Solid‑State Synthesized PEDOT/Au/GO Composite for Ultra‑Sensitive Electrochemical Detection of Nitrite and Iodate
Abstract
A ternary composite of poly(3,4‑ethylenedioxythiophene)/gold/graphene (PEDOT/Au/GO) was fabricated via a simple solid‑state heating protocol. The composite displays a shale‑like morphology with uniformly dispersed gold nanoparticles, as confirmed by TEM and XRD. Electrochemical studies reveal that the PEDOT/Au/GO‑modified glassy carbon electrode (GCE) exhibits excellent electrocatalytic activity toward both nitrite (NO2−) and iodate (IO3−) detection. Amperometric measurements demonstrate a linear response over 100–1000 µM, with limits of detection (LOD) of 0.53 µM for nitrite and 0.62 µM for iodate (S/N = 3). At 10 µM, the peak currents reach 9.59 µA for nitrite and 11.47 µA for iodate, underscoring the composite’s high sensitivity and rapid response.
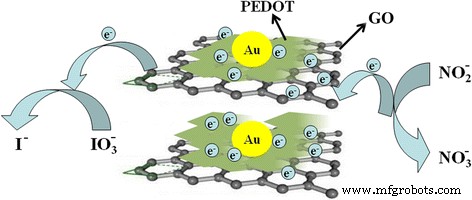
Mechanisms of the direct electron transfer between ion (nitrite or iodate) and the PEDOT/Au/GO composite
Background
Nitrite (NO2−) is a common contaminant in environmental, food, and agricultural matrices, and chronic exposure is linked to the formation of carcinogenic nitrosamines【1–5】. Iodate (IO3−), used in iodized salt, prevents iodine deficiency but excessive intake can cause thyroid disorders【6–7】. Consequently, reliable analytical methods for both anions are essential. While spectroscopic, chromatographic, chemiluminescence, and capillary electrophoresis techniques exist【8–16】, electrochemical sensors offer superior sensitivity, speed, and cost‑effectiveness. Traditional electrodes are often modified with nanostructured metals, metal oxides, or carbon nanomaterials to enhance performance【17–20】. Gold nanoparticles, for instance, provide excellent catalytic activity but suffer from high cost and limited stability【21】. Combining gold with conducting polymers such as PEDOT can overcome these drawbacks by exploiting Au–S interactions that boost electrocatalytic activity【27–28】.
Graphene’s high surface area, exceptional electronic transport, and chemical stability make it an attractive scaffold for ternary composites【31–33】. Recent reports demonstrate that PEDOT/gold/graphene hybrids can outperform binary counterparts in sensing applications【34–35】, yet conventional synthesis routes are often cumbersome. A green, scalable, and efficient solid‑state heating method can fill this gap by enabling the facile integration of PEDOT, gold nanoparticles, and graphene into a single composite.
In this study, we report the solid‑state synthesis of a PEDOT/Au/GO composite and evaluate its electrocatalytic performance for the simultaneous amperometric determination of nitrite and iodate. Comparative analyses with PEDOT and PEDOT/Au confirm the synergistic effect of graphene in enhancing sensitivity and stability.
Experimental
Chemicals and Reagents
3,4‑Ethylenedioxythiophene (EDOT) was obtained from Shanghai Aladdin Reagent Company and purified by distillation. Chloroauric acid tetrahydrate (HAuCl4·4H2O) and graphene oxide (GO) were purchased from Shanghai Aladdin and Strem Chemicals, respectively. All other reagents were analytical grade. 2,5‑Dibromo‑3,4‑ethylenedioxythiophene was synthesized according to literature【36】.
Synthesis of PEDOT/Au/GO and PEDOT/Au Composites
Gold nanoparticles were prepared by reducing HAuCl4 with NaBH4. A typical procedure involved dissolving 60 mg HAuCl4 in 100 mL water, adding 3.4 mL 1 % Na3C6H5O7 under vigorous stirring for 10 min, and then rapidly introducing 1.2 mg NaBH4; the solution instantly turned purple, indicating nanoparticle formation.
For the composite synthesis, 0.5 g (2 mmol) of the monomer and 10 mg GO were dispersed in 30 mL chloroform and ultrasonicated for 30 min to allow monomer adsorption onto GO. The solvent was evaporated, and the residue was ground for 5 min. The powder was then mixed with the gold nanoparticle sol and stirred for 10 min, filtered, washed with distilled water, and finally dried in a vacuum oven at 60 °C for 24 h. The product was designated PEDOT/Au/GO. Binary PEDOT/Au and pure PEDOT were prepared under identical conditions for comparison.
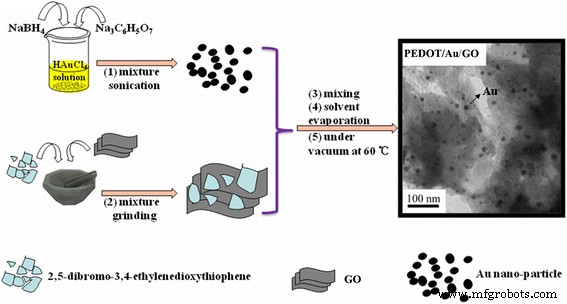
Schematic representation of the formation process of the PEDOT/Au/GO composite
Structure Characterization
Fourier‑transform infrared (FTIR) spectra were recorded on a BRUKER‑QEUINOX‑55 spectrometer using KBr pellets. UV‑vis absorption was measured with a UV‑4802 spectrophotometer. TEM images were obtained on a Hitachi 2600 microscope after depositing a few drops of ethanol suspension onto copper grids. Elemental composition was determined by EDS on a Leo 1430VP microscope (5 kV) with a 200 MPa‑pressed pellet.
Electrochemical Measurements
Cyclic voltammetry (CV) and chronoamperometry were performed on a CHI 660C workstation (three‑electrode setup: Pt counter, SCE reference, PEDOT/Au/GO‑modified GCE working electrode). The GCE (3 mm diameter) was modified by dropping 5 µL of a 30 mg L−1 PEDOT/Au/GO suspension, then air‑dried for 10 min. All experiments were conducted at ambient temperature in air.
Results and Discussion
FTIR spectra (Fig. 2a) confirm the presence of PEDOT functional groups in all samples, with subtle shifts indicating stronger Au–S coordination in the composites. The intensity ratio of the 690 and 830 cm−1 bands suggests that PEDOT/Au/GO has the highest degree of polymerization, followed by PEDOT/Au and pure PEDOT.
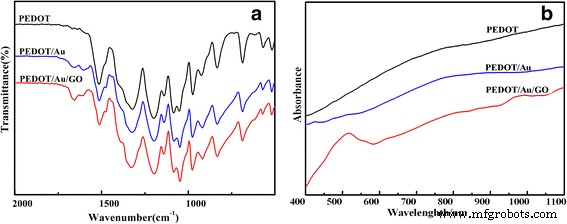
FTIR (a) and UV‑vis (b) spectra of PEDOT, PEDOT/Au, and PEDOT/Au/GO
UV‑vis (Fig. 2b) shows a pronounced free‑carrier tail for PEDOT/Au/GO, indicative of enhanced conjugation and conductivity. TEM images (Fig. 3) reveal a shale‑like layered morphology for PEDOT/Au/GO with uniformly distributed gold nanoparticles (10–15 nm). In contrast, PEDOT/Au exhibits a granular appearance with ~50 nm Au clusters.

TEM images of a PEDOT, b PEDOT/Au, and c PEDOT/Au/GO
XRD patterns (Fig. 4a) display characteristic peaks for PEDOT and GO; the absence of distinct Au peaks in PEDOT/Au/GO is attributed to the high dispersion and small size of the nanoparticles, corroborated by EDX (Fig. 4b) showing 1.71 wt % Au.
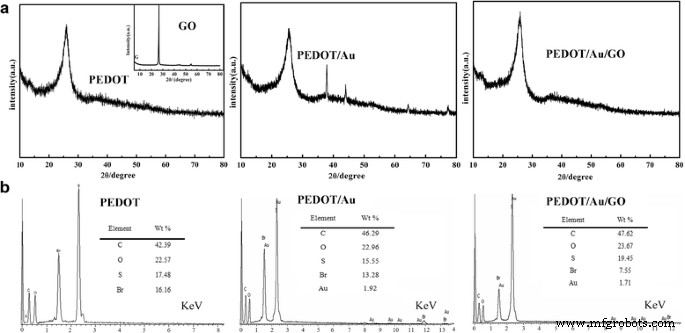
XRD (a) and EDX (b) of PEDOT, PEDOT/Au, and PEDOT/Au/GO
TGA (Fig. 5) demonstrates that PEDOT/Au/GO retains ~36.5 % residue at 800 °C, indicating superior thermal stability conferred by Au and GO.
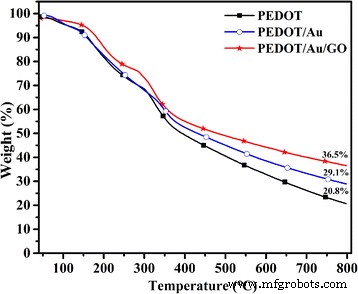
TGA curves of PEDOT, PEDOT/Au, and PEDOT/Au/GO
Electrochemical testing (Fig. 6) shows that the PEDOT/Au/GO‑modified GCE exhibits the highest reduction peak for iodate, confirming its superior catalytic activity compared to PEDOT/Au.
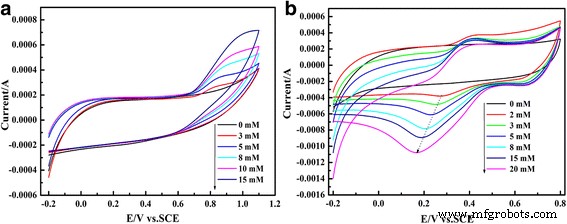
Cyclic voltammograms of PEDOT/Au/GO/GCE and PEDOT/Au/GCE in 0.1 M H2SO4 with 5 mM iodate
CVs in PBS (pH 6.86) and H2SO4 (Fig. 7) display increasing oxidation (nitrite) and reduction (iodate) currents with concentration, demonstrating linear behavior over 3–15 mM (nitrite) and 2–20 mM (iodate).

Cyclic voltammograms of PEDOT/Au/GO/GCE in 0.025 M PBS (pH 6.86) with nitrite (a) and 0.1 M H2SO4 with iodate (b)
Chronoamperometric responses (Fig. 8) illustrate a rapid, step‑wise current increase upon successive addition of nitrite (10−5–10−3 M) at 0.78 V and iodate at –0.25 V.
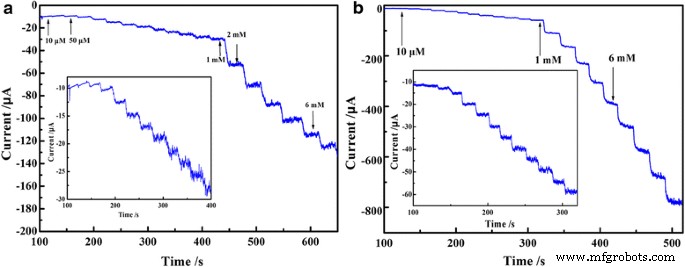
Steady‑state catalytic current–time response of PEDOT/Au/GO/GCE with successive addition of 10−5, 10−4, and 10−3 M nitrite (a) and iodate (b)
Extended chronoamperometry (Fig. 9) confirms a linear relationship between current and concentration in the 100–1000 µM range, with regression equations: I(µA) = 0.0322 C + 26.422 (R² = 0.9995) for nitrite and I(µA) = 0.13757 C + 6.80312 (R² = 0.999) for iodate. The sensor achieves LODs of 0.53 µM (nitrite) and 0.62 µM (iodate), with a response time of ~4 s.
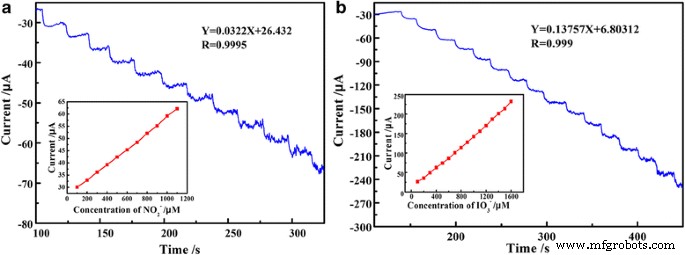
Steady‑state catalytic current–time response of PEDOT/Au/GO/GCE with 10−3 M nitrite (a) and iodate (b)
Stability tests (Fig. 10) demonstrate that the PEDOT/Au/GO/GCE maintains a stable response during a 1000‑s amperometric run for 1 mM analyte, indicating resistance to fouling by reduction products.
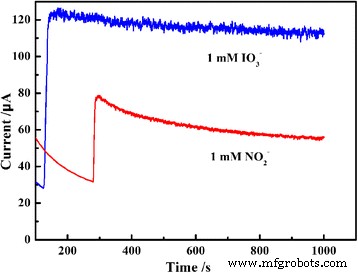
Long‑term amperometry of PEDOT/Au/GO/GCE in 1 mM nitrite (a) and iodate (b) over 1000 s
Mechanistic insight (Fig. 11) suggests that the lamellar PEDOT/GO matrix provides a high‑surface‑area, conductive scaffold that facilitates rapid electron transfer to the underlying GCE, while Au nanoparticles act as catalytic sites for nitrite oxidation and iodate reduction.
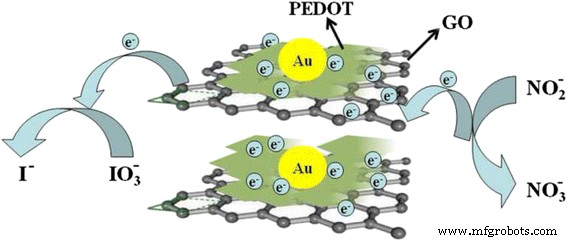
Mechanisms of the direct electron transfer between ion (nitrite or iodate) and GCE through the PEDOT/Au/GO composite
Real Sample Analysis
To validate practical applicability, the PEDOT/Au/GO/GCE was employed for nitrite determination in tap water using a standard‑addition approach. Recoveries ranged from 98.4 % to 104.3 %, confirming the sensor’s accuracy in real matrices.
Conclusion
We have successfully developed a solid‑state synthesized PEDOT/Au/GO composite that combines the high conductivity of PEDOT, the catalytic prowess of Au nanoparticles, and the large surface area of graphene. The resulting shale‑like structure promotes uniform Au dispersion and facilitates rapid electron transfer, yielding a sensor with low limits of detection (0.53 µM for nitrite, 0.62 µM for iodate) and fast response times (~4 s). The composite’s robust performance in real tap‑water samples demonstrates its promise for field‑ready monitoring of nitrite and iodate contaminants.
Nanomaterials
- Cellulose–POSS–Silica/Gold Core–Shell Hybrid Nanocomposites: One‑Step Sol‑Gel Synthesis and Multifunctional Properties
- Fabricating Electrically Conductive Silica Nanofiber–Gold Nanoparticle Composites via Laser Pulses and Sputtering
- Hybrid Graphene/WO₃ and Graphene/CeOx Electrodes for High‑Performance Supercapacitors
- A Recyclable Magnetic‑Silica Hybrid Probe for Fe³⁺ Detection and Live‑Cell Imaging
- Eco‑Friendly Co₃O₄ Nanowires with Graphene: A High‑Performance Anode for Lithium‑Ion Batteries
- Green Synthesis of Gold and Silver Nanoparticles with Platycodon Saponins from Platycodi Radix
- Heparanase‑Targeted Magnetic Gold Nanoparticle Probe Enhances MRI Detection of Tumor Metastasis
- Low‑Cost Hydrothermal Synthesis of Ultralong Copper Nanowires for Flexible Transparent Conductive Electrodes
- High‑Performance Flexible Microsupercapacitors Based on 3‑D rGO/PEDOT Open‑Network Architectures
- Efficient One-Step Green Synthesis of Multifunctional Gold Nanoparticles for Targeted Tumor Imaging and Therapy



