One‑Pot Synthesis of Color‑Tunable, Conductive CuS‑Coated CuSCN Composites
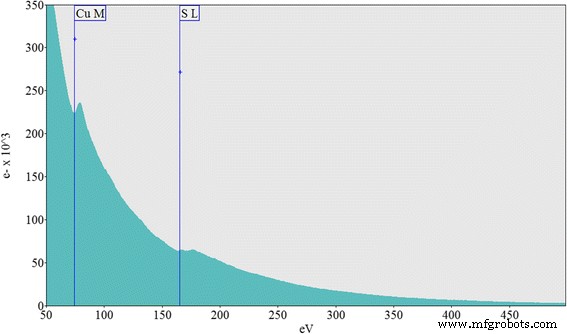
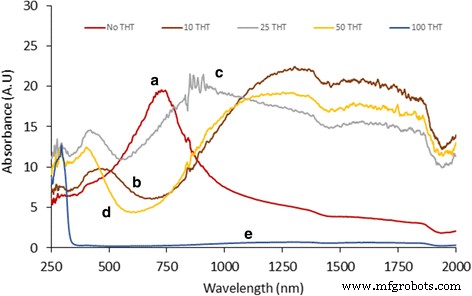
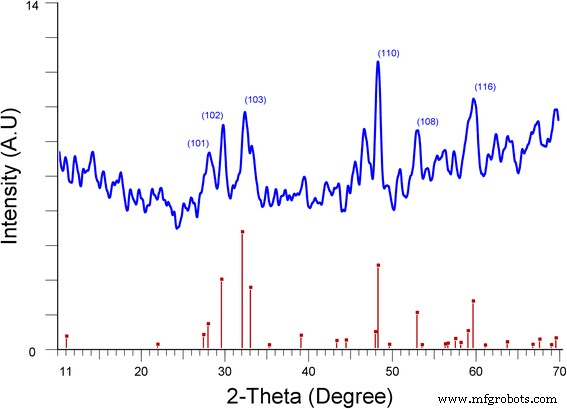
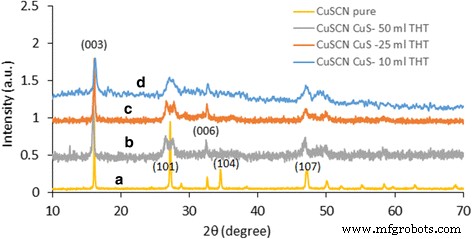
We report a simple, ambient‑temperature, one‑pot method for producing CuS nanoparticle‑coated CuSCN composites with tunable color and conductivity. By mixing copper sulfate, sodium thiosulfate, and triethyl amine hydrothiocyanate (THT) in a 1:1:1 molar ratio, a white‑gray CuSCN phase forms immediately. Removing THT yields dark blue CuS microparticles, whereas adding varying amounts of THT generates colored, conducting composites. CuS nanoparticles do not appear immediately; they form over ~12 h, as confirmed by TEM, which shows hexagonal CuS particles 3–10 nm in diameter. Remarkably, the composites exhibit higher conductivity than either pure CuS or CuSCN. Infrared absorption is also stronger in the coated composites. The lowest measured resistivity, 0.05 Ω cm, was obtained for annealed (250 °C) CuS‑coated CuSCN prepared with 10 ml THT under nitrogen. This scalable strategy can be extended to coat CuS onto other nanomaterials such as metal oxides, polymers, and metal nanoparticles. Nanostructured materials are prized for unique optical, electrical, and mechanical properties that bulk materials cannot provide. Copper sulfide (CuS) is especially attractive because its stoichiometry ranges from Cu₂S to CuS₂, and its p‑type, small‑band‑gap semiconducting behavior makes it a candidate for photovoltaics, field‑emission devices, and Li‑ion batteries. CuS (covellite) is highly conductive and even exhibits type‑I superconductivity at 1.6 K. Its versatility has led to applications in photocatalysis, photovoltaics, cathodes, supercapacitors, and Li‑ion batteries. Various morphologies—nanowires, nanodisks, hollow spheres, and flower‑like structures—have been fabricated, predominantly by hydrothermal routes. Previous work has demonstrated CuS‑based composites such as CuS nanoflowers on reduced graphene oxide for lithium storage, CuS/ZnS hollow spheres, CuS‑coated ZnO rods for piezo‑photocatalysis, and CuS‑coated activated carbon for dye removal. However, a straightforward, scalable synthesis of CuS‑coated CuSCN with color and conductivity tunability remains unexplored. In this study, we employ copper sulfate, sodium thiosulfate, and THT at room temperature to generate CuS‑coated CuSCN particles. By adjusting THT volume, we control the composite’s color and electrical properties. CuSCN, a stable p‑type semiconductor with a 3.6 eV bandgap, is chosen to match the p‑type nature of CuS. The method is also applicable to other nanomaterials, as demonstrated with CuS‑coated TiO₂ composites. All reagents—sodium thiosulfate pentahydrate, copper(II) sulfate, triethyl amine, and ammonium thiocyanate—were purchased from Sigma‑Aldrich and used without further purification. THT was synthesized according to our previous protocol. A 0.1 M solution of copper sulfate (100 ml) was mixed with an equal volume of 0.1 M sodium thiosulfate. After 30 min stirring, aliquots of 0.1 M THT (0–100 ml) were added dropwise. The mixture was stirred overnight at ambient conditions, then the precipitate was collected by centrifugation and washed repeatedly with deionized water before characterization. Morphology was examined using a Hitachi SU6600 SEM and a JEOL JEM‑2100 HRTEM. Elemental composition was confirmed by EELS (GATAN 963). Powder X‑ray diffraction (XRD) employed a Bruker D‑8 Focus with Cu‑Kα radiation (λ = 0.15418 nm). UV‑Vis and diffuse reflectance spectra were recorded on a Shimadzu UV‑3600 NIR spectrometer. Mixing equal volumes (100 ml) of 0.1 M copper sulfate and sodium thiosulfate produced a blue precipitate after an overnight reaction. Initially, the solution is light green with no solid. TEM shows a mixture of large, spherical microparticles and smaller nanoparticles. Adding THT (<0.1 M, 100 ml) immediately generates white CuSCN, which turns light brown as CuS nanoparticles nucleate on its surface. By varying THT volume from 0 to 100 ml, the composite’s color evolves, as illustrated in Fig. 2. With 100 ml THT, only gray CuSCN forms; without THT, only dark blue CuS appears. Intermediate THT volumes produce distinct colored composites, demonstrating the tunability of the process. SEM images: a) pure CuS, b) CuS‑coated CuSCN with 10 ml THT, c) CuS‑coated CuSCN with 25 ml THT, d) CuS‑coated CuSCN with 50 ml THT Thin‑film optical images: a) CuS (0 THT), b) CuS‑coated CuSCN (10 ml THT), c) CuS‑coated CuSCN (25 ml THT), d) CuS‑coated CuSCN (50 ml THT), e) CuSCN only (100 ml THT) Figure 1 reveals that CuS‑coated CuSCN particles exhibit uniform distribution of CuS nanoparticles, whereas bulk CuS consists mainly of microscale spheres. TEM confirms CuS nanoparticles (3–10 nm) embedded in a CuSCN matrix (Fig. 3). Sonication in ethanol separates CuS from CuSCN, resulting in a colored solution (Additional file 1: Figure S2). EELS detects only Cu (74 eV) and S (165 eV) peaks, confirming the identity of the nanoparticles (Fig. 4). TEM images: a) CuS nanoparticles, b) bulk CuSCN crystals in the composite (10 ml THT) EELS spectrum of a hexagonal CuS nanoparticle in the composite (10 ml THT) UV‑Vis absorption data (Fig. 5) show that pure CuS peaks at ~735 nm, while pure CuSCN shows negligible visible absorption due to its 3.6 eV bandgap. CuS‑coated CuSCN composites exhibit broad absorption across the visible and infrared (up to 1900 nm). The 10 ml THT composite displays the strongest IR absorption and a visible peak at 465 nm; increasing THT to 25 ml and 50 ml shifts the visible peak to 425 nm and 410 nm, respectively, and slightly reduces IR intensity. Absorbance spectra: a) pure CuS (no THT), b) CuS‑coated CuSCN (10 ml THT), c) CuS‑coated CuSCN (25 ml THT), d) CuS‑coated CuSCN (50 ml THT), e) pure CuSCN (100 ml THT) XRD patterns (Fig. 6) confirm that the dark blue sample is covellite CuS (JCPDS 03‑065‑3561). CuS‑coated CuSCN samples (Fig. 7) show overlapping peaks of CuS and CuSCN; weak CuS reflections are expected due to the 3–10 nm particle size and limited crystallinity, consistent with literature reports of amorphous‑like patterns for similarly sized CuS nanoparticles. XRD spectrum of CuS prepared without THT XRD spectra: a) pure CuSCN (100 ml THT), b) CuS‑coated CuSCN (50 ml THT), c) CuS‑coated CuSCN (25 ml THT), d) CuS‑coated CuSCN (10 ml THT) Electrical resistivity was measured on thin films deposited by the doctor‑blade method onto Cr/Pt‑sputtered glass electrodes. Pure CuS films exhibit high resistance, whereas CuS‑coated CuSCN films display resistivities in the 0.05–0.16 Ω cm range. Annealing the 10 ml THT composite at 250 °C for 20 min under nitrogen reduces resistance by 68 % (from 15.8 Ω to 5 Ω), attributable to enhanced crystallinity and interparticle connectivity (Additional file 1: Figure S3). We have demonstrated a versatile, ambient‑temperature synthesis of color‑tunable, conductive CuS‑coated CuSCN composites by adjusting THT volume. The composites absorb across the visible and infrared spectra and achieve a minimum resistivity of 0.05 Ω cm after annealing. The method is readily adaptable to other nanomaterials, offering a scalable route to CuS‑based nanocomposites with tailored optical and electrical properties.Abstract
Background
Methods
Materials
Synthesis of Nano‑CuS‑Coated CuSCN
Characterization
Results and Discussion



Conclusions
Nanomaterials
- Efficient Synthesis and Optical Characterization of Sub‑Micron Selenium Nanocrystals and Nanorods
- Sol‑Gel Fabricated SiO₂@C/MWNT Nanocomposites Deliver Superior Li‑Ion Battery Anodes
- High‑Performance Flexible Transparent Electrodes from Silver Nanowires with Tailored Aspect Ratios
- Rapid Fabrication of Hierarchical Porous Polyaniline/Polyurethane Sponge Composites for Flexible Pressure and Tunable Gas Sensors
- Eco‑Friendly Co₃O₄ Nanowires with Graphene: A High‑Performance Anode for Lithium‑Ion Batteries
- Ligand‑Free Iridium Nanoparticles: A Simple Aqueous Synthesis and Demonstrated In‑Vitro Biocompatibility
- Green Synthesis of MnO₂ Nanoparticles on Eggshell Membrane for Rapid Tetracycline Decontamination
- Comprehensive Review of Luminescent Silica Nanoparticles: Classification, Synthesis, and Practical Applications
- Reducing Cytotoxicity of Silver Nanoparticle Coatings with Graphene Oxide: Enhanced Antibacterial Performance and Biocompatibility of an Ag–GO Nanocomposite
- Biotin‑Functionalized Redox‑Sensitive Chitosan Nanoparticles Coated with Phycocyanin for Targeted Curcumin Delivery



