, 15 nm diameter) are uniformly distributed on transparent graphene sheets. TEM and SAED further confirm the crystallinity of both components. High‑resolution TEM shows lattice fringes of 0.22 nm corresponding to the (130) facet of Cu(OH)₂.

FESEM images of the Cu(OH)₂/graphene composite (low and high magnification)

a TEM image with SAED from graphene, b high‑magnification TEM of a Cu(OH)₂ branch, c HRTEM lattice fringe
XPS spectra confirm the presence of Cu²⁺ in Cu(OH)₂, graphene functional groups, and minor CuO. The Cu 2p peaks at 934.6 eV and 954.5 eV correspond to Cu²⁺; the C 1s peaks at 284.8 eV, 285.6 eV, and 288.5 eV indicate sp² carbon, C‑OH, and C=O groups, respectively.
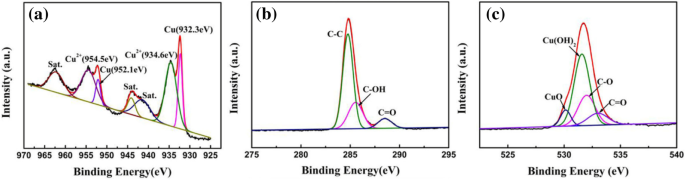
XPS spectra of a Cu 2p, b C 1s, and c O 1s
Electrochemical testing shows distinct redox peaks in CV curves, indicating reversible Cu²⁺/Cu⁺ transitions. The specific capacitance remains high across a wide scan‑rate range, demonstrating excellent rate capability. At 1 mA cm⁻², the area‑specific capacitance reaches 317 mF cm⁻², and it only decreases to 273 mF cm⁻² at 10 mA cm⁻² (a 14 % loss). The composite retains 100 % of its initial capacitance after 20 000 cycles at 2 mA cm⁻², with Coulombic efficiency consistently at 100 %. EIS reveals a low internal resistance (~2.35 Ω) and favorable Warburg impedance, indicative of efficient ion diffusion.
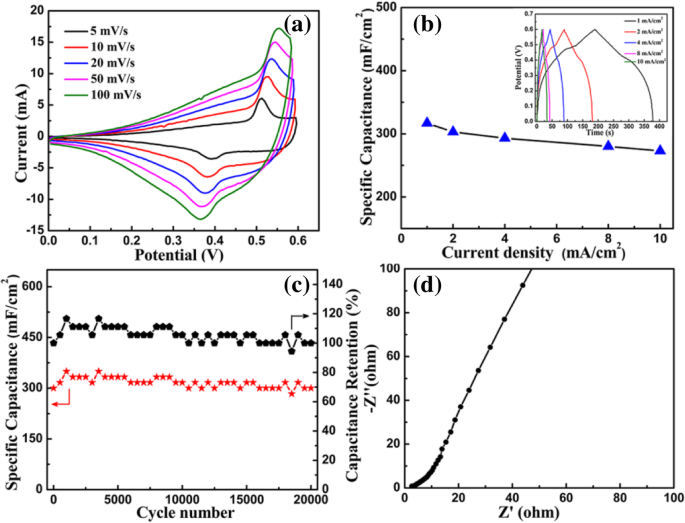
Electrochemical performance of the Cu(OH)₂/graphene composite: a CV curves, b GCD and capacitance, c area capacitance & Coulombic efficiency, d Nyquist plot
The superior performance stems from: (1) the conductive Cu foam scaffold providing a high surface area and mechanical support; (2) the binder‑free cypress‑leaf architecture minimizing dead volume and internal resistance; (3) graphene integration enhancing electrical conductivity and buffering volume changes; (4) the open, porous network ensuring ample ion access even at high rates; and (5) the intrinsic stability of the Cu(OH)₂/graphene hybrid during prolonged cycling.
Conclusions
We have demonstrated a simple, solution‑based, one‑step electrochemical synthesis of cypress‑leaf‑like Cu(OH)₂/graphene nanostructures directly on Cu foam, yielding a binder‑free electrode with outstanding supercapacitor performance. The hybrid delivers a reversible capacitance of 317 mF cm⁻², 100 % retention after 20 000 cycles, and excellent rate capability. This method offers a scalable route to high‑performance electrode materials for next‑generation energy storage devices.
Nanomaterials
- Hierarchical Activated Carbon Fibers from Waste Cotton Gloves: High‑Performance Electrodes for Supercapacitors
- Optimizing MoS2‑Graphene Hybrid Crystallization for Enhanced Electrocatalysis
- Ultra‑Sensitive Electrochemical DNA Biosensor for Vibrio cholerae Detection Using Polystyrene‑Acrylic Acid Nanoparticles and Gold Nanoparticles
- Hybrid Graphene/WO₃ and Graphene/CeOx Electrodes for High‑Performance Supercapacitors
- High‑Capacity Si/Graphene Composite Anode Fabricated via Magnesium‑Thermal Reduction for Li‑Ion Batteries
- Highly Stretchable, Electrically Conductive TPU–PANI Nanofibrous Membrane for Advanced Flexible Strain Sensors
- High‑Performance NiO Flakes@CoMoO₄ Nanosheets Core‑Shell Electrode on Ni Foam for Advanced Supercapacitors
- High‑Performance Flexible Microsupercapacitors Based on 3‑D rGO/PEDOT Open‑Network Architectures
- Egg‑Albumin Assisted Hydrothermal Synthesis of Mesoporous Co3O4 Quasi‑Cubes: High‑Performance Supercapacitor Electrode
- Engineered Nickel‑Cobalt Layered Double Hydroxides: Tunable Thin‑Layer Nanosheets for Superior Supercapacitor Performance



