Low‑Cost Hydrothermal Synthesis of Ultralong Copper Nanowires for Flexible Transparent Conductive Electrodes
Abstract
A facile hydrothermal approach has been developed to produce ultralong, thin copper nanowires (CuNWs) with an average diameter of 35 nm and a length of 100 µm. The synthesis uses inexpensive, non‑toxic precursors: copper(II) chloride dihydrate (CuCl2·2H2O), octadecylamine (ODA) as capping agent, and ascorbic acid as reducing agent. Reaction parameters such as time and molar ratios were systematically optimized to achieve the desired morphology. The resulting CuNWs were directly transferred onto polyethylene terephthalate (PET) substrates by vacuum filtration, yielding flexible transparent conductive electrodes (TCEs) with a sheet resistance of 26.23 Ω/□ and a transmittance of 89.06 % at 550 nm (excluding PET). A subsequent spin‑coating of poly(methyl methacrylate) (PMMA) reduced surface roughness and markedly improved oxidation resistance, forming hybrid TCEs (HTCEs) with superior chemical stability.
Background
Transparent conductive electrodes (TCEs) are essential components in a wide range of optoelectronic devices, including organic light‑emitting diodes (OLEDs), solar cells, liquid crystal displays, and flexible sensors. Indium tin oxide (ITO) has long dominated the market because of its low resistivity (≈ 10–30 Ω/□) and high transparency (≈ 90 %). However, indium’s scarcity drives up ITO costs, motivating the search for alternatives that are cheaper, more abundant, and compatible with low‑temperature processing. Metallic nanowires, particularly silver (AgNWs) and copper (CuNWs), have emerged as promising candidates due to their excellent electrical conductivity and tunable optical properties. While AgNWs can rival ITO in performance, silver’s high cost and limited supply make copper an attractive substitute. Copper’s bulk resistivity (1.67 nΩ·m) is only slightly higher than silver’s (1.59 nΩ·m), yet copper is far more abundant and inexpensive. Consequently, research efforts have intensified to produce high‑quality CuNWs and integrate them into flexible TCEs.
Numerous synthesis routes have been explored for CuNWs, including chemical vapor deposition, electrochemical deposition, template methods, and membrane processes. These approaches often involve complex steps, toxic reagents, or expensive catalysts. Hydrothermal synthesis offers a simpler, scalable alternative, allowing CuNWs to grow from aqueous solutions under moderate temperatures and pressures. Recent studies have demonstrated CuNWs with diameters ranging from 30 to 100 nm and lengths spanning from several micrometers to millimeters, depending on the choice of reducing and capping agents. Despite these advances, CuNW‑based TCEs still face challenges such as high surface roughness, susceptibility to oxidation, and limited stability over time. Strategies to mitigate these issues include incorporating high‑aspect‑ratio nanowires and embedding them in protective polymer matrices.
In this work, we report a hydrothermal method that produces ultralong, thin CuNWs with a high aspect ratio (~ 2857) using cheap, non‑toxic reagents. We systematically investigate the influence of reaction time and reagent ratios on nanowire morphology. The CuNWs are then transferred onto PET substrates via vacuum filtration, forming flexible TCEs with record low sheet resistance and high transparency without post‑treatment. Finally, we demonstrate that coating the CuNW network with PMMA reduces surface roughness and enhances oxidation resistance, yielding hybrid TCEs with superior performance.
Experimental
Synthesis of CuNWs
In a typical experiment, 140 mg of ascorbic acid (C6H8O6) and 270 mg of CuCl2·2H2O were dissolved in 282 mL of an ODA aqueous solution (26.3 mmol L−1). The resulting mixture was stirred at room temperature for 60 min to form a homogeneous suspension. This suspension was then transferred to a Teflon‑lined autoclave and sealed at 120 °C for 20 h. After cooling to room temperature, excess reagents were removed by washing with deionized water and ethanol. The final CuNWs were stored in 130 mL of glacial acetic acid to prevent oxidation.
Fabrication of CuNW TCEs and CuNW/PMMA HTCEs
CuNW TCEs were fabricated on 188 µm thick PET substrates. A small volume of the CuNW dispersion in glacial acetic acid was diluted with 500 mL of deionized water and filtered through a 0.45 µm mixed cellulose ester (MCE) membrane. The deposited film was then transferred onto the PET by applying uniform pressure and peeling off the MCE membrane. For the hybrid electrodes, 100 µL of a 20 mg mL−1 PMMA solution was spin‑coated onto the CuNW film at 800 rpm for 5 s and 2500 rpm for 30 s, then air‑dried without additional thermal treatment.
Structural, Optical, and Electrical Characterization
Nanowire morphology and dimensions were examined by SEM (JSM‑7500F, JEOL) and TEM (FEI‑TECNAL G20). Optical microscopy (BX51M, Olympus) provided surface images. Transmittance spectra were measured with a UV‑vis spectrophotometer (GZ502A). Surface roughness was assessed by atomic force microscopy (AFM, Dimension Edge, Bruker). Powder X‑ray diffraction (XRD, Bruker) identified crystal phases. Sheet resistance was determined from four‑probe measurements using a two‑probe method on aluminum contacts; the relationship Rs = R·W/L (Eq. 1) was applied.
$$R_{s}=R rac{W}{L}$$
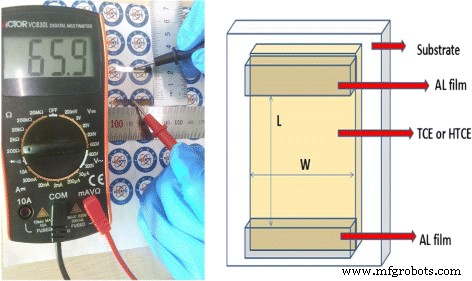
Photographic image and schematic illustration of the resistance measurement setup. Left: photo of the test; right: diagram of length L and width W of the TCE.
Results and Discussion
Synthesis of CuNWs
The hydrothermal method yielded CuNWs with an average diameter of 35 nm and a length of 100 µm, giving an aspect ratio of ~ 2857. ODA served as the capping agent, while ascorbic acid reduced Cu2+ to metallic Cu. The resulting nanowires were uniform, highly crystalline, and free of copper oxides, as confirmed by XRD and HRTEM.
(A) Characterization of CuNWs
Figure 2a shows a photograph of the CuNW dispersion. SEM images (Figures 2b and 2c) reveal densely packed, straight nanowires with the reported dimensions. The XRD pattern (Figure 2d) displays sharp peaks at 43.316°, 50.448°, and 74.124°, corresponding to the {111}, {200}, and {220} planes of face‑centered cubic copper, confirming phase purity and preferential {111} growth. TEM images (Figure 3a–b) further illustrate the smooth surfaces and uniform diameters, while HRTEM (Figure 3c) shows a lattice spacing of 0.21 nm matching the {111} plane.
(B) Time‑Dependent Analysis
SEM images (Figure 4) illustrate the evolution of the CuNW morphology with hydrothermal time. At 1 h, the product consists mainly of copper nanocubes due to insufficient ODA coverage. Between 2–14 h, the nanowires grow rapidly, reaching lengths of 80 µm by 14 h. Beyond 20 h, growth plateaus, and the nanowires maintain their dimensions, indicating that the reaction is largely complete after 20 h.
(C) Amount of Ascorbic Acid
Adjusting the ascorbic acid to CuCl2·2H2O ratio (maintaining constant ODA and water) revealed that a 0.5:1 molar ratio produces the highest yield of CuNWs with minimal copper nanoparticles. Ratios of 1:1 or 2:1 result in increased CuNP formation, while a 0.2:1 ratio yields few nanowires due to limited reducing power. This confirms that an optimal reducing‑to‑copper ratio is essential for selective nanowire growth.
Fabrication of CuNW TCE
The vacuum‑filtration transfer method produced flexible TCEs with tunable conductivity and transparency. Table 1 (not shown) lists sheet resistances and transmittances for five samples. A trade‑off between sheet resistance and transmittance is observed: the lowest sheet resistance (26.23 Ω/□) corresponds to a transmittance of 89.06 % at 550 nm. Figure 7 (photograph of sample C) confirms the high optical quality of the electrode.
Fabrication of CuNW/PMMA HTCE
Coating the CuNW network with PMMA dramatically reduces surface roughness from 31.2 nm (RMS) to 4.8 nm (RMS) and suppresses rapid resistance increase under ambient storage. While the sheet resistance of the bare CuNW TCE rises from 32.1 Ω/□ to 93.5 Ω/□ within 3 h, the PMMA‑coated HTCE remains stable, reaching only 74 Ω/□ after 72 h. The improved chemical stability arises from the PMMA layer blocking moisture and oxygen ingress.
Conclusions
We have developed a simple, low‑cost hydrothermal route to produce ultralong, thin CuNWs (35 nm × 100 µm) using non‑toxic reagents. The resulting CuNWs enable fabrication of flexible TCEs with a sheet resistance of 26.23 Ω/□ and 89.06 % transmittance at 550 nm without any post‑treatment. A subsequent PMMA coating further lowers surface roughness and enhances oxidation resistance, yielding hybrid TCEs with superior performance.
Nanomaterials
- Efficient Visible‑Light Photocatalysis via WS₂/Bi₂MoO₆ Heterostructures: A Simple Solvothermal Approach
- High‑Performance Flexible Transparent Electrodes from Silver Nanowires with Tailored Aspect Ratios
- Controlled Hydrothermal Synthesis of ZnO Nanocrystals for Enhanced Inverted Polymer Solar Cells
- Solid‑State Synthesized PEDOT/Au/GO Composite for Ultra‑Sensitive Electrochemical Detection of Nitrite and Iodate
- Eco‑Friendly Co₃O₄ Nanowires with Graphene: A High‑Performance Anode for Lithium‑Ion Batteries
- Ligand‑Free Iridium Nanoparticles: A Simple Aqueous Synthesis and Demonstrated In‑Vitro Biocompatibility
- Gold Nanomesh Electrodes: Flexible, Transparent, and Highly Conductive for Advanced Electronics
- High-Performance Recyclable Starch‑Ag Networks for Transparent Conductive Electrodes in Flexible Joint Sensors
- Comprehensive Review of Luminescent Silica Nanoparticles: Classification, Synthesis, and Practical Applications
- Ultra‑Sensitive Paper‑Based Capacitive Flexible Pressure Sensor for Wearable and Artificial‑Skin Applications



