Optimizing MoS2‑Graphene Hybrid Crystallization for Enhanced Electrocatalysis
Abstract
This study investigates how hydrothermal temperature (150–240 °C) controls the crystallization of molybdenum disulfide (MoS2) nanoflakes on graphene nanosheets. Flower‑shaped MoS2 nanoflakes are successfully anchored onto graphene, and their morphology, crystal phase, and electrochemical performance are correlated. The hybrid synthesized at 180 °C delivers the highest efficiency in both dye‑sensitized solar cells (DSSCs) and hydrogen evolution reactions (HERs), striking a balance between high reactivity and conductivity. These findings clarify temperature‑dependent MoS2 growth and provide actionable guidance for designing next‑generation catalytic hybrids.
Temperature‑dependent crystallization of MoS2 nanoflakes on graphene nanosheets for electrocatalysis
Background
Two‑dimensional (2D) hybrid nanomaterials are rapidly emerging across photovoltaics, water splitting, sensors, and energy storage. Their exceptional surface‑area‑to‑volume ratios and tunable electronic properties make them ideal for heterojunctions or 3D frameworks that integrate transition‑metal dichalcogenides (TMDs) with conductive graphene. In particular, MoS2/graphene hybrids combine the high surface reactivity of MoS2 with the exceptional electron transport of graphene, yielding catalysts that rival Pt in performance while dramatically reducing cost.
While pristine MoS2 suffers from low intrinsic conductivity and graphene alone exhibits modest redox activity, their synergistic assembly creates a network of active sites and continuous charge pathways. Prior work has shown that hydrothermal routes—leveraging graphene as a crystallization scaffold—offer a low‑cost, scalable approach to fabricate these hybrids. However, the precise influence of hydrothermal temperature on MoS2 crystallization and phase evolution in the presence of graphene remains underexplored.
Here we systematically vary the hydrothermal temperature (150–240 °C) to elucidate how growth conditions shape the morphology, crystal phase, and electrocatalytic performance of MoS2/graphene hybrids. By integrating structural characterization with DSSC and HER testing, we identify 180 °C as the optimal temperature that balances high crystallinity, defect‑mediated activity, and electrical conductivity.
Methods
Material Preparation and Characterization
Hydrothermal synthesis of MoS2/graphene hybrids was performed using microwave‑exfoliated graphene oxide (MEGO) as the substrate. MEGO was produced by 900 W microwave irradiation of graphite oxide for 90 s under argon, simultaneously reducing the material. 2.8 mg of MEGO was dispersed in 20 mL deionized water, followed by sequential addition of 42 mg sodium molybdate dihydrate and 84 mg thiourea (the excess thiourea further reduces MEGO). The suspension was transferred to 50 mL autoclaves and heated at 150, 180, 210, or 240 °C for 24 h, yielding MG‑150, MG‑180, MG‑210, and MG‑240, respectively. The solids were collected, washed, and dried under vacuum at 70 °C overnight.
Morphology and composition were examined by FE‑SEM (Hitachi S‑4800) and EDS mapping (Bruker detector). TEM and HRTEM (Hitachi H 9000 NAR) probed crystal structure, while XRD (Bruker D8 Discover) and Raman spectroscopy (Renishaw 1000B, 633 nm laser) assessed phase and crystallinity. XPS (VG ESCA 2000, Mg Kα) quantified Mo and S oxidation states and phase ratios.
DSSC Fabrication and Tests
FTO glass was sequentially cleaned with acetone, isopropanol, and DI water. A TiO2 photoanode was assembled by doctor‑blading a commercial TiO2 paste and sintering to 500 °C over 30 min. The TiO2 layer was then sensitized with a 0.5 mM N719 ethanolic solution for 24 h. Counter electrodes were fabricated by doctor‑blading the hybrid slurries (20 mg sample + 5 µL Triton X‑100 in 500 µL DI water) onto FTO, followed by annealing at 500 °C for 30 min in argon. Pt counter electrodes were prepared similarly using a 0.01 M H2PtCl6 ethanolic solution. DSSCs were assembled with commercial electrolyte injection and sealed with thermoplastic film.
Photovoltaic performance was measured under AM 1.5G (100 mW cm–2) illumination using a Keithley 2420 source meter. The system was calibrated with a Si reference cell. Electrochemical impedance spectroscopy (EIS) spanned 0.1–10 kHz under one‑sun illumination at 0.7 V bias, recorded on a CHI 760D workstation.
Electrochemical Measurements
HER and electrochemical tests employed a standard three‑electrode glass cell with a saturated Ag/AgCl reference (converted to RHE). All measurements were conducted in 0.5 M H2SO4 using a CHI 760D. Working electrodes were glassy‑carbon discs (3 mm diameter) coated with 5 mg of hybrid dispersed in 50 µL 5 % Nafion ethanol + 450 µL DI water, followed by drying. Linear sweep voltammetry (LSV) ran from 0.2 to –0.8 V vs. Ag/AgCl at 5 mV s–1; Tafel plots were derived from LSV. Cyclic voltammetry (CV) scanned –1 to 1 V at 50 mV s–1. EIS covered 0.1–10 kHz at 0.5 mV bias. Stability was evaluated over 20,000 s at –0.5 V vs. Ag/AgCl.
Results and Discussion
FE‑SEM images (Figure 1a–h) reveal that MoS2 nucleates as perpendicularly oriented flower‑like nanoflakes across all temperatures. As synthesis temperature rises, the flake size enlarges, and the MG‑240 sample transitions from layered to nanoparticle morphology, indicating a loss of layer‑by‑layer growth at the highest temperature. The increased branching at mid‑range temperatures is advantageous because edges and defects act as catalytic active sites.
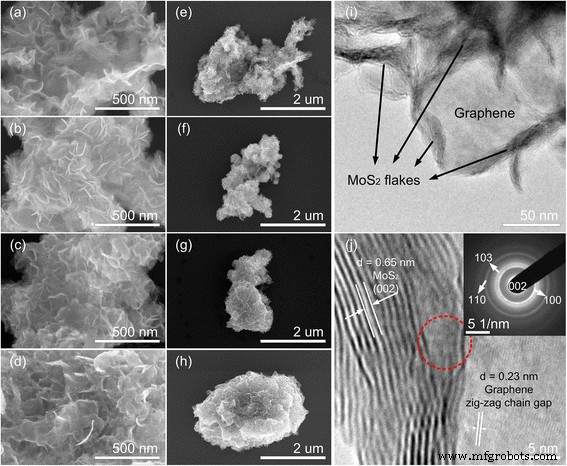
Morphology of MoS2/graphene hybrids. SEM images of hybrids at 150 °C (a, e), 180 °C (b, f), 210 °C (c, g), and 240 °C (d, h); TEM/HRTEM of MG‑180 (i, j). Inset of (j) shows the SAED pattern (dashed circle). Lattice spacings are annotated in (j).
TEM/HRTEM of the MG‑180 sample confirms the intimate interfacial contact between MoS2 lamellae and graphene. The 0.65 nm spacing matches the 2H‑MoS2 (002) plane, while the 0.23 nm spacing corresponds to the graphene zig‑zag chain. SAED patterns corroborate the 2H‑MoS2 crystallinity and the seamless stitching of graphene, indicating efficient electron transfer across the interface. Across all hybrids, higher temperatures yield sharper lattice fringes and better crystallinity.
XRD and Raman spectroscopy (Figure 2) further illustrate the temperature‑driven evolution. MG‑150 displays weak MoS2 peaks, reflecting limited crystallization. At 180 °C, the (103) and (105) 2H peaks sharpen, and an additional (006 + 104) feature suggests the emergence of a 1T phase. The (100) peak broadening and shift confirm lattice rearrangement. Raman A1g and E2g intensities rise with temperature, and the high A1g intensity in MG‑210 and MG‑240 indicates a perpendicularly oriented MoS2 arrangement. The D/G ratio of graphene also increases, signaling stronger van der Waals coupling with MoS2 at elevated temperatures.
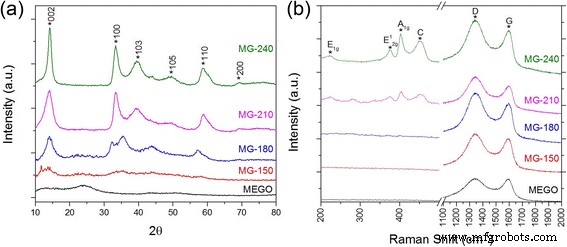
Crystallization comparison of MoS2/graphene hybrids. (a) XRD spectra of hybrids prepared at 150, 180, 210, and 240 °C versus MEGO. (b) Raman spectra of hybrids and MEGO. 2H peaks of MoS2 are labeled.
XPS analysis (Figure 3) confirms a temperature‑dependent phase transition. Peak sharpening from MG‑150 to MG‑240 indicates progressive crystallization. The Mo 3d binding energy shifts ~0.63 eV from 180 °C to 240 °C, consistent with a 1T→2H transition. Deconvolution yields 2H:1T ratios of 4.84:1 (MG‑150), 3.01:1 (MG‑180), 13.7:1 (MG‑210); MG‑240 shows only 2H. Thus, optimal performance requires a balanced 1T content to provide metallic conductivity without excessive defect‑induced charge trapping.
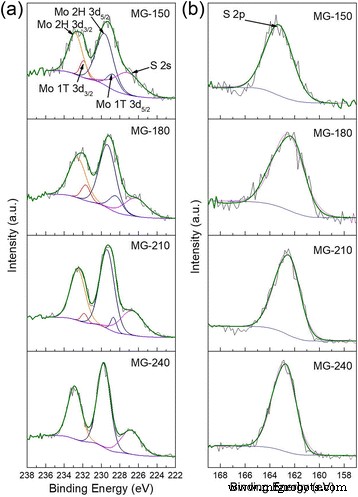
Binding analysis of MoS2/graphene hybrids. XPS spectra of hybrids at 150, 180, 210, and 240 °C. (a) Mo 3d orbits; (b) S 2p orbits.
Electrochemical testing reveals that the MG‑180 hybrid delivers the best catalytic performance. In DSSCs, all hybrids maintain a VOC of ~0.7 V, but MG‑180 achieves the highest short‑circuit current (isc) and power conversion efficiency, approaching that of Pt counter electrodes. MG‑210 and MG‑240 exhibit reduced isc due to diminished charge transport stemming from over‑stacked MoS2 layers. EIS data confirm the lowest charge‑transfer resistance for MG‑180, aligning with its superior performance.
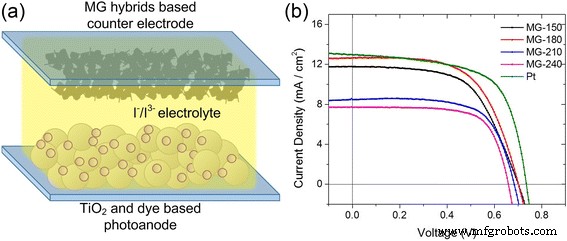
DSSC schematic and performance. (a) Schematic of DSSC with MoS2/graphene counter electrode. (b) J–V curves for hybrids; Pt delivers the highest efficiency, while MG‑180 closely follows. VOC declines as synthesis temperature increases beyond 210 °C.
HER testing shows MG‑180 also outperforms its counterparts. Onset potentials are –176 mV (MG‑150), –179 mV (MG‑180), and –287 mV (MG‑210). The MG‑180 Tafel slope (74.5 mV dec–1) is markedly lower than MG‑150 (137 mV dec–1) and MG‑210 (not shown), indicating faster kinetics. EIS confirms a smaller semicircle for MG‑180, reflecting efficient interfacial charge transfer. BET analysis yields a specific surface area of 73.5 m2 g–1 for MG‑180, comparable to MG‑210 but far exceeding MG‑150 (49.5 m2 g–1), corroborating the branched morphology observed by SEM.
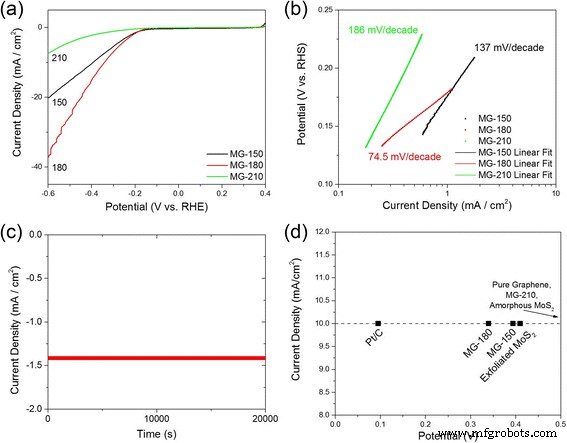
HER performance comparison. (a) Polarization curves (IR‑corrected). (b) Tafel plots for hybrids at 150, 180, and 210 °C. (c) Stability test of MG‑180 over 20,000 s at –0.5 V. (d) Overpotential comparison at 10 mA cm–2 versus Pt/C, exfoliated MoS2, and amorphous MoS2.
The MG‑180 hybrid demonstrates robust stability, maintaining performance over 20,000 s at –0.5 V, and outperforms commercial Pt/C and exfoliated MoS2 at the same current density. These results confirm that a hydrothermal temperature of 180 °C optimizes the defect density, 1T fraction, and morphological branching necessary for high catalytic activity.
Conclusions
By correlating structural evolution with electrocatalytic performance, this work establishes that MoS2 crystallization on graphene is highly temperature‑dependent. At < 180 °C, the hybrid retains a 1T‑rich, defect‑laden phase; above 210 °C, the material transitions to the more stable 2H phase with diminished defects. The 180 °C hybrid exhibits the optimal blend of high crystallinity, active edge sites, and robust charge transport, delivering DSSC efficiencies and HER kinetics that rival Pt‑based catalysts. These insights provide a clear design principle for tailoring 2D hybrid electrocatalysts via controlled hydrothermal synthesis.
Abbreviations
- 2D
Two‑dimensional
- 3D
Three‑dimensional
- BET
Brunauer‑Emmett‑Teller
- CV
Cyclic voltammetry
- DFT
Density functional theory
- DSSC
Dye‑sensitized solar cell
- EDS
Energy‑dispersive X‑ray spectroscopy
- EIS
Electrochemical impedance spectroscopy
- FE‑SEM
Field‑emission scanning electron microscope
- FF
Fill factor
- GCE
Glassy carbon electrode
- HER
Hydrogen evolution reaction
- HRTEM
High‑resolution transmission electron microscope
- isc
Short‑circuit current
- LSV
Linear sweep voltammetry
- MEGO
Microwave‑exfoliated graphene oxide nanosheets
- RHE
Reversible hydrogen electrode
- SAED
Selected area electron diffraction
- TEM
Transmission electron microscope
- TMD
Transition metal dichalcogenide
- VOC
Open‑circuit voltage
- XPS
X‑ray photoelectron spectroscopy
- XRD
X‑ray diffraction
Nanomaterials
- MoS₂ Films of Precise Thickness Enhance Electrocatalytic Hydrogen Evolution
- High-Capacity Few-Layered MoS₂/Acetylene Black Anode for Li‑Ion Batteries
- Controlling Photoresponsivity in Multi‑Layer MoS₂ Phototransistors Through Applied Bias
- Hybrid Graphene/WO₃ and Graphene/CeOx Electrodes for High‑Performance Supercapacitors
- Binder‑Free 3‑D MoS₂/Graphene Aerogel Anode Delivering 1,041 mAh g⁻¹ for Lithium‑Ion Batteries
- Predicting Surface Impedance of Metasurface–Graphene Hybrid Structures in the Terahertz Regime
- One‑Step In Situ Assembly of Cypress‑Leaf‑Like Cu(OH)₂/Graphene Nanosheets for Ultra‑Stable, High‑Performance Supercapacitor Electrodes
- Precise Band Alignment of 2D MoS₂ on 3D β‑Ga₂O₃ Heterojunctions: Impact of Nitridation
- R‑Scheme Ti3C2 MXene/MoS2 2D/2D Heterojunctions: A Breakthrough in Photocatalytic Efficiency
- Hierarchically Porous MoS₂/C Nanospheres from Nanosheets: Exceptional Electrochemical Energy Storage



