Binder‑Free 3‑D MoS₂/Graphene Aerogel Anode Delivering 1,041 mAh g⁻¹ for Lithium‑Ion Batteries
Abstract
Hybrid MoS₂/reduced graphene aerogels with a rich microporous network were fabricated by hydrothermal synthesis followed by freeze‑drying and high‑temperature annealing. The resulting porous structure functions directly as a binder‑free electrode, enhancing electron transport and creating a three‑dimensional ion‑transport pathway. Consequently, the aerogel delivers a specific capacity of 1,041 mAh g⁻¹ at 100 mA g⁻¹ and retains 667 mAh g⁻¹ after 100 cycles—58.6 % retention—at 100 mA g⁻¹. These impressive metrics arise from the synergistic combination of MoS₂ nanosheets and conductive graphene, coupled with the binder‑free architecture. This work demonstrates a scalable route to integrate transition‑metal dichalcogenides with graphene for high‑performance, porous electrode materials in energy storage devices.
Introduction
The accelerating adoption of electric vehicles and flexible electronics has intensified research into next‑generation energy storage. Lithium‑ion batteries (LIBs) remain the benchmark for high energy density and long cycle life, making them a focal point for material innovation.
Two‑dimensional transition‑metal dichalcogenides (TMDs) have attracted significant attention as LIB anodes because of their layered structure, high theoretical capacity (~670 mAh g⁻¹ for MoS₂), and relatively large interlayer spacing, which facilitate Li⁺ diffusion. However, volume expansion during lithiation/delithiation and low electronic conductivity limit practical performance. Strategies to mitigate these drawbacks include nanosizing the active material and integrating carbonaceous hosts such as graphene.
Graphene’s exceptional conductivity and large specific surface area make it an ideal scaffold to support MoS₂ nanosheets, preventing restacking and buffering volume changes. Recent studies—e.g., Teng et al. achieving 1,077 mAh g⁻¹ at 100 mA g⁻¹ after 150 cycles, and Liu et al. reporting 1,300–1,400 mAh g⁻¹—demonstrate the promise of MoS₂/graphene composites. Yet, a binder‑free architecture that combines a hierarchical porous network with strong MoS₂/graphene interactions remains underexplored.
Here we present a facile, low‑cost synthesis of a hierarchical MoS₂/reduced graphene oxide (RGO) aerogel via solvothermal treatment, freeze‑drying, and annealing. The resulting binder‑free aerogel offers a three‑dimensional conductive network that promotes rapid electron transfer, reduces Li⁺ diffusion paths, and accommodates volume changes, achieving superior electrochemical performance as an LIB anode.
Materials and Methods
Synthesis of MoS₂/RGO Aerogels
All reagents were of analytical grade. Graphene oxide (GO) was prepared by a modified Hummers’ method. For the aerogel synthesis, 60 mg of (NH₄)₂MoS₄ was dissolved in 10 mL DMF, then 5 mL of 5 mg mL⁻¹ GO aqueous solution was added. The mixture was sonicated for several hours to achieve a homogeneous dispersion. It was sealed in a Teflon‑lined autoclave and heated at 200 °C for 12 h. The resulting hydrogel was washed with ethanol and deionised water, then freeze‑dried and annealed at 700 °C for 2 h to yield the final MoS₂/RGO aerogel. For comparison, MoS₂ powder was prepared following the same procedure without GO addition.
Characterization
Thin sections of the aerogel were examined by FESEM (JEOL JSM‑6700F) and FETEM (FEI, Tecnai G2 F30). XRD (PANalytical PW3040/60, Cu Kα, λ = 1.5406 Å) confirmed phase composition from 10° to 80°. Raman spectra were recorded to assess MoS₂ layer thickness and graphene defect density.
Electrochemical Measurements
The aerogel film served as a binder‑free anode in coin‑type half‑cells (Li foil counter, Celgard 2400 separator). The electrolyte was 1 M LiPF₆ in EC/DEC. After a 24 h rest, galvanostatic charge/discharge (GCD) tests were conducted with a Land system; cyclic voltammetry (CV) was performed on an Autolab PGSTAT‑302N within 0.01–3.0 V (vs Li⁺/Li). Electrochemical impedance spectroscopy (EIS) used a 10 mV amplitude from 100 kHz to 0.01 Hz.
Results and Discussion
The synthesis route—hydrothermal treatment, freeze‑drying, and annealing—is illustrated in Figure 1. The resulting aerogel retained an integrated, porous architecture (Additional file 1: Fig. S1, Additional file 2: Fig. S2), confirming its suitability as a binder‑free electrode.
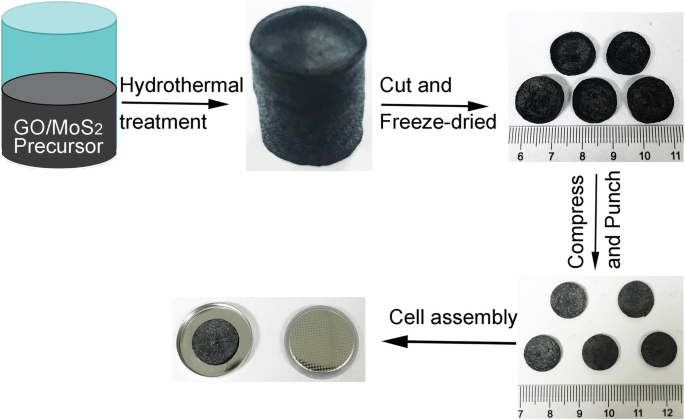
Schematic of fabrication of hybrid nanostructure of MoS₂/RGO
SEM images (Figure 2a) reveal a wrinkled graphene network interlaced with MoS₂ nanosheets. TEM confirms uniform MoS₂ coverage (Figures 2c–2f) with lattice spacings of 0.61 nm (002) and 0.27 nm (100), indicating high crystallinity. EDX mapping (Figures 2g–2j) shows homogeneous distribution of Mo, S, and C.

Figure 2. a, b SEM images; c–f TEM/HRTEM; g–j EDX mapping of Mo, S, and C. Inset in f: SAED pattern.
XRD (Figure 3a) shows the characteristic peaks of hexagonal 2H‑MoS₂ (JCPDS 37‑1492). A prominent (002) peak at 14.2° (d = 0.62 nm) and a graphene (002) peak at 26.3° confirm successful composite formation. The reduced intensity of MoS₂ (002) indicates a few‑layer configuration, while the graphene peaks are more pronounced, evidencing extensive MoS₂ wrapping by RGO.
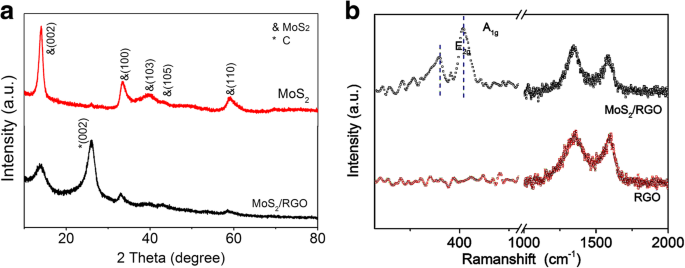
Figure 3. a XRD patterns of MoS₂/RGO and MoS₂; b Raman spectra.
Raman analysis (Figure 3b) shows E₂g and A₁g peaks at 380.2 cm⁻¹ and 403.6 cm⁻¹, respectively, consistent with few‑layer MoS₂. The D and G bands of graphene appear at 1354.3 cm⁻¹ and 1591.6 cm⁻¹, with an I_D/I_G ratio of 1.08, indicating reduced graphene with moderate defect density.
CV curves (Figure 4a) display a broad shoulder at 0.95 V and a reduction peak at 0.65 V during the first cathodic sweep, corresponding to Li⁺ intercalation (forming LixMoS₂) and subsequent conversion to Li₂S and metallic Mo. Subsequent cycles exhibit stable redox peaks, confirming reversible behavior.
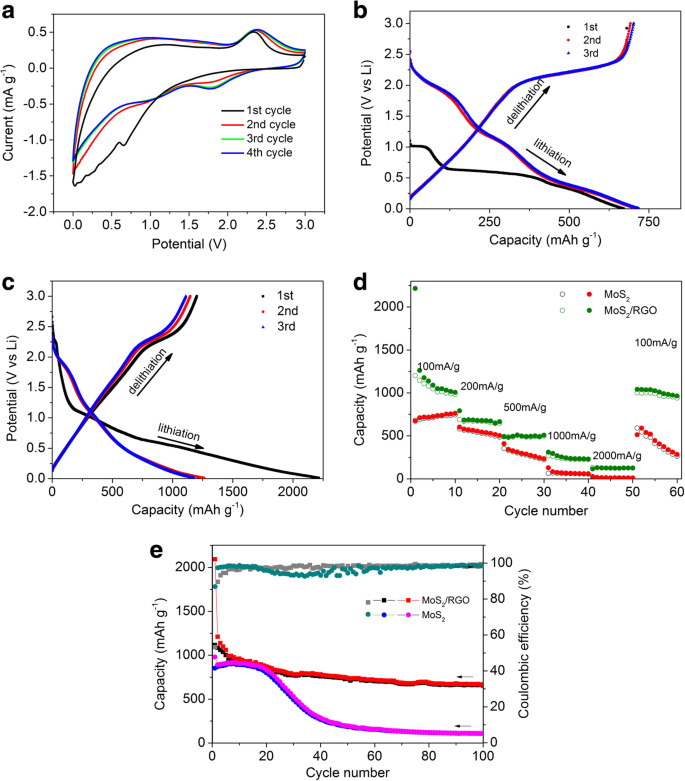
Figure 4. a First three CV curves at 0.5 mV s⁻¹; b GCD curves of MoS₂/RGO; c GCD curves of MoS₂; d Rate performance; e Cycling performance at 100 mA g⁻¹.
The initial discharge capacity of the MoS₂/RGO aerogel reaches 2,215 mAh g⁻¹, with a reversible charge capacity of 1,202 mAh g⁻¹. In contrast, pure MoS₂ delivers 671.1 mAh g⁻¹ (discharge) and 680.5 mAh g⁻¹ (charge). After 50 cycles, MoS₂/RGO retains 1,041 mAh g⁻¹, whereas MoS₂ falls to 512 mAh g⁻¹.
Cycling at 100 mA g⁻¹ shows MoS₂/RGO maintaining 667 mAh g⁻¹ (58.6 % retention) after 100 cycles, while MoS₂ drops to 110 mAh g⁻¹ (12.3 % retention). RGO alone delivers 297.8 mAh g⁻¹ at 100 mA g⁻¹, indicating that the MoS₂/RGO composite leverages both high capacity and conductivity.
EIS (Figure 5a) reveals a lower charge‑transfer resistance (R_ct = 10.74 Ω) for MoS₂/RGO compared to MoS₂ (44.07 Ω), underscoring the conductive advantage of the graphene network.
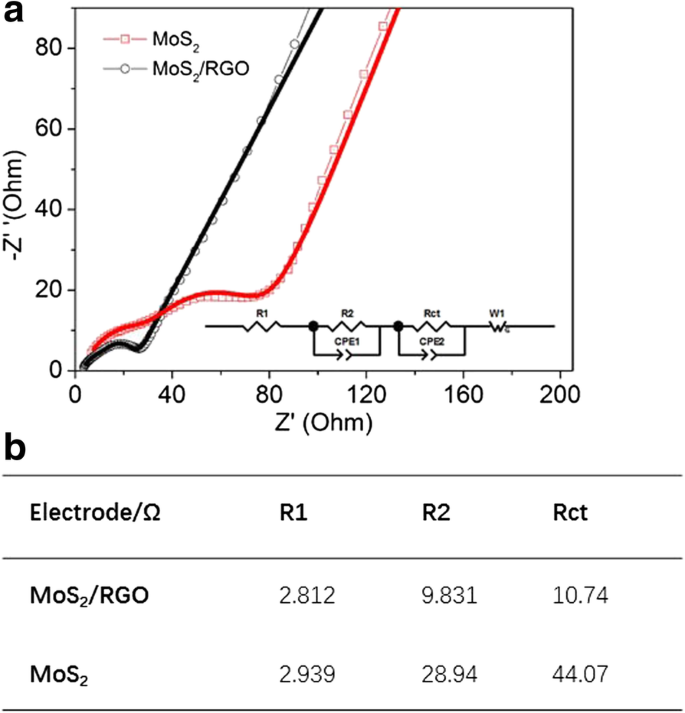
Figure 5. a Nyquist plots after 100 cycles at 100 mA g⁻¹; b Extracted resistances (R1, R2, R_ct) from fitting.
SEM after 100 cycles (Additional file 1: Figure S1) shows the aerogel retains its porous structure with no cracks, whereas pristine MoS₂ exhibits severe cracking due to volume expansion. This confirms the buffering role of the graphene scaffold.
Conclusion
We have fabricated a binder‑free MoS₂/RGO aerogel featuring abundant micropores and a three‑dimensional conductive network. The aerogel delivers an outstanding specific capacity of 1,041 mAh g⁻¹ at 100 mA g⁻¹ and retains 667 mAh g⁻¹ after 100 cycles (58.6 % retention). These results arise from the synergistic interplay between MoS₂ nanosheets, reduced graphene, and the porous architecture, which collectively enhance electron transport, shorten Li⁺ diffusion paths, and accommodate volume changes. This strategy offers a practical route to high‑capacity, long‑life LIB anodes based on transition‑metal dichalcogenide/graphene composites.
Abbreviations
- 2H:
Trigonal prismatic
- CV:
Cyclic voltammograms
- EIS:
Electrochemical impedance spectroscopy
- GCD:
Galvanostatic charge/discharge
- GO:
Graphene oxide
- HRTEM:
High‑resolution TEM
- LIBs:
Lithium‑ion batteries
- MoS₂:
Molybdenum disulfide
- MoS₂/RGO:
MoS₂/reduced graphene
- R1:
Lithium ion migration resistance through the SEI films
- R2:
Resistance of the electrolyte
- Rct:
Charge‑transfer resistance
- SAED:
Selected area electron diffraction
- TMDs:
2D transition metal dichalcogenides
- XRD:
X‑ray diffraction
Nanomaterials
- MoS₂ Films of Precise Thickness Enhance Electrocatalytic Hydrogen Evolution
- Optimizing MoS2‑Graphene Hybrid Crystallization for Enhanced Electrocatalysis
- High-Capacity Few-Layered MoS₂/Acetylene Black Anode for Li‑Ion Batteries
- Hybrid Graphene/WO₃ and Graphene/CeOx Electrodes for High‑Performance Supercapacitors
- Highly Compressible Graphene/Polyaniline Aerogel: Superelasticity Meets 713 F g⁻¹ Capacitance for All‑Solid‑State Supercapacitors
- Polyaniline‑Coated Activated Carbon Aerogel/Sulfur Composite Yields Superior Lithium‑Sulfur Battery Performance
- Secondary Transfer of Graphene Electrodes for Highly Stable Flexible OLEDs
- High‑Performance Flexible Microsupercapacitors Based on 3‑D rGO/PEDOT Open‑Network Architectures
- Binder‑Free Electrospun Fiber Electrodes for High‑Performance Lithium‑Ion Batteries via Simple Rolling Assembly
- Advancing Lithium‑Ion Batteries: Binder‑Free Electrode Technology for Higher Energy Density



