Eco‑Friendly Co₃O₄ Nanowires with Graphene: A High‑Performance Anode for Lithium‑Ion Batteries
Abstract
We present a green, scalable route to synthesize Co₃O₄ nanowires (NWs) via a two‑step process: magnetic‑field‑assisted assembly of Co nanowires (CoNWs) followed by gentle air oxidation. The resulting 1‑D NWs, 3–8 µm long with aspect ratios exceeding 15, combine a high crystalline quality with a large specific surface area. When paired with only 3 wt % defect‑free graphene (df‑GNS), the Co₃O₄ NW/graphene composite delivers a reversible lithium‑storage capacity of ~790 mAh g⁻¹ and exhibits excellent rate capability, underscoring its promise as a next‑generation LIB anode.
Background
As global demand for clean, high‑energy‑density storage escalates, lithium‑ion batteries (LIBs) remain the benchmark technology due to their high specific energy, long cycle life, and low cost. Transition‑metal oxides, particularly Co₃O₄, offer theoretical capacities (~890 mAh g⁻¹) that surpass graphite, but their practical performance is limited by poor electronic conductivity, pronounced volume expansion, and low utilization [1–4].
One‑dimensional nanostructures—nanowires, nanobelts, and nanofibers—provide intrinsic advantages such as high surface area, short ion‑transport paths, and structural flexibility to accommodate volume changes [5–10]. However, conventional synthesis methods (hydrothermal, template‑based, wet‑chemical reduction) often involve harsh conditions, expensive templates, or corrosive reagents, hampering scalability and environmental friendliness [11–16].
Herein, we introduce a facile, environmentally benign synthesis of Co₃O₄ NWs that avoids high‑temperature, high‑pressure, or hazardous processes. The resulting NWs, integrated with df‑GNS, form a 1D–2D hybrid architecture that enhances electron transport and mitigates mechanical degradation during cycling.
Experimental
Materials
All reagents were analytical grade: CoCl₂·6H₂O (99 wt%), EDTA‑2Na (99 wt%), NaOH (98 wt%), NaCl/KCl (99.5 wt%), PVP‑K30 (Mₙ ≈ 29–35 kDa), hydrazine monohydrate (80 vol%), and H₂PtCl₆·6H₂O (38 wt%). De‑ionized water (DI) was used for all solutions. Commercial Co₃O₄ nanoparticles (Co₃O₄NPs) served as a control.
Synthesis of CoNWs
In a 100 mL PTFE beaker, 0.6 mmol CoCl₂·6H₂O and 0.6 mmol EDTA‑2Na were dissolved in 60 mL DI water. The pH was adjusted to 14 with dilute NaOH, and 0.15 g PVP was added under vigorous stirring. The solution was placed between two permanent magnets (35 mT) in an 80 °C water bath. Upon reaching the target temperature, 0.30 mL hydrazine and 0.12 mL of 0.0253 M H₂PtCl₆·6H₂O were introduced, initiating rapid Co²⁺ reduction. CoNWs formed within 10 min, were magnetically harvested, and washed repeatedly with ethanol and DI water before vacuum‑freeze drying for 12 h.
Synthesis of Co₃O₄ NWs
The dried CoNWs (0.01 g) were mixed with 0.3 g KCl and 0.2 g NaCl, then calcined in a muffle furnace at 650 °C for 4 h. After natural cooling, the product was washed thrice with DI water and dried at 80 °C for 4 h. The transformation from CoNWs to Co₃O₄ NWs is illustrated in Figure 1.
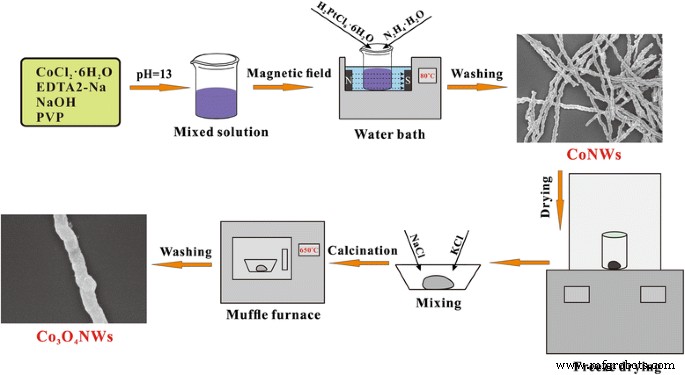
Schematic illustration of Co₃O₄ NW synthesis.
Characterization
Structural Analysis
X‑ray diffraction (Cu Kα, λ = 0.1542 nm) confirmed phase purity and high crystallinity of CoNWs (fcc Co) and Co₃O₄ NWs (cubic Co₃O₄) with lattice parameters a = b = c = 8.084 Å. Scherrer analysis yielded crystallite sizes of 18.7 nm (CoNWs) and 25.4 nm (Co₃O₄ NWs), indicating multi‑grain structures within each wire. SEM revealed uniform 150 nm‑diameter CoNWs up to 20 µm long; post‑oxidation NWs maintained ~180 nm diameter and wire‑like morphology (Figure 3).

SEM images of Co₃O₄NPs (a), CoNWs (b), and Co₃O₄ NWs (c, d).
Microstructural Imaging
TEM and HRTEM confirmed the wire morphology and crystalline planes: CoNWs displayed (111) and (220) planes, while Co₃O₄ NWs showed (111), (211), (220), (311), (440), and (511) planes with lattice spacings matching standard data. SAED patterns corroborated XRD results, confirming phase purity (Figure 4).

TEM, SAED, and HRTEM images of Co₃O₄NPs (a), CoNWs (b), and Co₃O₄ NWs (c). Insets show corresponding SAED patterns.
Electrochemical Measurements
Half‑coin cells (CR2025) were assembled in an Ar glovebox (<0.1 ppm H₂O/O₂). Working electrodes consisted of Co₃O₄ NWs or Co₃O₄NPs mixed with 3 wt % df‑GNS (weight ratio 100:3) and coated onto Cu foil; active loading was 0.5–1 mg per cell. The electrolyte was 1 M LiPF₆ in EC/PC/DEC (1:1:1 v/v/v). Galvanostatic charge–discharge tests were performed between 0.01–3 V using a LAND 2001A system. CVs were recorded at 0.5 mV s⁻¹, and EIS measurements spanned 100 kHz–0.1 Hz (5 mV amplitude).
Results and Discussion
The XRD patterns (Figure 2) confirm phase purity of all samples. SEM/TEM images (Figures 3–4) illustrate the successful synthesis of uniform Co₃O₄ NWs that retain their morphology after calcination. The high aspect ratio (≥15) and smooth surface facilitate efficient electron/ion transport and mitigate mechanical stress during cycling.
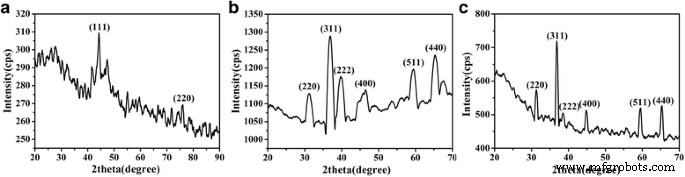
XRD patterns of CoNWs (a), Co₃O₄ NWs (b), and Co₃O₄NPs (c).
Electrochemical performance (Figure 6) shows that the Co₃O₄ NW/df‑GNS electrode delivers ~790 mAh g⁻¹ after 20 cycles at 50 mA g⁻¹, with negligible capacity fade. In contrast, Co₃O₄NPs/df‑GNS exhibit a high initial discharge capacity (~1130 mAh g⁻¹) that collapses to ~400 mAh g⁻¹ by cycle 2 due to irreversible SEI formation and volume‑induced pulverization. The NWs maintain >650 mAh g⁻¹ even at 200 mA g⁻¹ and >400 mAh g⁻¹ at 1000 mA g⁻¹, highlighting superior rate capability.
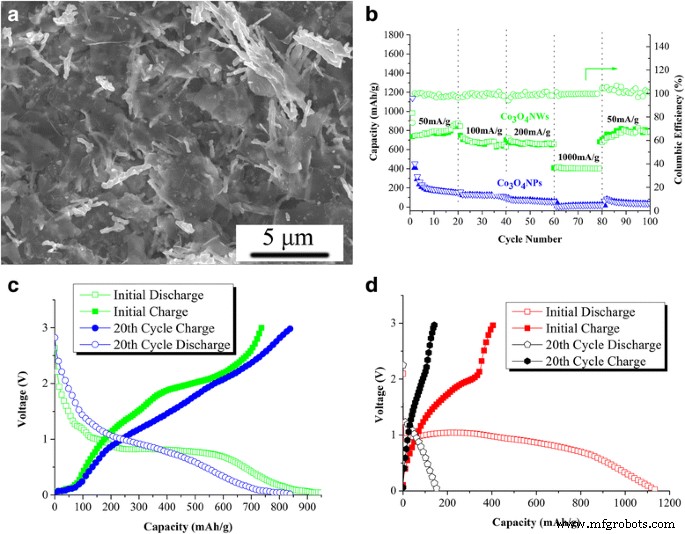
SEM image of Co₃O₄ NW/df‑GNS electrode (a); rate performance comparison (b); first and 20th cycle charge–discharge curves for NWs (c) and NPs (d).
CV curves (Figure 7a) display characteristic cathodic peaks at ~1.1 V and ~0.4 V, corresponding to multi‑step Co₃O₄ reduction to metallic Co and Li₂O. The anodic peak at ~2.2 V confirms reversible Co₃O₄ re‑oxidation. EIS analysis (Figure 7b) reveals a charge‑transfer resistance (R_ct) of 52.6 Ω for NW/df‑GNS versus 109 Ω for NP/df‑GNS, indicating superior electronic conductivity of the 1D–2D hybrid.
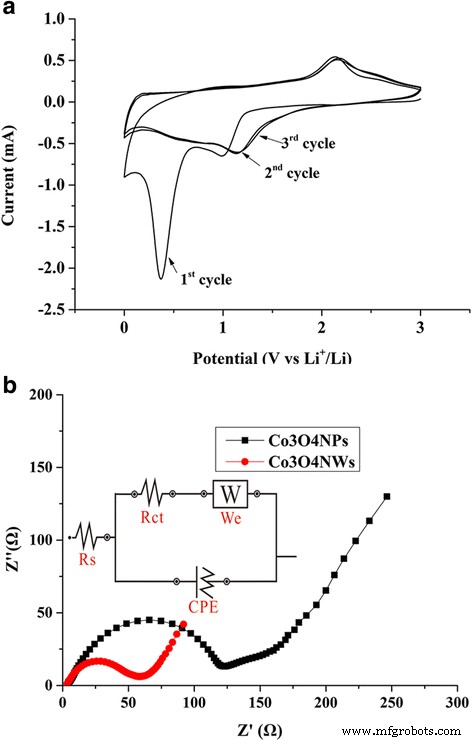
CV (a) and EIS (b) of Co₃O₄ NW/df‑GNS and Co₃O₄NP/df‑GNS electrodes.
Conclusions
We have demonstrated a green, scalable synthesis of Co₃O₄ nanowires that, when combined with a minimal amount of defect‑free graphene, delivers exceptional lithium‑ion storage performance (>700 mAh g⁻¹) and robust rate capability. The 1D–2D hybrid architecture synergistically enhances electronic conductivity, accommodates volume changes, and prevents self‑aggregation, making Co₃O₄ NW/graphene an attractive anode candidate for next‑generation LIBs.
Abbreviations
- Co₃O₄
Cobalt oxide
- Co₃O₄NPs
Co₃O₄ nanoparticles
- Co₃O₄NWs
Co₃O₄ nanowires
- CV
Cyclic voltammograms
- DEC
Diethyl carbonate
- df‑GNS
Defect‑free graphene nanosheets
- EC
Ethylene carbonate
- EIS
Electrochemical impedance spectroscopy
- GNS
Graphene nanosheets
- HRTEM
High‑resolution TEM
- LIBs
Lithium‑ion batteries
- NMP
N‑methyl‑pyrrolidone
- PC
Propylene carbonate
- SAED
Selected area electron diffraction
- SEI
Solid electrolyte interface
- SEM
Scanning electron microscope
- TEM
Transmission electron microscopy
- XRD
X‑ray diffraction analysis
Nanomaterials
- LiNi0.5Mn1.5O4 Cathodes with Cr3+ and F− Composite Doping: Synthesis, Stability, and Electrochemical Performance
- One‑Pot Synthesis of Color‑Tunable, Conductive CuS‑Coated CuSCN Composites
- High‑Performance Flexible Transparent Electrodes from Silver Nanowires with Tailored Aspect Ratios
- Efficient One‑Step Photo‑Ultrasonic Synthesis of rGO/Ag₃PO₄ Quantum‑Dot Composites for Enhanced Visible‑Light Photocatalysis
- Optimized Fe³⁺‑Grafted BiOCl for Rapid Adsorption of Trace Cationic and Anionic Dyes in Aqueous Wastewater
- Low‑Cost Hydrothermal Synthesis of Ultralong Copper Nanowires for Flexible Transparent Conductive Electrodes
- Ligand‑Free Iridium Nanoparticles: A Simple Aqueous Synthesis and Demonstrated In‑Vitro Biocompatibility
- CuGeO3 Nanowires: A High‑Capacity, Stable Anode for Advanced Sodium‑Ion Batteries
- FeF3·0.33H2O Cathode Enhanced by CNTs and Graphene: A High‑Performance Solution for Lithium‑Ion Batteries
- Advancing Lithium‑Ion Batteries: Binder‑Free Electrode Technology for Higher Energy Density



